Signal-to-Noise Ratio in Neural Recordings: A Comprehensive Comparison of EEG, ECoG, and Intracortical Technologies
This article provides a systematic analysis of the signal-to-noise ratio (SNR) across the primary neural recording modalities: electroencephalography (EEG), electrocorticography (ECoG), and intracortical microelectrodes.
Signal-to-Noise Ratio in Neural Recordings: A Comprehensive Comparison of EEG, ECoG, and Intracortical Technologies
Abstract
This article provides a systematic analysis of the signal-to-noise ratio (SNR) across the primary neural recording modalities: electroencephalography (EEG), electrocorticography (ECoG), and intracortical microelectrodes. Aimed at researchers and drug development professionals, we explore the fundamental biophysical principles governing SNR, detail methodological advances for its enhancement in applications from basic science to brain-computer interfaces (BCIs), and address critical troubleshooting and optimization strategies. The content validates these approaches through direct comparative studies and discusses the implications of SNR performance for the accuracy of neural decoding, particularly in clinically relevant domains such as speech prostheses and somatosensory mapping. By synthesizing foundational knowledge with current technological frontiers, this review serves as a vital resource for selecting, optimizing, and validating neural recording methodologies.
Fundamental Principles and Biophysical Trade-offs in Neural Recording SNR
In electrophysiology, the signal-to-noise ratio (SNR) serves as a fundamental quantitative metric for evaluating the fidelity of recorded neural signals. It compares the power of meaningful biological information—such as action potentials, local field potentials (LFPs), or evoked responses—against the power of background interference. Achieving a high SNR is critical for distinguishing genuine neural activity from artifacts, thereby enabling accurate interpretation of brain function across research and clinical applications. The specific definitions and calculations of SNR vary significantly between different recording modalities, including electroencephalography (EEG), electrocorticography (ECoG), and intracortical microelectrode arrays (MEAs), each presenting unique trade-offs between invasiveness and signal quality [1].
This guide provides a systematic comparison of SNR definitions, methodologies, and performance across these primary electrophysiological recording techniques. We synthesize key metrics and formulas, supported by experimental data and detailed protocols, to inform researchers, scientists, and drug development professionals in selecting and optimizing neural interfaces for specific applications.
Core Mathematical Definitions of SNR
The foundational definition of SNR, common across engineering and science, is the ratio of signal power to noise power [2]. This core concept is adapted in electrophysiology to accommodate the specific characteristics of neural data, which can range from continuous voltage traces to discrete spike events.
Standard Power-Based Definitions
For continuous voltage signals, such as those recorded by EEG, ECoG, or MEAs, SNR is most commonly defined using power-based metrics, often expressed in decibels (dB) [2].
- Power Ratio:
SNR = P_signal / P_noise, wherePrepresents average power. - Amplitude Ratio: Since power is proportional to the square of amplitude, SNR can also be calculated as
SNR = (A_signal / A_noise)², whereArepresents root mean square (RMS) amplitude [2]. - Decibel Conversion: For a logarithmic scale,
SNR_dB = 10 * log10(P_signal / P_noise)orSNR_dB = 20 * log10(A_signal / A_noise)[2].
Alternative Definitions for Specific Contexts
In some electrophysiological contexts, alternative definitions are more practical.
- Mean-to-Standard Deviation Ratio: An alternative definition uses the ratio of the mean (µ) of a signal to its standard deviation (σ), expressed as
SNR = µ / σ[2]. This is closely related to the sensitivity index d'. The square of this ratio,SNR = µ² / σ², is equivalent to the power-based definition if the signal is a constant value [2]. - Point Process SNR for Single Neurons: For the binary, discrete nature of neural spiking activity, the standard Gaussian-model-based SNR definitions are inappropriate. A specialized approach uses point process generalized linear models (PP-GLMs) to define SNR based on a ratio of expected prediction errors, accounting for the influence of stimuli and the neuron's intrinsic biophysical properties [3]. Studies using this method report single-neuron SNRs typically ranging from -29 dB to -3 dB across various brain regions, underscoring the noisiness of individual neurons [3].
Comparative SNR Metrics Across Recording Modalities
The practical application of these formulas differs significantly across electrophysiological techniques. The table below summarizes the key SNR definitions and their typical applications for EEG, ECoG, and intracortical recordings.
Table 1: SNR Definitions and Applications by Recording Modality
| Modality | Typical Signal Type | Common SNR Definition | Key Application Context |
|---|---|---|---|
| EEG | Event-Related Potentials (ERPs) | SNR = ERP_amplitude / SD_EEG_epoch [4] |
Quantifying evoked responses against background brain activity [4]. |
| ECoG / µECoG | Sensory Evoked Potentials (SEPs), LFPs | Power ratio or amplitude ratio, compared directly with intracortical recordings [1]. | Assessing signal quality and spatial resolution on the cortical surface [1]. |
| Intracortical MEA | Action Potentials (Spikes), LFPs | Power ratio; PP-GLM for single-unit activity [3] [1]. | Isolating single-neuron activity and high-frequency components from noise [1]. |
Experimental Data and Performance Comparison
Direct comparisons between modalities reveal how technological trade-offs impact measurable signal quality. A recent study comparing subdural µECoG and intracortical MEA recordings of somatosensory evoked potentials (SEPs) in pigs provides illustrative data.
Table 2: Experimental Comparison of SEP Recordings between µECoG and Intracortical MEA
| Parameter | µECoG (Subdural) | Intracortical MEA | Statistical Significance |
|---|---|---|---|
| SEP Amplitude | Lower | Higher | Significant [1] |
| Spectral Power | Lower, especially at high frequencies | Higher, contains more high-frequency content | Significant [1] |
| Measured SNR | Comparable | Comparable | Not Significant [1] |
| Key Advantage | Less invasive, stable chronic recordings | High spatial resolution, access to deep layers | N/A |
This data demonstrates a critical insight: while intracortical MEAs record larger amplitude signals and broader frequency content, the inherent noise levels are also higher, resulting in SNRs that are not statistically superior to those achieved with less invasive µECoG arrays [1]. This supports the utility of µECoG as a balanced solution for chronic applications.
Detailed Experimental Protocols for SNR Calculation
To ensure reproducibility and valid comparisons, standardized protocols for data acquisition and processing are essential. The following workflow details a methodology for calculating SNR from evoked potentials, adaptable across EEG, ECoG, and MEA setups.
Protocol for SNR Calculation from Evoked Potentials
This protocol is based on methods used for quantifying SNR in Event-Related Potentials (ERPs) and Somatosensory Evoked Potentials (SEPs) [4] [1].
Subject Preparation & Electrode Implantation:
- Animals: Perform a craniotomy over the target brain region (e.g., primary somatosensory cortex, S1).
- EEG: Apply a scalp electrode cap according to the 10-10 system.
- µECoG: Implant the array subdurally on the cortical surface.
- MEA: Penetrate the array perpendicularly into the cortex to a depth of 1.5-2.0 mm.
- Allow tissue responses and impedances to stabilize for approximately one hour post-implantation [1].
Stimulus Presentation & Data Acquisition:
- Apply controlled sensory stimuli (e.g., electrical stimulation of a peripheral nerve).
- Use a constant-current stimulator with defined parameters (e.g., 1 mA amplitude, 500 μs pulse duration) [1].
- Deliver multiple trials (e.g., 50-100 pulses) with a pseudo-random inter-stimulus interval to avoid habituation.
- Record neural signals synchronously with stimulus triggers.
- Acquisition Settings: Sample rate ≥ 1 kHz, appropriate hardware filtering (e.g., 0.1 Hz - 5 kHz bandpass) [1].
Signal Preprocessing:
- Apply a band-pass digital filter (e.g., 4th order Butterworth, 0.1-40 Hz for ERPs) to remove drifts and high-frequency noise [4] [1].
- For EEG, perform Independent Component Analysis (ICA) to identify and remove artifacts from eye movements or muscle activity [4].
- Re-reference signals to a common average or a quiet reference electrode.
Epoch Extraction:
Signal and Noise Estimation:
- Noise Estimation (σ_noise): Calculate the standard deviation (SD) of the voltage across the entire epoch, including both pre- and post-stimulus periods. This SD represents the amplitude of the background noise [4].
- Signal Estimation (A_signal): Measure the peak amplitude of the evoked response relative to the baseline. For an SEP, this might be the amplitude of the N1 or P1 component [4].
SNR Calculation:
- Compute the SNR as the ratio of the signal amplitude to the noise standard deviation:
SNR = A_signal / σ_noise[4]. - This unitless ratio can be converted to decibels:
SNR_dB = 20 * log10(SNR).
- Compute the SNR as the ratio of the signal amplitude to the noise standard deviation:
Statistical Analysis & Reporting:
- Calculate the mean and standard deviation of SNR values across all trials and subjects.
- Use appropriate statistical tests (e.g., t-tests, ANOVA) to compare SNR between groups or recording modalities, as demonstrated in the comparative study between µECoG and MEA [1].
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful electrophysiology experiments rely on a suite of specialized equipment and materials. The following table details key components used in the featured comparative studies.
Table 3: Essential Materials for In Vivo Electrophysiology Research
| Item Name & Specifications | Function in Experiment | Example Model / Source |
|---|---|---|
| Intracortical Microelectrode Array (MEA) | Records high-resolution neural signals (spikes & LFPs) from within brain tissue. 16-channel, Pt/Ir, 75μm tip. | Microprobes Inc. (Model #MEA-PI-A3-00-16-0.6-2.0-3-1.0-1.0-1-1SS-1) [1] |
| µECoG Array | Records cortical surface potentials with high spatial density. 32-channel, 200μm diameter electrodes. | Neuronexus Technologies (Model #E32-1000-30-200-HZ32) [1] |
| Programmable Stimulator | Generates precise, timed electrical pulses for sensory stimulation. | Multichannel Systems (STG4008) [1] |
| Data Acquisition System | Amplifies, filters, and digitizes analog neural signals from electrodes. | Tucker-Davis Technologies (RZ2 bioamp processor, ZIF-Clip headstage) [1] |
| Faraday Cage & Grounding Setup | Shields the preparation and equipment from environmental electromagnetic interference, reducing noise. | Custom-built enclosure with single-point grounding [5] |
| Differential Amplifier (High CMRR) | The core of signal amplification, rejecting noise common to both signal and reference inputs. | Specialized bioamplifier (CMRR > 100 dB) [5] |
The definition of Signal-to-Noise Ratio in electrophysiology is not monolithic but is instead tailored to the specific recording modality, signal type, and experimental question. While intracortical MEAs provide superior signal amplitude and access to high-frequency neural activity, less invasive techniques like µECoG can achieve comparable SNR for evoked potentials, making them viable for chronic applications. The choice of interface should therefore be guided by a balanced consideration of SNR, spatial resolution, invasiveness, and long-term stability. Standardizing the methodologies for calculating SNR, as outlined in this guide, is crucial for generating reproducible and comparable data across the field, ultimately accelerating progress in neuroscience research and therapeutic development.
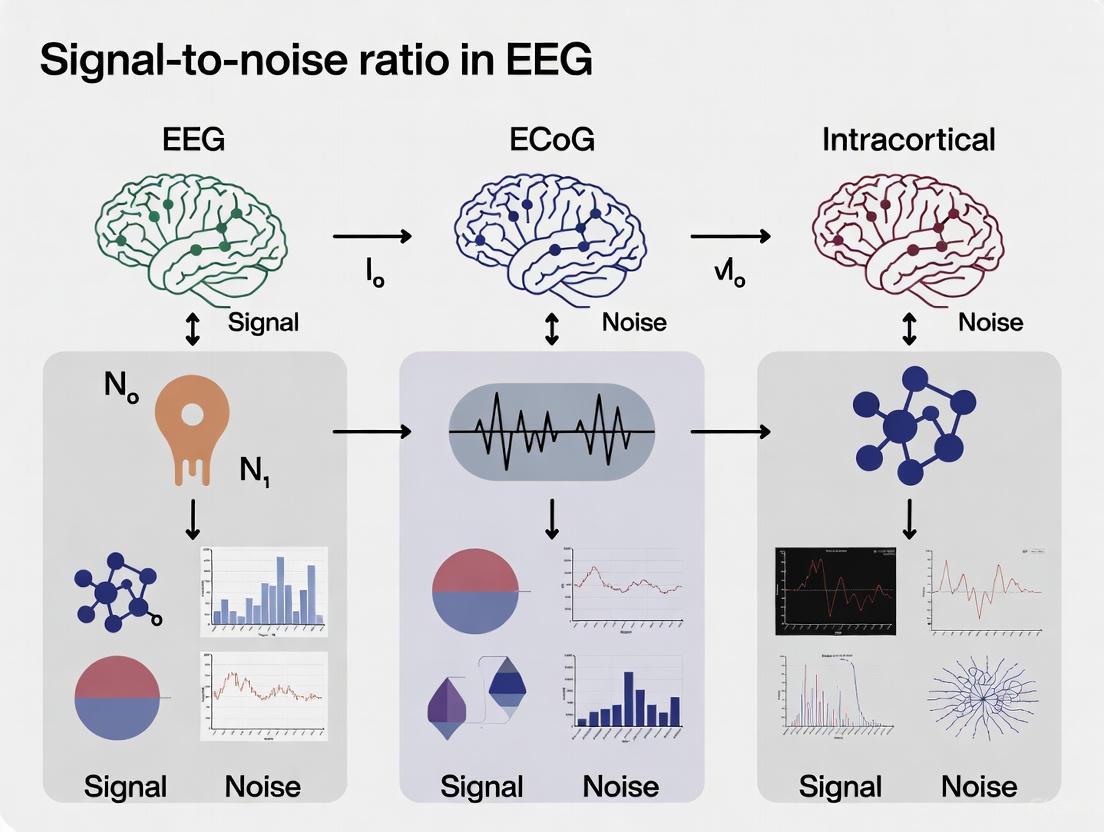
Electrophysiological recording techniques form a hierarchy, balancing invasiveness with resolution. On one end, scalp Electroencephalography (EEG) offers a non-invasive window into brain activity, while on the other, intracortical microelectrodes provide high-resolution data at the cost of penetrating brain tissue. Bridging this gap is Electrocorticography (ECoG), which records from the cortical surface. Understanding the spatial and temporal resolution, along with the signal-to-noise ratio (SNR) characteristics of each modality, is fundamental for selecting the appropriate tool for neuroscience research, clinical diagnosis, and the development of brain-computer interfaces (BCIs). This guide objectively compares the performance of these primary recording modalities, underpinned by experimental data and detailed methodologies.
Hierarchical Comparison of Recording Modalities
The table below summarizes the core characteristics of scalp EEG, ECoG, and intracortical recordings, illustrating the inherent trade-offs.
Table 1: Key Characteristics of Neural Recording Modalities
| Feature | Scalp EEG | ECoG | Intracortical (LFP & Spikes) |
|---|---|---|---|
| Spatial Resolution | Low (cm range) [6] | Moderate (mm-cm range); local signal with ~3 mm diameter spatial spread [7] | High (LFP: ~0.5- several mm; Spikes: ~100 µm) [7] [8] |
| Temporal Resolution | High (milliseconds) [9] | High (milliseconds) [9] | Very High (sub-millisecond) |
| Invasiveness | Non-invasive | Invasive (requires craniotomy, subdural/epidural placement) [10] | Highly Invasive (penetrates brain parenchyma) [8] |
| Typical Signal Amplitude | Low (microvolts, µV) [11] | Medium (tens to hundreds of µV) [6] | High (LFP: hundreds of µV; Spikes: millivolts, mV) [8] |
| Signal-to-Noise Ratio (SNR) | Low; highly affected by skull and non-neural sources [12] [10] | Moderate to High; less susceptible to biological noise than EEG [10] | High for local neural populations [8] |
| Primary Neural Sources | Synchronous activity of large cortical areas (>8-15 cm²) [6] | Cortical surface local field potentials (LFPs) [7] [8] | LFP: dendritic inputs & local processing; Spikes: output of neurons near the electrode tip [8] |
| Key Clinical/Research Use | Epilepsy monitoring, sleep studies, cognitive neuroscience | Pre-surgical epilepsy mapping, cognitive studies, BCI [7] [13] | Fundamental neuroscience, high-fidelity BCI, mapping microcircuits [8] |
Experimental Evidence and Detection Thresholds
The relationship between these signals is not merely theoretical but is quantifiable through simultaneous recording experiments. A key finding is that scalp EEG requires substantial cortical synchronization to become visible.
Table 2: Experimental Detection Thresholds and Signal Correlations
| Experiment Focus | Key Finding | Quantitative Data / Threshold | Citation |
|---|---|---|---|
| Cortical Area for Scalp EEG | Ictal EEG discharges become detectable only when a sufficient cortical area is synchronously active. | Synchronous ECoG discharges with amplitudes of 200–2000 µV recorded over >8–15 cm² of cortex were required for a correlate on scalp EEG. | [6] |
| Deep Source Visibility | Ictal discharges originating in deep structures (e.g., hippocampus) were not detectable on scalp EEG until they spread to the cortical convexity. | Deep source activity was not visible on scalp EEG, highlighting its limitation in capturing subcortical or deeply sulcal activity. | [6] |
| SNR and Source Localization | The spatial scatter of single-dipole localizations for interictal spikes is highly dependent on the SNR of the scalp recording. | A strong correlation was found between dipole scatter and SNR (r = -0.83, p < 0.0001). Averaging spikes improves SNR and provides a more stable localization. | [12] |
| Uncovering Occult Scalp Potentials | Averaging scalp EEG time-locked to intracranial interictal epileptiform discharges (iIEDs) can reveal potentials not seen on visual inspection of the raw scalp trace. | In one case, 522 of 548 intracranial spikes had no recognizable correlate on unaveraged scalp EEG, but averaging revealed a clear scalp potential. | [11] |
| Endovascular Recording Quality | The signal quality of endovascular (EV) recordings, a minimally invasive method, was compared to subdural (SD) and epidural (ED) arrays. | The bandwidth and SNR of EV signals were not significantly different from conventional SD and ED interfaces after chronic implantation, supporting its use as a viable less-invasive alternative. | [10] |
Detailed Experimental Protocols
To ensure reproducibility and critical evaluation, this section outlines the methodologies from key studies cited in this guide.
Protocol 1: Simultaneous EEG-ECoG Recording During Seizures
This protocol investigates the correlation between scalp and cortical surface activities during epileptic seizures [6].
- Objective: To determine the conditions under which ictal (seizure) discharges recorded in ECoG become visible on simultaneous scalp EEG.
- Subjects: Eight human patients with partial epilepsy undergoing pre-surgical evaluation.
- Recording Setup: Chronic subdural ECoG electrodes were implanted. Simultaneous recordings of scalp EEG and ECoG were performed during the patients' ictal periods.
- Data Analysis: The study compared the onset, spread, amplitude, and synchronicity of ictal discharges between the two modalities. The cortical area of involvement was estimated based on the number and location of active ECoG electrodes.
- Key Parameters: Amplitude of ECoG discharges (µV), spatial extent of cortical involvement (cm²), degree of synchronization, and anatomical origin of discharge (deep vs. lateral convexity).
Protocol 2: Estimating Spatial Spread of ECoG and LFP
This protocol uses a model-based approach to estimate the spatial resolution of ECoG and LFP in the primary visual cortex of awake monkeys [7].
- Objective: To accurately determine the spatial spread (the extent of cortical tissue contributing to the signal) of ECoG and LFP.
- Subjects: Awake, behaving monkeys (Macaca radiata and Macaca mulatta).
- Recording Setup: A custom-designed hybrid electrode array was implanted in the visual cortex (V1). This array allowed for simultaneous recording of multi-unit activity (MUA), LFP, and ECoG from the same cortical region.
- Stimulus & Task: Monkeys performed a fixation task while visual stimuli (drifting gratings) were presented on a screen to map the receptive fields (RFs).
- Data Analysis:
- The receptive fields for MUA, LFP, and ECoG were mapped.
- The model by Xing et al. (2009) was applied, which uses the difference in RF sizes between MUA and LFP (or ECoG) to estimate the spatial spread of the latter, canceling out common inflationary factors like eye movements.
- An alternative method involved simulating ECoG as a sum of LFPs over a progressively larger area and comparing the slope of the power spectral density of the simulated and actual ECoG.
- Key Parameters: Receptive field size (degrees of visual angle), cortical magnification factor (mm/degree), and calculated spatial spread (mm).
Protocol 3: Depth-to-Scalp Electric Source Imaging (dsESI)
This protocol leverages simultaneous stereo-EEG (sEEG) and scalp EEG to analyze the spatiotemporal dynamics of interictal epileptiform discharges (IEDs) [11].
- Objective: To extract information about epileptic network nodes and propagation by analyzing scalp potentials averaged from intracranial IEDs.
- Subjects: Human patients with drug-resistant epilepsy undergoing sEEG monitoring with concurrent scalp EEG.
- Data Processing Workflow:
- IED Identification & Clustering: Intracranial IEDs were manually marked and then clustered using fuzzy c-means clustering based on their morphology and topography.
- Averaging Scalp Correlates: The scalp EEG, time-locked to the peak of the iIEDs within a cluster, was averaged to create an averaged scalp field (ASF), enhancing the SNR of otherwise occult scalp potentials.
- Temporal Analysis: The timing difference (delta) between the peak of the intracranial IED and the peak of the ASF was calculated for each cluster.
- Source Localization: Electric Source Imaging (ESI) using sLORETA was performed on the ASFs to estimate the cortical sources of the averaged potentials (this step is termed dsESI).
- Key Parameters: Inter-channel jitter (variability in spike timing), ASF amplitude (µV) and width (ms), source localization solution, and spatial dispersion metric.
The following diagram illustrates the logical workflow and relationships of the dsESI protocol.
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful execution of the protocols above relies on a suite of specialized materials and data analysis solutions.
Table 3: Key Reagents and Solutions for Electrophysiology Research
| Item Name | Function / Application | Specific Examples / Notes |
|---|---|---|
| Hybrid Electrode Array | Allows simultaneous recording of multiple signal types (e.g., ECoG, LFP, MUA) from the same cortical region, enabling direct correlation. | Custom 3x3 ECoG with integrated 9x9 microelectrodes [7]; Flexible electrode-mesh with fenestrae for microelectrode penetration [8]. |
| Stereotactic EEG (sEEG) Electrodes | Minimally invasive depth electrodes for recording from cortical and subcortical structures. Used for epilepsy monitoring and cognitive tasks like speech production. | Platinum-iridium sEEG electrode shafts (e.g., Microdeep intracerebral electrodes) with multiple contacts [13]. |
| Endovascular Stentrode | A minimally invasive neural interface implanted via blood vessels, avoiding the need for a craniotomy. | Stent-mounted electrode array placed in the superior sagittal sinus for chronic recording of cortical signals [10]. |
| High-Density Amplifier System | Acquires neural data from multiple channels with high fidelity and sampling rate. | Micromed SD LTM amplifiers for sEEG [13]; Systems from Blackrock Microsystems for intracortical arrays [7]. |
| Anatomical Labeling Software Suite | Co-registers post-implantation CT scans with pre-implantation MRI to precisely localize electrode contacts in the brain. | img_pipe Python package [13]; Freesurfer for cortical parcellation and atlas-based labeling (e.g., Destrieux atlas) [13]. |
| Multivariate Pattern Analysis (MVPA) | A class of machine learning techniques that analyzes patterns of neural activity across multiple electrodes or voxels to decode cognitive states or stimulus content. | Used to relate EEG and fMRI data to underlying ECoG signals and to decode object category information from neural responses [9]. |
| Brain-Computer Interface (BCI) Software Platforms | Toolboxes and libraries for real-time signal processing, decoding, and application control in BCI research. | MATLAB with Psychophysics Toolbox for stimulus presentation [9]; LabStreamingLayer (LSL) for data synchronization [13]. |
Signaling Pathways and Experimental Workflows
The relationship between neural events and the signals captured by different modalities is hierarchical. The following diagram synthesizes the core concepts from the cited research into a signaling pathway that shows how micro-scale neural activity is filtered and integrated to produce macro-scale signals like ECoG and EEG.
The choice of neural recording modality is a direct trade-off between spatial resolution and invasiveness. Scalp EEG provides a safe and accessible tool for measuring large-scale cortical synchronization but is inadequate for pinpointing deep or highly focal sources. ECoG offers a superior SNR and spatial resolution for cortical surface mapping, which is invaluable in clinical and cognitive applications. For the highest resolution analysis of neural microcircuits, intracortical recordings are necessary, though they carry the greatest risk. Emerging technologies like endovascular electrodes and advanced analytical methods such as dsESI and MVPA are progressively enhancing our ability to interpret these complex signals, bridging the gaps in this resolution hierarchy and expanding the frontiers of neuroscience and neurotechnology.
Understanding the origins and characteristics of neural signals is fundamental to neuroscience research and the development of brain-computer interfaces (BCIs). The two primary signal sources—population synaptic activity measured by EEG/ECoG and single-unit spikes recorded intracortically—offer distinct advantages and limitations. This guide provides an objective comparison of these signal modalities, focusing on their physiological origins, signal-to-noise ratios (SNR), spatiotemporal resolution, and suitability for various research and clinical applications. Framed within the broader context of SNR comparison research, this analysis synthesizes empirical data to inform researchers, scientists, and drug development professionals in selecting appropriate methodologies for specific investigative needs.
Core Physiological Origins and Signal Characteristics
The fundamental difference between these neural signals lies in their physiological generators and the recording techniques used to measure them.
EEG and ECoG primarily reflect the summed synaptic activity of neuronal populations. When neurotransmitters act on postsynaptic receptors, they generate transmembrane currents that flow through the extracellular space. These synchronized postsynaptic potentials, particularly from pyramidal cells with their aligned apical dendrites, create dipoles that summate to produce measurable field potentials [14]. EEG records these potentials from the scalp, while ECoG (electrocorticography) records from the cortical surface, bypassing the signal-attenuating skull [14] [15].
Intracortical recordings directly capture action potentials (spikes) from individual neurons or small neuronal clusters near the microelectrode tip. These fast Na+ spikes generate strong, localized transmembrane currents detectable as brief (~1 ms), high-amplitude extracellular voltage deflections [14] [16]. Multi-unit activity (MUA), derived from higher-frequency components of the raw signal, represents the superimposed spiking activity of multiple neurons near the electrode without isolating individual units [16].
The table below summarizes the fundamental characteristics of these signal types:
Table 1: Fundamental Characteristics of Neural Signal Sources
| Characteristic | EEG | ECoG | Intracortical Signals (Single-Unit & MUA) |
|---|---|---|---|
| Primary Physiological Origin | Summed synaptic potentials & population activity [14] | Summed synaptic potentials & population activity [14] | Extracellular action potentials (spikes) [14] [16] |
| Typical Amplitude Range | Microvolts (μV) [17] | Microvolts (μV) [17] | Microvolts to Millivolts (μV - mV) for spikes [17] |
| Typical Bandwidth | 0-40 Hz (commonly analyzed) [9] | 0-500 Hz [17] [10] | 0-7,000 Hz (Spikes: ~300-6,000 Hz; LFP: <500 Hz) [16] [17] |
| Key Signal Components | Evoked potentials, oscillatory activity in delta, theta, alpha, beta, gamma bands [18] | Similar to EEG, but with richer high-frequency content (e.g., high-gamma) [18] | Single-Unit Activity (SUA), Multi-Unit Activity (MUA), Local Field Potentials (LFP) [16] |
Figure 1: Neural Signaling Pathways. EEG/ECoG signals originate from synchronized population synaptic activity, while intracortical recordings capture single-unit spikes and their derivatives.
Quantitative Comparison of Signal Properties
Signal-to-Noise Ratio (SNR) and Spatial Resolution
SNR and spatial resolution are critical metrics for evaluating neural recording techniques. The table below synthesizes quantitative data from empirical studies for a direct comparison.
Table 2: Signal-to-Noise Ratio and Spatial Resolution Comparison
| Recording Method | Representative SNR Findings | Spatial Resolution | Key Factors Influencing SNR |
|---|---|---|---|
| EEG | Lower SNR than ECoG; blink artifacts cause significant contamination [15]. SNR for deep sources can be higher than in MEG [19]. | Low (cm-scale) due to skull and tissue blurring [14] [19]. | Distance from sources, skull conductivity, environmental noise, artifacts from blinks/eye movements [19] [15]. |
| ECoG | Superior signal quality and less artifact contamination compared to EEG [15]. Provides stable, long-term SNR [17]. | Moderate (mm-scale, typically 1-10 mm) [17]. Direct cortical contact bypasses skull [14]. | Electrode size and proximity to cortex, brain region, cortical folding, less affected by blink artifacts than EEG [17] [15]. |
| Intracortical | High inherent SNR for spike detection; single MUA recordings are highly informative [16]. SNR can degrade over time due to tissue response [17]. | Very High (μm-scale, 50-100 μm) [17]. Can resolve individual neurons. | Electrote proximity to neurons, glial scarring over time, immune response, electrode material and size [16] [17]. |
Spatiotemporal Resolution and Invasiveness
The choice of recording technique often involves a trade-off between spatial/temporal resolution and the invasiveness of the procedure.
Table 3: Spatiotemporal Resolution and Practical Trade-offs
| Characteristic | EEG | ECoG | Intracortical Recordings |
|---|---|---|---|
| Spatial Resolution | Low (cm-scale) [14] | Moderate (mm-scale) [17] | Very High (μm-scale) [17] |
| Temporal Resolution | High (millisecond) [9] | High (millisecond) [9] | Very High (sub-millisecond) [16] |
| Invasiveness | Non-invasive | Semi-invasive (requires craniotomy, subdural grids) [17] | Highly invasive (penetrating brain tissue) [17] |
| Typical Coverage | Whole-brain | Large cortical areas (lobar/hemispheric) [17] | Focal, small brain regions [17] |
| Long-Term Stability | Excellent (for repeated sessions) | Good (stable for long-term implants) [17] | Variable (can degrade due to glial scarring) [17] |
Figure 2: Invasiveness vs. Spatial Resolution Trade-off. Techniques offering higher spatial resolution typically require more invasive implantation procedures.
Experimental Protocols for Key Comparisons
Protocol 1: Multivariate Comparison of EEG, fMRI, and ECoG
This protocol outlines the methodology for a study that directly compared population-level signals (EEG and ECoG) using a multivariate pattern analysis approach [9].
- Objective: To understand the relationship between EEG and ECoG data at the level of neural population codes and how this relationship is affected by stimulus variations [9].
- Participants: 21 healthy volunteers for EEG and fMRI, with previously published ECoG data from epileptic patients used for comparison [9].
- Stimulus Set: 125 grayscale images from five object categories (animals, chairs, faces, fruits, vehicles). Each object was presented under five different viewing conditions (varying size and orientation) [9].
- EEG Acquisition: Signals were recorded from 64 sensors using a g.GAMMAsys cap with a g.HIamp amplifier, digitized at 1,100 Hz. The left mastoid served as the reference [9].
- EEG Preprocessing: Data were low-pass filtered at 40 Hz, resampled to 1,000 Hz, and epoched from -100 to +600 ms relative to stimulus onset. Infomax Independent Component Analysis (ICA) was applied to remove artifacts [9].
- Data Analysis: Multivariate pattern analysis was used to assess the correlation between EEG and ECoG signals at different time points after stimulus onset, specifically comparing responses to different object categories and viewing conditions [9].
Protocol 2: Predicting Movement from Multiunit Activity (MUA)
This protocol details an experiment demonstrating the decoding capability of intracortical signals, specifically comparing Single-Units (SUs), MUA, and LFPs [16].
- Objective: To compare the accuracy of predicting arm movements based on different intracortical signals: Single-Units (SUs), Multiunit Activity (MUA), and Local Field Potentials (LFP) [16].
- Animal Model: Three monkeys (Macaca fascicularis) performing either a prehension (grasping) task or a continuous tracing movement task [16].
- Task Design (Prehension): Monkeys initiated a trial by pressing a button. After a cue and delay period, they reached for and grasped one of two objects located in different positions, without visual feedback of their hand during the movement [16].
- Intracortical Recording: Up to 16 glass-coated tungsten microelectrodes were implanted in the dorsal and ventral premotor cortex (PMd and PMv). Electrodes were lowered until spiking activity was encountered [16].
- Signal Extraction:
- Single-Units (SUs): The amplified and filtered (300-6000 Hz) signal was fed to template-matching devices to isolate the activity of up to three single units per electrode [16].
- LFP: Obtained by low-pass filtering the raw trace, typically below 100 Hz [16].
- MUA: Computed as the root mean square (RMS) of the signal in the 300-6000 Hz frequency band, reflecting spiking activity within ~100 μm of the recording electrode without explicit spike sorting [16].
- Data Analysis: Movement parameters (reach direction, grasp type) were predicted from each signal type using decoding algorithms, and prediction accuracies were compared [16].
Figure 3: Experimental Workflow for Neural Signal Comparison. Common workflow for studies comparing EEG, ECoG, and intracortical signals, covering acquisition, processing, and analysis stages.
Suitability for Research and Clinical Applications
The distinct characteristics of each signal type make them suitable for different applications in research and clinical settings.
Table 4: Application Suitability in Research and Clinical Contexts
| Application Domain | EEG | ECoG | Intracortical Recordings |
|---|---|---|---|
| Basic Neuroscience | Large-scale network dynamics, sleep studies, cognitive states (attention) [17] [18] | Cortical processing with better spatiotemporal resolution than EEG, high-gamma activity [18] | Microcircuit computation, causal manipulation, columnar organization, detailed process analysis (e.g., decision-making) [17] |
| Brain-Computer Interface (BCI) | Spelling interfaces, basic neuroprosthetic control [17] | Gross motor control (e.g., arm reaching), suitable for long-term implants [17] | Fine dexterous control (e.g., individual finger movements), enables complex language production [16] [17] |
| Clinical/Medical | Epilepsy monitoring (non-invasive screening), diagnosis of encephalopathies [19] | Surgical mapping for epilepsy, localization of epileptogenic foci [19] [15] | Currently limited to research or severe paralysis cases; higher invasiveness raises ethical and safety concerns [17] |
| Drug Development | Pharmaco-EEG for assessing drug effects on brain states and network oscillations | Potential for direct cortical monitoring of drug effects in patients with implanted grids | High-precision investigation of drug effects on single-neuron and microcircuit activity |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 5: Key Materials and Solutions for Neural Signal Research
| Item | Function/Application | Key Considerations |
|---|---|---|
| High-Density EEG Systems (e.g., 64+ channels) [9] | Recording scalp potentials for cognitive studies and clinical monitoring. | High-density arrays improve spatial resolution via source modeling [14]. Use of electroconductive gel is required. |
| ECoG Grids/Strips (Platinum-Iridium) [14] | Subdural recording of cortical surface potentials in surgical patients. | Flexible, closely spaced (<5 mm) electrodes for improved resolution [14]. Requires sterile surgical implantation. |
| Intracortical Microelectrodes (e.g., Utah Array, Michigan Probe) [16] [17] | Recording single/multi-unit activity and LFPs within brain tissue. | Material biocompatibility (e.g., silicon, tungsten), electrode density, and long-term stability are critical [16] [17]. |
| Endovascular (Stentrode) Array [10] | Minimally-invasive recording of cortical signals from within blood vessels. | Does not require craniotomy. Signal quality is comparable to epidural arrays after vessel incorporation [10]. |
| Neural Signal Amplifiers & Data Acquisition Systems [9] [16] | Amplification, filtering, and digitization of raw neural signals. | High sampling rate (>1 kHz for EEG/ECoG; >25 kHz for spikes), appropriate bandpass filtering, and low noise are essential. |
| Computational Tools for Signal Processing (e.g., EEGLAB [9]) | Preprocessing, artifact removal (e.g., ICA), and time-frequency analysis. | Capability for multivariate analysis and machine learning-based decoding is increasingly important [9] [16]. |
In neuroscience research and the development of brain-computer interfaces (BCIs), a fundamental challenge persists: achieving high-quality neural signals often requires invasive methods that inevitably cause tissue damage. This creates a critical trade-off where researchers must balance the fidelity of neural data against the degree of invasiveness imposed on neural tissue. This guide objectively compares the performance of three primary neural recording modalities—electroencephalography (EEG), electrocorticography (ECoG), and intracortical microelectrode arrays (MEAs)—within the context of this invasiveness-signal-to-noise ratio (SNR) continuum.
EEG represents the least invasive approach, recording from the scalp surface, while intracortical MEAs are the most invasive, penetrating brain tissue to record single-neuron activity. ECoG occupies a crucial middle ground, involving electrodes placed on the cortical surface, either subdurally or epidurally [20] [21]. Understanding the quantitative relationship between the anatomical placement of these interfaces and their resulting signal characteristics is essential for selecting the appropriate technology for specific research or clinical applications, from basic neurophysiology to next-generation BCIs.
Quantitative Comparison of Recording Technologies
The following table summarizes the key performance characteristics and technological trade-offs of the three main neural recording modalities.
Table 1: Performance Comparison of Neural Recording Technologies
| Feature | EEG (Non-invasive) | ECoG (Minimally Invasive) | Intracortical MEA (Fully Invasive) |
|---|---|---|---|
| Anatomical Placement | Scalp surface | Cortical surface (subdural/epidural) | Penetrates cortex into brain tissue |
| Spatial Resolution | Low (centimeters) [22] | Moderate (millimeters) [21] | High (micrometers) [20] |
| Signal Amplitude | 10-20 μV [22] | 50-100 μV [22] | Highest (microvolts for spikes) [20] |
| Frequency Bandwidth | 0-50 Hz [22] | 0-500 Hz [22] | 0-5000 Hz+ (includes spike activity) [20] |
| Signal-to-Noise Ratio (SNR) | Lowest, susceptible to artifacts [23] [24] | Moderate to High [24] [21] | Highest [20] |
| Tissue Damage / Biostability | None | Minimal; lower risk of medical complications [21] | Significant; tissue damage, scarring, signal deterioration over time [20] [21] |
| Long-Term Stability | Excellent (non-invasive) | Good; stable for chronic implants [24] [21] | Variable; can deteriorate due to tissue response [20] [25] |
| Primary Signal Content | Averaged synaptic potentials | Local field potentials (LFPs), high-frequency gamma | Single- and multi-unit activity (spikes), LFPs [20] |
Experimental Evidence and Methodologies
Direct comparative studies are essential to quantitatively evaluate the trade-offs summarized above. The following section details key experimental protocols and their findings.
Direct Comparison of ECoG and Intracortical MEA
A 2024 study provides a direct, quantitative comparison of somatosensory evoked potentials (SEPs) recorded using both µECoG and intracortical MEAs in a pig model [20].
Experimental Protocol:
- Animal Model: Twenty healthy female Danish Landrace pigs.
- Electrode Implantation: Nine animals were implanted with a 16-channel intracortical MEA (4x4 array, 1.0 mm spacing, 2 mm length) penetrated 1.6-1.8 mm into the primary somatosensory cortex (S1). Eleven animals were implanted subdurally with a 32-channel µECoG array (8x4 configuration, 1 mm spacing) over S1 [20].
- Stimulation: SEPs were generated by applying electrical stimulation to the contralateral ulnar nerve.
- Signal Acquisition & Processing: Signals were acquired at 24 kHz. Analysis focused on SEP amplitude, SNR, power spectral density (PSD), and correlation structure. Signals were filtered (0.1 Hz - 5 kHz) and notch-filtered (50 Hz & harmonics) [20].
Key Findings:
- Amplitude and Spectral Power: SEPs from intracortical MEA recordings exhibited higher amplitudes and contained significantly more spectral power, particularly at higher frequencies, compared to µECoG [20].
- SNR: Despite the amplitude differences, the SNRs for SEPs were similar between the two interfaces [20].
- Conclusion: The study demonstrated that µECoG is capable of decoding SEPs with performance comparable to MEAs in certain metrics, positioning it as a viable compromise that balances signal quality with reduced invasiveness [20].
ECoG as a High-Performance BCI Modality
The potential of ECoG is further illustrated by its success in BCI applications. A foundational study demonstrated that humans can achieve rapid two-dimensional cursor control using ECoG signals.
Experimental Protocol:
- Subjects: Five human patients with temporarily implanted subdural electrode arrays for epilepsy monitoring.
- Electrode Specification: Arrays consisted of 26-64 electrodes (4 mm diameter, 1 cm spacing) placed over fronto-parietal-temporal regions [22].
- Task & Decoding: Subjects performed various motor and motor imagery tasks (e.g., hand movement, tongue protrusion). ECoG features (spectral amplitudes at specific locations and frequencies) with the largest task-related changes were identified and used for online cursor control [22].
Key Findings:
- Subjects acquired substantial control in brief training periods (12-36 minutes) and achieved high success rates (53-73%) in a two-dimensional task [22].
- ECoG provided a broader bandwidth (0-200+ Hz) and higher amplitude than EEG, enabling faster acquisition of control than typically reported for non-invasive systems [22].
- The study concluded that ECoG-based BCIs combine high performance with technical and clinical practicality [22].
Visualizing the Invasiveness-SNR Trade-off
The relationship between the anatomical depth of an interface and its resulting signal characteristics can be visualized as a continuum. The following diagram illustrates this core trade-off, the associated signal content, and the primary technological challenges at each level.
Diagram 1: The fundamental trade-off in neural recording technologies. As the physical invasiveness of the interface increases (from top to bottom), so does the signal-to-noise ratio (SNR) and the richness of the obtainable neural information. However, this comes at the cost of increased tissue damage and potential long-term instability.
The Scientist's Toolkit: Essential Research Reagents & Materials
Selecting the appropriate tools is critical for designing experiments within the invasiveness-SNR paradigm. The following table details key materials and their functions as derived from the cited experimental methodologies.
Table 2: Key Research Materials for Neural Interface Studies
| Material / Solution | Function & Specification | Relevance to Trade-off |
|---|---|---|
| Intracortical MEA (e.g., Pt/Ir) [20] | 16-channel array (4x4), 2mm length, 75µm tip diameter. Records high-frequency spikes and LFPs directly from neural tissue. | Provides highest signal fidelity but causes significant tissue penetration and damage. |
| µECoG Array [20] [8] | 32-channel flexible array (8x4), 200µm diameter electrodes, 1mm spacing. Conforms to cortical surface. | Optimizes balance; high SNR and broad bandwidth with minimal cortical penetration. |
| Insulated Screw ECoG [24] | Novel screw-shaped electrode with zirconia insulator. Designed for chronic implantation through the skull. | Offers stable, high-SNR recording (better than EEG) with minimal invasiveness compared to grid ECoG. |
| Flexible Electrode-Mesh [8] | Parylene-C-based mesh with fenestrae, allowing simultaneous ECoG and intracortical penetration. | Enables direct comparison of surface vs. depth signals, advancing understanding of signal sources. |
| Deep Learning Decoders (e.g., EEGNet) [26] | Convolutional neural networks optimized for EEG/ECoG signal classification. | Mitigates lower SNR of non-/minimally-invasive signals via advanced processing, improving BCI performance. |
| Somatosensory Evoked Potential (SEP) Protocol [20] | Electrical stimulation of peripheral nerve (e.g., ulnar nerve) to generate controlled, measurable cortical responses. | Provides a standardized, quantitative method for comparing signal quality across different recording modalities. |
The choice of a neural recording technology is not a simple selection of the "best" interface but a deliberate compromise based on the specific requirements of an application. EEG remains the most accessible and safe option for many basic research and clinical monitoring tasks, though its utility is limited by poor spatial resolution and low SNR. Intracortical MEAs provide unparalleled resolution for decoding fine-grained neural commands, a necessity for advanced motor BCIs, but this comes with significant biological costs and long-term stability challenges. ECoG, particularly in its modern µECoG and high-density forms, successfully navigates the middle path, offering a favorable balance of high signal fidelity, clinical practicality, and reduced tissue damage [20] [21] [25].
Future progress will likely stem from both technological and algorithmic advances. The development of flexible, high-density electrode arrays [25] and chronically stable, insulated designs [24] pushes the boundary of what is possible from the cortical surface. Concurrently, sophisticated deep learning decoders are increasingly capable of extracting meaningful information from noisier, non-invasive signals [26]. For researchers and clinicians, a clear understanding of this invasiveness-SNR trade-off is fundamental to driving innovations that are not only technologically powerful but also biologically compatible and clinically viable.
The fidelity of neural recordings is fundamentally governed by the physical principles of volume conduction and the biological reality of signal attenuation through various tissue layers. For researchers and drug development professionals, the choice of neural recording modality—from non-invasive electroencephalography (EEG) to invasive electrocorticography (ECoG) and intracortical methods—represents a critical trade-off between signal quality and procedural invasiveness. The Signal-to-Noise Ratio (SNR) is a pivotal metric that quantifies this trade-off, directly impacting the resolution and reliability of neural data for both basic research and clinical applications. This guide provides an objective comparison of these modalities based on experimental data, focusing on how brain geometry and tissue composition influence recording quality. A foundational understanding begins with the sources of these signals: the extracellular field is a superposition of all transmembrane currents, with synaptic activity being the most significant contributor under physiological conditions, alongside action potentials and intrinsic membrane oscillations [14].
Fundamental Principles of Signal Generation and Attenuation
The Biophysics of Extracellular Signals
Neuronal activity generates transmembrane currents that propagate through the extracellular medium. The recorded signal is a spatial and temporal integration of all active cellular processes within a volume of tissue. The key distinction between recording modalities lies in their proximity to these neural sources and the number and composition of tissue layers the signal must traverse.
- Signal Sources: The primary source of measurable extracellular potentials is synaptic transmembrane currents. The influx of cations at excitatory synapses creates a local extracellular sink, which is balanced by a passive return current (source) along the neuron, forming a current dipole [14].
- Impact of Tissue Layers: Different tissues have varying electrical conductivity. The cerebrospinal fluid (CSF) is highly conductive, while the skull is a strong attenuator, and the scalp further distorts and smears the signal. The presence of insulating layers (e.g., dura) or conductive layers (e.g., saline) above the cortex can dramatically alter the spatial profile and amplitude of recorded potentials [27] [14].
The Role of Geometry and Boundary Conditions
The geometry of both the brain and the recording device itself influences the measured signal. When an electrode is placed on the brain's surface, the boundary condition (e.g., insulating vs. conductive) can no longer be neglected. For instance, an insulating boundary, such as a dry cortical surface or an insulating electrode array, can amplify surface potentials by about a factor of 4 compared to a conductive boundary like a saline-covered cortex [27]. This demonstrates that experimenters can exert some control over spatial selectivity and amplitude through appropriate device design and implantation procedures.
The following diagram illustrates the core concepts of how neural signals are generated and attenuated before reaching different types of sensors.
Quantitative Comparison of Recording Modalities
Experimental data from animal models and human studies provide direct comparisons of SNR and bandwidth across different recording technologies. The following table summarizes key performance metrics from empirical studies.
Table 1: Signal Quality Comparison Across Neural Recording Modalities
| Recording Modality | Implantation Depth & Tissue Layers | Typical SNR (for VEP) | Maximum Bandwidth | Key Signal Characteristics |
|---|---|---|---|---|
| Scalp EEG | On scalp, signal passes through skin, skull, CSF, dura, pia | Lower (reference for comparison) [10] | Limited high-frequency content [14] | Spatially smoothed, integrated over ~10 cm² [14]; highly susceptible to blink/movement artifacts [15] |
| Endovascular (EV) | Within cortical blood vessel (e.g., superior sagittal sinus) | Comparable to ED and macro SD arrays [10] | Up to 200-500 Hz [28] [10] | Minimally invasive; signal quality comparable to conventional sensors after 3-4 weeks post-implantation [10] |
| Sub-Scalp (on Periosteum) | Above the skull, beneath the scalp | Lower than Peg and ECoG [28] | Up to 120 Hz [28] | Stable, discrete; suitable for chronic use; signal quality improves with closer proximity to the brain [28] |
| Sub-Scalp (Peg) | Partially embedded within the skull (bur hole) | Approaches that of ECoG [28] | Up to 180 Hz [28] | Highest quality among sub-scalp options; requires more complex implantation [28] |
| Epidural (ED) | On the dura mater, beneath the skull | Comparable to EV and macro SD arrays [10] | < 500 Hz [10] | Requires craniotomy; avoids direct cortical contact [10] |
| Subdural (SD) / ECoG | On the cortical surface (pia mater), beneath the dura | High [28] [15] | < 500 Hz [10]; High Gamma (70-190 Hz) is a robust marker [29] | High spatial (<5 mm²) and temporal resolution; less susceptible to movement artifacts than EEG [15] [14] [29] |
| Intracortical/LFP | Penetrating the cortex, within brain tissue | Highest for local sources | Up to 40 kHz (can record unit spikes) [14] | Records highly local activity; can resolve single-neuron spiking; assumption of infinite homogeneous medium may be valid for deep sources [27] [14] |
Experimental Protocols and Methodologies
To ensure the reproducibility of findings and validate the comparisons drawn, this section details the key experimental methodologies from the cited research.
Protocol: Boundary Condition Manipulation on Cortical Surface
This experiment quantified the impact of surface boundary conditions on ECoG recordings [27].
- Objective: To determine how insulating versus conductive layers above the cortical surface affect the amplitude of recorded evoked potentials.
- Animal Model: Anesthetized mice.
- Electrode: Laminar array with 22 contacts inserted into the whisker barrel cortex.
- Procedure:
- The exposed cortex was covered with artificial CSF (ACSF), creating a conductive boundary.
- Evoked potentials in response to whisker stimulation were recorded.
- The ACSF was wicked away using a Kimwipe, creating a dry (insulating) boundary.
- The stimulation and recording were repeated.
- Signal Processing: Local Field Potential (LFP) was analyzed by calculating the Root Mean Square (RMS) value over the response window to estimate overall magnitude. Multi-unit activity (MUA) was high-pass filtered at 350 Hz, and its amplitude was computed using the Hilbert transform.
- Key Finding: The insulating (dry) boundary amplified potentials by a factor of approximately 4 compared to the conductive (ACSF) boundary [27].
Protocol: Comparative SNR of Minimally Invasive Interfaces
This study systematically compared the signal quality of various electrode placements in a sheep model [28].
- Objective: To quantify and compare the SNR and maximum bandwidth of sub-scalp EEG, ECoG, and endovascular recordings.
- Animal Model: Anesthetized sheep.
- Electrode Placements (sequential):
- Endovascular: A stent-electrode array was deployed in the transverse sinus.
- Periosteum: A disc electrode array was sutured onto the periosteum over the occipital bone.
- Skull Surface: The same array was screwed into the skull after periosteum removal.
- Peg: A bur hole was made, and a peg electrode array was inserted.
- ECoG: A craniotomy was performed, and an ECoG array was placed on the visual cortex.
- Stimulus: Visual Evoked Potentials (VEP) were elicited using a full-field flash stimulator at 0.99 Hz for 5 minutes.
- Analysis: SNR was calculated from the VEP responses. The maximum bandwidth was determined by identifying the frequency at which the power spectrum during a background (no-stimulus) recording fell to the noise floor.
- Key Finding: Peg electrodes achieved VEP SNR approaching that of ECoG, and endovascular arrays exhibited SNR comparable to periosteum electrodes [28].
The workflow for a typical comparative experiment is summarized below.
The Scientist's Toolkit: Key Research Reagents and Materials
Selecting the appropriate tools is critical for designing experiments in neural recording. The following table lists essential materials and their functions as derived from the featured experiments.
Table 2: Essential Materials for Neural Interface Experiments
| Material / Device | Function in Research | Specific Examples & Notes |
|---|---|---|
| Laminar Electrode Array | Records neural activity at multiple depths simultaneously. | Used to measure depth-dependence of boundary effects (e.g., 22 contacts, 100 µm spacing) [27]. |
| Micro-Electrocorticography (µECoG) Array | High-density surface recording from the cortex with sub-millimeter resolution. | Flexible, closely spaced grid electrodes; subject to ongoing materials research for improved flexibility and biocompatibility [30]. |
| Endovascular Stent-Electrode Array | Minimally invasive neural recording from within a blood vessel. | "Stentrode"; requires ~2 weeks for incorporation into the vessel wall for stable recording [10]. |
| Sub-Scalp EEG Electrodes (Peg, Ring, Disc) | Chronic, stable neural recording from beneath the scalp but above the brain. | Peg electrodes (partially embedded in skull) offer superior SNR to disc/ring electrodes [28]. |
| Artificial Cerebrospinal Fluid (ACSF) | Maintains physiological ionic environment and creates a conductive boundary condition. | Used experimentally to manipulate surface boundary conditions on the cortex [27]. |
| Intan Technologies RHD2000 Amplifier | Acquires and digitizes raw neural signals with high fidelity. | A common data acquisition system used in experimental setups (e.g., sampled at 20 kHz) [27] [28]. |
| Silicon-Based Polytrodes | Records extracellular action potentials and LFP from dense neuronal populations within the brain. | Enables high-density recording of elementary processes for intracortical studies [14]. |
The selection of a neural recording modality is a decisive factor that directly shapes the quality and interpretation of experimental data. As the comparative data demonstrates, a clear trade-off exists: as one moves from minimally invasive sub-scalp EEG to highly invasive intracortical methods, the SNR and spatial resolution increase substantially, primarily due to the reduction of signal-attenuating tissue layers. Furthermore, the geometry and material properties of the recording interface itself, such as the boundary condition it creates, can actively shape the recorded signal. For researchers and drug development professionals, this guide underscores that there is no single "best" technology, but rather a spectrum of tools suited for different experimental questions and risk tolerances. The ongoing innovation in high-density µECoG [30] and minimally invasive endovascular and sub-scalp arrays [28] [10] continues to push the boundaries, offering ever-improving signal quality for both scientific discovery and clinical application.
Technological Advancements and SNR-Driven Applications in Research and Clinics
In the pursuit of advanced brain-computer interfaces (BCIs) for restoring communication, researchers have long faced a fundamental trade-off: the choice between the fine-grained signals captured by invasive, penetrating electrodes and the broader, safer coverage of surface recordings. Electroencephalography (EEG), while non-invasive, suffers from limited spatial resolution due to signal attenuation and scattering by the skull [10]. Traditional electrocorticography (ECoG) and stereoencephalography (SEEG) electrodes offer improved signal quality by recording from the cortical surface or depth, but their relatively large size (typically several millimeters) inadequately captures the rich spatio-temporal structure of human brain signals [31] [32]. Into this landscape emerge high-density micro-electrocorticography (µECoG) arrays, which represent a paradigm shift by offering an optimal balance between invasiveness and signal quality. These arrays feature electrode diameters and spacings an order of magnitude smaller than conventional ECoG, enabling unprecedented access to the neural signatures of speech with 57 times higher spatial resolution and 48% higher signal-to-noise ratio (SNR) compared to macro-ECoG and SEEG [31]. This breakthrough in recording technology has directly translated to a 35% improvement in speech decoding accuracy, bringing us closer than ever to viable neural speech prostheses for patients with debilitating neurodegenerative diseases [31] [32].
Quantitative Performance Comparison of Neural Recording Technologies
The performance advantages of high-density µECoG can be fully appreciated through direct, quantitative comparison with established neural recording methods. The table below synthesizes key metrics from recent studies, highlighting the transformative potential of µECoG.
Table 1: Performance Metrics of Neural Recording Technologies
| Recording Technology | Spatial Resolution | Signal-to-Noise Ratio (SNR) | Speech Decoding Accuracy | Key Advantages | Primary Limitations |
|---|---|---|---|---|---|
| High-Density µECoG | 57x higher than macro-ECoG/SEEG [31] | 48% higher than macro-ECoG/SEEG [31] | 35% improvement over standard intracranial signals [31] | Excellent balance of high resolution and minimal tissue damage [31] [33] | Still a surface recording; may not access deepest cortical layers |
| Macro-ECoG | Baseline (e.g., 2-3 mm electrodes) [7] | Baseline [31] | Baseline | Tried-and-tested clinical tool; broad coverage [7] | Coarse spatial resolution blurs fine-scale neural activity [31] |
| SEEG (Depth Electrodes) | Varies with electrode design | Lower than µECoG [31] | Lower than µECoG [31] | Accesses deep brain structures | Inherently damaging; higher risk profile [20] [33] |
| Intracortical Microelectrode Arrays (MEA) | Highest (records single neurons) [20] | High amplitude signals, but similar SNR to µECoG for evoked potentials [20] | Excellent for motor control, but less stable over time [33] | Unmatched resolution for single-unit activity [20] | Significant tissue damage; instability in chronic recordings [20] [33] |
| Endovascular (Stentrode) | Comparable to subdural/epidural arrays [10] | Comparable to subdural/epidural arrays [10] | Comparable decoding accuracy to conventional electrodes [10] | Minimally invasive implantation [10] | Lower spatial resolution than µECoG; confined to vascular architecture [10] |
Beyond the metrics in Table 1, the spatial scale of information captured by µECoG is critical. A foundational study demonstrated that even conventional ECoG is a surprisingly local signal, with a spatial spread of approximately 3 mm in diameter [7]. High-density µECoG, with its sub-millimeter electrodes and spacing, capitalizes on this locality to resolve neural population activity with unprecedented clarity, capturing information that is spatially discriminative at scales smaller than 2 mm [31] [32]. This fine-grained mapping is a primary driver of its superior decoding performance.
Experimental Protocols for Evaluating µECoG Performance
Intra-operative Speech Production and Decoding
The remarkable results demonstrating a 35% improvement in speech decoding were generated using a specific and rigorous experimental protocol [31]. During intra-operative procedures, researchers performed high-resolution neural recordings while participants engaged in speech production tasks. The core technology was a liquid crystal polymer thin-film (LCP-TF) µECoG array, which possesses up to 57 times the electrode density of standard macro-ECoG arrays [31] [32].
The experimental workflow can be summarized as follows:
Diagram 1: Speech Decoding Experimental Workflow
Key Methodological Details:
- Neural Feature Extraction: The primary feature for decoding was the high-gamma band (HG, ~70-170 Hz) power, a well-established correlate of local cortical activity and neural firing. Signals were processed to compute the HG envelope, which showed a 48% higher SNR in µECoG compared to standard intracranial recordings [31] [34] [35].
- Decoding Models: Researchers employed both linear models, such as Linear Discriminant Analysis (LDA), and non-linear models, specifically recurrent neural networks (RNNs). The non-linear models, designed to leverage the enhanced spatio-temporal information from µECoG, consistently produced superior results [31] [35]. A key innovation for training models without precise time-aligned data in paralyzed participants was the use of Connectionist Temporal Classification (CTC) loss [35].
- Performance Validation: Decoding accuracy was quantified by the model's ability to predict phonemes or whole words from the neural data. The performance was tested against baseline models using spatially down-sampled data to isolate the benefit of high spatial resolution [31].
Comparative Protocol: µECoG vs. Intracortical Arrays for Evoked Potentials
Another critical protocol directly compared µECoG signals against the gold standard of intracortical Microelectrode Arrays (MEAs) in the somatosensory cortex [20]. This study design allows for a fundamental understanding of how well surface µECoG approximates signals from within the brain tissue.
Methodology Overview:
- Implantation: In a porcine model, a µECoG array was placed subdurally on the cortical surface, while a penetrating MEA was implanted into the primary somatosensory cortex.
- Stimulation: Somatosensory Evoked Potentials (SEPs) were generated by electrically stimulating the ulnar nerve.
- Signal Analysis: The amplitude, SNR, and power spectral density (PSD) of the SEPs recorded by both interfaces were systematically analyzed and compared [20].
Key Finding: While SEPs from intracortical MEAs had higher raw amplitudes and contained more spectral power at higher frequencies, the SNRs between the two interfaces were similar. This demonstrates that µECoG can decode critical neural events with a fidelity comparable to penetrating electrodes for certain frequency bands, but with reduced invasiveness [20].
The Scientist's Toolkit: Essential Research Reagents and Materials
The advancement of high-density µECoG technology relies on a specialized set of materials and instruments. The table below details key components of the research toolkit for this field.
Table 2: Research Reagent Solutions for High-Density µECoG
| Item Name/Type | Function/Application | Key Characteristics | Example Specifications |
|---|---|---|---|
| LCP-TF µECoG Array [31] [32] | High-resolution neural recording from cortical surface | High electrode density, flexible substrate, biocompatible | Electrode density ≤57x > macro-ECoG; ≤9x > standard HD-ECoG [31] |
| Thin-Film Polyimide Arrays [33] | Scalable, minimally invasive cortical mapping | Ultra-conformable, high channel count, suitable for micro-slit insertion | 1,024 channels; 50 µm electrode diameter; 400 µm pitch [33] |
| Custom Hybrid Array (Microelectrodes + ECoG) [7] | Simultaneous recording of MUA, LFP, and ECoG for validation | Integrates multiple electrode types on a single platform | 9 ECoG electrodes (2.3 mm) + 81 microelectrodes [7] |
| High-Gamma Band (HG) Power Analysis [31] [34] [35] | Extraction of speech-related neural features | Tracks local cortical activity and neural firing rates | Frequency: 70-170 Hz; Computed from raw ECoG signals [34] |
| RNN with CTC Loss Decoder [35] | Mapping neural features to text/speech without precise time alignment | Enables training on silently attempted speech from paralyzed users | For large-vocabulary decoding; handles alignment ambiguity [35] |
| Cranial Micro-Slit Delivery System [33] | Minimally invasive surgical implantation of µECoG arrays | Avoids full craniotomy, reduces surgical risk and time | 500-900 µm wide skull incisions; guided by fluoroscopy/endoscopy [33] |
High-density µECoG arrays have decisively addressed the long-standing trade-off between signal quality and invasiveness in neural interface design. By providing a 57-fold increase in spatial resolution and a significant boost in SNR, this technology has enabled a 35% leap in speech decoding accuracy, demonstrating its potential as the foundation for next-generation neural speech prostheses [31]. The experimental data clearly shows that the enhanced spatial sampling of µECoG captures neural information that is simply inaccessible to conventional macro-electrodes, and that non-linear decoding models are best suited to leverage this rich spatio-temporal information [31] [35].
The future of this field points toward even greater integration and miniaturization. Scalable arrays exceeding 1,000 channels are now a reality, and novel surgical techniques, such as guidewire-driven deployment and cranial micro-slit insertions, are paving the way for safer, large-scale cortical coverage [36] [33]. These innovations will be crucial for translating high-performance laboratory demonstrations into practical, clinically viable BCIs that can restore full, embodied communication to individuals living with paralysis.
Interfacial Materials and Electrode Modifications for Lower Impedance and Higher SNR
The performance of neural interfaces, from non-invasive electroencephalography (EEG) to intracortical microelectrodes, is fundamentally constrained by the electrode-tissue interface. The electrode-skin interfacial impedance and the ensuing signal-to-noise ratio (SNR) are pivotal parameters determining the fidelity of the recorded neural signal [37] [38]. High impedance attenuates signal amplitude and increases susceptibility to environmental noise, while a low SNR obscures crucial neural information, compromising the reliability of brain-computer interfaces (BCIs) for both research and clinical applications [39]. Consequently, the development of advanced interfacial materials and electrode modifications is a primary focus in neural engineering. This guide provides a objective comparison of leading-edge technologies aimed at lowering impedance and boosting SNR across the spectrum of neural recording modalities, presenting key performance data and detailed experimental methodologies to inform material and protocol selection for researchers and drug development professionals.
Comparative Performance of Materials and Modifications
The quest for superior neural interfaces has driven innovation in material science, yielding a diverse array of solutions. The following table summarizes the core performance metrics of various state-of-the-art materials and modifications as validated by experimental data.
Table 1: Performance Comparison of Electrode Materials and Modifications for Neural Recording
| Material/Modification | Application Context | Key Performance Findings | Quantitative Data (vs. Baseline) |
|---|---|---|---|
| POLiTAG (PEDOT:PSS/PAMPS Hydrogel) [37] | Wearable EEG | Low electrode-skin impedance & long-term stability. | Impedance: 20.7 kΩ·cm² (lower than gel-based electrodes).Stability: Maintained lower impedance for 29 days. |
| CF4 Plasma-Modified Gold [40] | Intracortical Microelectrodes | Increased surface area, lowered impedance, reduced noise. | Impedance: 98% decrease for 15 µm electrodes.Noise: -106 dB (vs. -86 dB baseline).Evoked LFP: 7x higher amplitude. |
| Sputtered Iridium Oxide Film (SIROF) [41] | Chronic Intracortical Microelectrodes | Superior chronic recording capability despite physical degradation. | Recording Likelihood: 2x higher than Platinum (Pt) electrodes (based on SNR). |
| Close-Packed Gold Nanoparticles (AuNPs) [42] | Electrochemical Sensing (e.g., Dopamine) | Improved signal reproducibility via homogenous active sites. | High signal reproducibility within the same batch of electrodes. |
| Dry Electrodes (Solid Materials) [43] | Wearable ECG | High SNR and low skin-electrode impedance in stationary testing. | SNR Ranking (Stationary): Stainless Steel > Platinum > Silver > Conductive Polymer > Conductive Fabric. |
Detailed Experimental Protocols
Fabrication and Testing of POLiTAG EEG Electrodes
The development of the POLiTAG electrode exemplifies a rational design approach for a wearable EEG interface [37].
- Fabrication Protocol: The precursor solution is prepared by mixing poly(ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS) with the anionic poly(2-acrylamido-2-methyl-1-propanesulfonic acid) (PAMPS) hydrogel network, which boasts a 97.4% water ratio. Secondary additives include Triton X-100 (a non-ionic surfactant to improve conductivity and soften the blend), glycerol (to enhance water-retaining capability and electrical conductivity), and lithium chloride (LiCl to vastly improve ionic conductivity). This mixture is then molded into the desired electrode shape, typically a circle with an area of 2.23 cm², and cured.
- Electrical Characterization Protocol: Electrode-skin interfacial impedance is quantified over time using electrochemical impedance spectroscopy (EIS). The long-term stability is assessed by monitoring impedance values under controlled environmental conditions for up to 29 days. Functional validation involves deploying the electrodes in a wireless single-channel EEG device to detect alpha rhythms (eye-open/close conditions) and to capture oscillatory rhythms in motor imagery protocols. Performance is benchmarked against commercial gel-based Ag/AgCl electrodes.
In Vivo SNR Quantification Using Cortical Slow Oscillations
A novel methodology for quantifying the SNR of recording devices leverages the intrinsic properties of cortical slow oscillations (SOs), providing rich spectral information [38].
- Physiological Basis: Slow oscillations, prevalent during slow-wave sleep or under anesthesia, consist of alternating Up states (periods of neuronal firing) and Down states (periods of neuronal silence). The Up states are treated as the "signal," as they contain a broad band of meaningful neural information, while the Down states represent the "noise."
SNR Calculation Protocol: Local field potentials (LFPs) are recorded from active cortical tissue generating spontaneous SOs. The Power Spectral Density (PSD) is computed for multiple identified Up states and Down states. The spectral SNR in decibels (dB) is then calculated using the formula:
(SNR(f) = 10 \log{10}\left(\frac{\frac{1}{N}\sum{i=1}^{N}(PSD{Up})i}{\frac{1}{N'}\sum{j=1}^{N'}(PSD{Down})_j}\right) [dB])
where (N) and (N') are the total numbers of Up and Down states, respectively. This approach allows for the evaluation of SNR across different frequency bands (5–1500 Hz), contrasting with amplitude-based methods that evaluate only a single frequency.
Plasma Surface Modification for Intracortical Microelectrodes
Surface roughening via plasma treatment is a effective method to enhance the performance of metallic microelectrodes [40].
- Modification Protocol: A tetrafluoromethane (CF4) plasma is applied to the gold electrode sites of polyimide-based neural probes. The plasma treatment parameters are optimized to controllably increase the surface roughness without compromising the structural integrity of the electrode. For 15 µm diameter electrodes, this process can increase the average surface roughness from 1.7 nm to 22 nm.
- Validation Protocol: The efficacy is characterized in vitro through EIS measurements. Subsequent in vivo validation involves acute implantation in the anterior cingulate cortex (ACC) of animal models to record spontaneous and electrically evoked LFPs. The performance of plasma-modified electrodes is directly compared to unmodified controls from the same implant, measuring signal amplitude and background noise power.
Signaling Pathways and Experimental Workflows
The following diagrams illustrate the core concepts of SNR quantification and the logical relationship between electrode properties and functional outcomes.
SNR Quantification Using Cortical Slow Oscillations
Electrode Property-Performance Relationship
The Scientist's Toolkit: Research Reagent Solutions
Successful experimentation in this field relies on a set of key materials and reagents. The following table details essential components, their functions, and examples of their application.
Table 2: Essential Research Reagents and Materials for Neural Interface Development
| Reagent/Material | Function in Research | Application Example |
|---|---|---|
| PEDOT:PSS | Conducting polymer providing mixed ionic/electronic conductivity, mechanical flexibility, and biocompatibility. | Primary conductive component in POLiTAG hybrid hydrogel for EEG electrodes [37]. |
| PAMPS Hydrogel | Ionic hydrogel matrix with high water content (~97.4%) for maintaining low interfacial impedance. | Hydrating matrix in POLiTAG to overcome lack of water in non-invasive skin contact [37]. |
| Triton X-100 | Non-ionic surfactant to improve conductivity of PEDOT:PSS and act as a plasticizer. | Secondary additive to increase linearity of PEDOT and soften the electrode blend [37]. |
| Lithium Chloride (LiCl) | Hygroscopic salt to vastly improve the ionic conductivity of the electrode composite. | Additive to enhance ionic charge transport in polymer-hydrogel blends [37]. |
| Tetrafluoromethane (CF4) | Plasma gas used for surface etching to create nano-scale roughness on electrode surfaces. | Surface modification of gold microelectrodes to drastically increase effective surface area [40]. |
| Sputtered Iridium Oxide Film (SIROF) | High-charge-capacity coating for microelectrodes, enabling superior recording and stimulation. | Tip metal for chronic intracortical microelectrodes in humans, showing greater longevity than Pt [41]. |
Leveraging High Gamma Band (70-150 Hz) as a High-Fidelity Signal for ECoG-based BCIs
Electrocorticographic (ECoG) recordings of high-gamma activity (HGA, 70-150 Hz) have emerged as a particularly robust neural correlate of cognition and behavior, offering an optimal balance of spatial resolution, temporal fidelity, and signal stability for brain-computer interface (BCI) applications [44] [45]. Unlike scalp electroencephalography (EEG), which suffers from significant signal attenuation in high-frequency bands due to the skull's low-pass filtering effect, ECoG provides direct access to high-frequency neural signals generated by cortical networks [46] [47]. This technical comparison guide objectively evaluates the performance characteristics of ECoG-based HGA against alternative neural recording methodologies within the broader context of signal-to-noise ratio optimization for next-generation BCIs.
Comparative Signal Profiles Across Recording Modalities
Quantitative Performance Metrics
Table 1: Signal characteristics across neural recording modalities
| Recording Method | High-Gamma Signal Quality | Spatial Resolution | Temporal Resolution | Invasiveness | Stability |
|---|---|---|---|---|---|
| ECoG | High SNR, robust task modulation [44] [48] | ~1 cm (standard arrays) [8] | Millisecond [45] | Moderate (subdural) | Stable over ≥12 months [48] |
| scalp EEG | Low SNR, attenuated by skull [46] [47] | ~3-5 cm | Millisecond | Non-invasive | Session-dependent |
| Intracortical (LFP) | Excellent, correlates with spiking [49] | ~100-500 μm | Millisecond | High (penetrating) | Variable, tissue response |
| Subgaleal | Moderate HG preservation (70-110 Hz) [46] | ~1-2 cm | Millisecond | Minimal (extracranial) | Limited long-term data |
Table 2: High-gamma band specification across functional domains
| Cognitive Domain | Typical Frequency Range | Amplitude Modulation | Temporal Dynamics |
|---|---|---|---|
| Motor Control | 60-300 Hz [44] | Significant power increase [45] | Smooth time course with relatively slow transients [44] |
| Speech Processing | 70-150 Hz [48] [47] | Stable long-term responses [48] | Burst-like patterns during articulation [45] |
| Visual Perception | 70-150 Hz [49] | Tuned to stimulus features [49] | Stimulus-locked responses [50] |
| Auditory Processing | 70-110 Hz [46] | Robust envelope tracking [47] | Phasic responses to acoustic features [44] |
Signal Origin and Neural Correlates
High-gamma activity in ECoG reflects a broadband power increase that corresponds to localized cortical processing. Unlike narrowband gamma oscillations (30-80 Hz), HGA represents a broadband phenomenon that indexes the average firing rate of neuronal populations near the electrode [49]. Intracortical recordings have demonstrated that HGA in local field potentials (LFP) tightly correlates with multi-unit spiking activity, suggesting it provides a direct window into local neural computation [49]. ECoG electrodes capture this activity from the cortical surface with minimal spatial smearing, as evidenced by similar stimulus tuning properties between ECoG and directly adjacent intracortical microelectrodes [49].
Experimental Protocols for HGA Characterization
ECoG Signal Acquisition and Processing
Table 3: Essential research reagents and materials
| Item | Function | Example Specifications |
|---|---|---|
| ECoG Electrode Array | Neural signal acquisition | 1 cm inter-electrode distance, 256 channels [36] |
| Biosignal Amplifier | Signal conditioning | 2 kHz sampling rate, high input impedance [46] |
| Hybrid Micro/Macro Arrays | Signal validation | Simultaneous LFP/ECoG recording [49] |
| Flexible Electrode-Mesh | Chronic recording | Parylene-C substrate with fenestrations [8] |
| Guidewire Deployment | Minimally invasive implantation | Millimeter-sized skull access [36] |
Standard HGA Processing Pipeline:
- Signal Acquisition: Record continuous ECoG at sampling rates ≥2 kHz to adequately capture high-frequency components [46]
- Spectral Decomposition: Apply time-frequency analysis (e.g., multitaper method) to compute power spectral density [49]
- High-Gamma Extraction: Bandpass filter in 70-150 Hz range (adjusting for task-specific variations) [44]
- Power Calculation: Compute amplitude envelope via Hilbert transform [47]
- Baseline Normalization: Express HGA as percent change from pre-stimulus or resting baseline [48]
Paradigms for Functional Mapping
Motor Tasks: Participants perform repetitive motor acts (hand clenching, foot movement) while ECoG is recorded from sensorimotor cortices. HGA power increases are quantified during active movements compared to rest periods [44] [45].
Auditory/Speech Tasks: For speech decoding applications, participants listen to or produce words/syllables while ECoG is recorded from temporal and frontal regions. Stimulus-locked or production-locked HGA responses are analyzed for envelope tracking or articulation-related modulation [48] [47].
Visual Perception Tasks: Participants view category-specific visual stimuli (bodies, faces, objects) while ECoG is recorded from occipitotemporal cortex. Body-selective responses in HGA emerge at 100-150 ms post-stimulus, demonstrating rapid category discrimination [50].
Signaling Pathways and Experimental Workflows
Neural signaling pathway for HGA-based BCI
Stability and Long-Term Performance Evidence
Longitudinal studies provide compelling evidence for HGA stability in chronically implanted ECoG arrays. Research with an ALS participant demonstrated stable speech-related HGA responses over 12 months, with preserved signal-to-noise ratios and activation patterns across multiple syllables [48]. This stability is crucial for clinical BCI applications requiring reliable long-term operation. The observed preservation of HGA tuning properties over extended periods suggests that ECoG provides a robust platform for permanent assistive communication devices [48].
Emerging Technologies and Future Directions
Advanced Electrode Designs
Next-generation ECoG technologies focus on improving spatial resolution while maintaining broad coverage. High-density arrays (256 electrodes per 4 cm²) deployed via minimally invasive guidewire techniques now enable large-scale mapping with millimeter precision [36]. Flexible electrode-mesh designs with fenestrated structures allow better cortical conformity and simultaneous intracortical validation [8]. Subgaleal recording approaches represent an intermediate solution, capturing attenuated but measurable HGA signals from outside the skull [46].
Complementary Signal Integration
While HGA provides excellent spatial and temporal specificity, combining it with lower-frequency bands can enhance decoding performance. Research shows that integrating low-frequency (<16 Hz) EEG with HGA improves speech envelope reconstruction compared to either signal alone [47]. This multimodal approach leverages the complementary information across frequency domains, potentially boosting BCI performance for complex tasks like continuous speech decoding.
ECoG-based high-gamma activity represents a uniquely powerful signal modality for brain-computer interfaces, offering an optimal combination of spatial localization, temporal dynamics, and long-term stability. The systematic characterization of HGA properties across cognitive domains provides a solid foundation for designing next-generation BCI systems. While ECoG requires more invasive implantation than scalp EEG, its superior signal-to-noise ratio for high-frequency neural activity justifies this tradeoff for clinical applications requiring robust, high-performance communication and control. Continued technological innovations in electrode design, signal processing, and multimodal integration will further enhance the fidelity of HGA as a neural control signal for assistive technologies.
Decoding Motor Imagery from EEG: Machine Learning Strategies to Overcome Low SNR
Electroencephalography (EEG) provides a non-invasive window into brain activity, offering high temporal resolution for decoding motor imagery (MI)—the mental rehearsal of physical movements. A primary obstacle in this domain is the inherently low signal-to-noise ratio (SNR) of EEG signals, which are often contaminated by physiological artifacts and environmental noise. This guide compares modern machine learning (ML) strategies designed to overcome this challenge, framing them within the broader context of signal quality comparison across EEG, Electrocorticography (ECoG), and intracortical recordings. For researchers and scientists, understanding these trade-offs and the corresponding algorithmic innovations is critical for developing robust Brain-Computer Interfaces (BCIs) for neurorehabilitation and assistive technologies [51] [52] [53].
The SNR Landscape: EEG in Perspective
The choice of neural recording modality involves a direct trade-off between invasiveness, spatial resolution, and resultant signal quality. EEG sits at one end of this spectrum, providing a practical but noisy signal source.
- EEG (Non-invasive): Characterized by a low amplitude and low spatial resolution, EEG signals are susceptible to noise from muscle movements, eye blinks, and other environmental sources. This results in the lowest baseline SNR among the three modalities, making feature extraction and classification particularly challenging [53].
- ECoG (Minimally Invasive): Involving electrodes placed on the surface of the brain, ECoG offers higher spatial resolution and signal amplitude than EEG. It bypasses the signal-attenuating skull, leading to a higher SNR and richer feature content for motor decoding [53].
- Intracortical (Fully Invasive): Microelectrode arrays (MEAs) implanted directly into brain tissue can record from individual neurons. This provides the highest spatial resolution and SNR, capturing fine-grained neural activity but at the cost of maximum invasiveness and long-term stability concerns [53].
Table 1: Comparison of Neural Signal Recording Modalities for Motor Decoding
| Modality | Invasiveness | Spatial Resolution | Typical SNR | Primary Use Cases |
|---|---|---|---|---|
| EEG | Non-invasive | Low (cm) | Low | Research, portable & clinical BCIs, neurofeedback |
| ECoG | Minimally Invasive | Medium (mm) | Medium to High | Intraoperative monitoring, advanced BCI prototypes |
| Intracortical (MEA) | Fully Invasive | High (µm) | High | Fundamental neuroscience, high-performance BCIs |
The following conceptual workflow illustrates how machine learning strategies are integrated to combat low SNR at various stages of EEG signal processing:
Diagram 1: A conceptual workflow for decoding Motor Imagery from EEG signals, highlighting key ML strategies to overcome low SNR.
Comparative Analysis of Machine Learning Strategies
Recent advances employ sophisticated preprocessing, feature enhancement, and specialized deep learning architectures to extract robust patterns from noisy EEG data. The table below compares several state-of-the-art approaches.
Table 2: Comparison of Modern ML Strategies for MI-EEG Decoding
| Strategy (Year) | Core Methodology | Key Innovation | Reported Accuracy | Advantages | Limitations |
|---|---|---|---|---|---|
| Hybrid Preprocessing + ADBN [54] (2025) | Hybrid EMD + CWT; SPoC + CSP features; Adaptive Deep Belief Network. | End-to-end optimized preprocessing & classification. | 95.7% (BCI IV-2a) | High accuracy; Robust feature extraction. | Computational complexity. |
| Feature Reweighting [55] (2025) | CNN with Temporal & Channel Feature Score modules. | Suppresses irrelevant/noisy features via learned reweighting. | ~9.34% improvement (Physionet) | Effectively suppresses noise; Improved generalizability. | Module integration complexity. |
| HA-FuseNet [56] (2025) | End-to-end network with multi-scale dense connectivity & hybrid attention. | Lightweight design; Fuses local (CNN) and global (LSTM) features. | 77.89% (BCI IV-2a) | Handles spatial resolution variation; Lower compute. | Lower raw accuracy than some models. |
| AMD-KT2D [57] (2025) | 2D Spectrograms (OptSTFT); Guide-learner CNN with domain adaptation. | Knowledge transfer improves cross-subject generalization. | 96.75% (Subject-dependent) | Excellent for cross-subject use; High accuracy. | Depends on quality of 2D transformation. |
| HBA-Optimized BPNN [58] (2025) | HHT preprocessing; PCMICSP features; BPNN optimized with Honey Badger Algorithm. | Nature-inspired optimization avoids local minima in training. | 89.82% (EEGMMIDB) | Effective for non-linear, non-stationary signals. | Optimization can be complex. |
Detailed Experimental Protocols
To ensure reproducibility and provide depth, here are the detailed methodologies for two high-performing strategies from the comparison.
This protocol describes a comprehensive method that achieves high accuracy through advanced signal decomposition and an optimized deep learning model.
- Dataset: BCI Competition IV Dataset 2a.
- Preprocessing:
- Empirical Mode Decomposition (EMD): Decomposes the raw EEG signal into Intrinsic Mode Functions (IMFs) to isolate underlying oscillatory components.
- Continuous Wavelet Transform (CWT): Applied to the IMFs for multi-resolution time-frequency analysis, creating a detailed representation of the signal.
- Feature Extraction & Enhancement:
- Common Spatial Patterns (CSP): Extracts spatial features that maximize the variance between two different motor imagery classes (e.g., left hand vs. right hand).
- Source Power Coherence (SPoC): Integrated with CSP to enhance features by aligning them with the source of brain activity, improving robustness.
- Classification:
- Model: An Adaptive Deep Belief Network (ADBN) is used for the final classification of MI tasks.
- Optimization: The parameters of the ADBN are fine-tuned using the Far and Near Optimization (FNO) algorithm to achieve superior performance, resulting in reported metrics of 95.7% accuracy, 96.2% recall, and 95.9% precision on the BCI IV 2a dataset.
This protocol focuses on a novel architectural innovation within a CNN to dynamically suppress noise, offering significant performance improvements.
- Dataset: Physionet motor imagery dataset.
- Base Model: A Convolutional Neural Network (CNN) forms the backbone for initial feature map generation.
- Feature Reweighting Module:
- Temporal Feature Score (TFS): This sub-module analyzes the sequence of feature maps and learns a relevance score for each time point, down-weighting temporally noisy segments.
- Channel Feature Score (CFS): This sub-module learns the importance of each EEG channel (or feature map channel), suppressing contributions from uninformative or noisy channels.
- Score Fusion (SF): The scores from TFS and CFS are fused and used to reweight the original feature maps adaptively.
- Classification: The reweighted, "cleaner" feature maps are passed to the final fully connected layers for classification. This approach demonstrated a 9.34% performance gain over standard CNNs on the Physionet dataset.
The Scientist's Toolkit: Essential Research Reagents & Materials
Translating these algorithms from theory to practice requires a suite of key resources, from benchmark data to hardware.
Table 3: Essential Resources for MI-EEG Research
| Category | Item | Function & Application |
|---|---|---|
| Public Datasets | BCI Competition IV (2a & 2b) [54] [55] | Benchmarking algorithms; Provides multi-class MI EEG data from multiple subjects. |
| Physionet EEG Motor Movement/Imagery Dataset [54] [55] | Evaluating generalizability; Contains a large set of MI and execution tasks. | |
| Algorithms & Features | Common Spatial Patterns (CSP) [54] [58] | A foundational spatial filtering technique for feature extraction in MI tasks. |
| Empirical Mode Decomposition (EMD) [54] | Adaptive, data-driven signal decomposition for non-stationary EEG analysis. | |
| Wavelet Transforms (CWT/DWT) [54] [58] | Provides time-frequency representation of signals, crucial for localizing MI patterns. | |
| Hardware | Emotiv Epoc Flex (32-ch) [57] | A research-grade, flexible saline-based EEG system for data acquisition. |
| Portable, Low-channel Systems [59] | Systems focusing on a reduced number of central electrodes for practical, real-world BCI applications. | |
| Computational Tools | Elastic Net Regression [59] | Regularized linear regression used for channel selection and signal prediction. |
| Honey Badger Algorithm (HBA) [58] | A nature-inspired metaheuristic used for optimizing neural network parameters and avoiding local minima. |
The frontier of motor imagery decoding from EEG is being pushed forward by machine learning strategies that directly confront the challenge of low SNR. As evidenced by the comparative data, approaches that integrate advanced preprocessing (like EMD and CWT), noise-aware architectures (like feature reweighting), and sophisticated optimization (like HBA and FNO) are setting new benchmarks in classification accuracy. The trend is moving towards models that are not only accurate but also computationally efficient and generalizable across subjects, a crucial step for clinical viability. Future work will likely focus on standardizing datasets and protocols while further refining these adaptive, lightweight models to bridge the gap between laboratory research and real-world neurorehabilitation applications [56] [60].
The development of brain-computer interfaces (BCIs) for restoring communication represents one of the most promising clinical applications of modern neuroscience. For individuals with amyotrophic lateral sclerosis (ALS), brainstem stroke, or other neurological conditions that result in locked-in syndrome (LIS), BCIs offer the potential to restore the fundamental human ability to communicate. However, a significant translational challenge emerges when patients progress to complete loss of voice: conventional supervised learning approaches for speech decoding require time-aligned acoustic ground truth for training, which becomes impossible when users cannot speak. This limitation has catalyzed research into unsupervised methods that can detect speech production directly from neural signals without labeled data.
Central to this endeavor is the fundamental relationship between neural recording modality and signal fidelity. Electrocorticography (ECoG), which records electrical activity directly from the cortical surface, provides a critical balance of invasiveness and signal quality, positioned between non-invasive electroencephalography (EEG) and highly invasive intracortical microelectrodes. ECoG offers superior spatial resolution and signal-to-noise ratio (SNR) compared to EEG, while avoiding the tissue damage associated with penetrating electrodes [9] [15]. This technical sweet spot makes ECoG particularly suitable for clinical BCIs, especially for decoding complex processes like speech production. Recent advances have demonstrated that ECoG signals can be leveraged to detect voice activity without acoustic labels, representing a crucial step toward BCIs for completely locked-in patients who can no longer provide supervised training data [34] [61].
Neural Recording Modalities: A Quantitative Comparison of Signal Fidelity
The selection of neural recording modality fundamentally determines the information bandwidth available for brain-computer interfaces. Each technique offers distinct trade-offs between signal quality, spatial resolution, temporal resolution, and clinical risk, making certain modalities better suited for specific applications.
Table 1: Comparison of Neural Recording Modalities for BCI Applications
| Modality | Spatial Resolution | Temporal Resolution | SNR Characteristics | Invasiveness | Primary Clinical Considerations |
|---|---|---|---|---|---|
| EEG | ~1-2 cm (limited by skull/signal spread) [9] | Excellent (milliseconds) [9] | Lower; susceptible to blink, eye movement, and muscle artifacts [15] | Non-invasive | Easy to apply but limited signal fidelity for complex decoding |
| ECoG | ~0.5-1 cm [10] | Excellent (milliseconds) | Higher than EEG; less susceptible to distant artifacts [15] [10] | Minimally invasive (requires craniotomy) | Ideal balance for clinical BCIs; better signal quality than EEG with acceptable risk |
| Endovascular (Stentrode) | ~0.5-1 cm [10] | Excellent (milliseconds) | Comparable to ECoG; signal quality not significantly different from conventional neural sensors [10] | Minimally invasive (via blood vessels) | No craniotomy required; recently shown to have comparable SNR to ECoG |
| Intracortical (Microelectrodes) | ~0.05-0.1 mm (single neuron level) | Excellent (milliseconds) | Highest for unit recordings; potential signal degradation from tissue response over time | Highly invasive (penetrates brain tissue) | Highest signal quality but increased biological risk and potential for scar formation |
Quantitative analyses demonstrate that ECoG provides substantially better signal quality compared to non-invasive alternatives. Simultaneous recordings of ECoG and EEG during eye blinks and saccades have revealed that blink-related artifacts are significantly more prominent in EEG recordings, with ECoG showing markedly better signal-to-noise characteristics except at electrodes positioned immediately adjacent to the eyes [15]. Furthermore, recent comparisons of endovascular, subdural, and epidural signals have found that the quality of signals (bandwidth and signal-to-noise ratio) recorded via endovascular neural interfaces is not significantly different from conventional subdural ECoG arrays, supporting the viability of minimally-invasive approaches for high-quality neural recording [10].
Unsupervised Voice Activity Detection from ECoG: Methodology and Experimental Validation
Experimental Protocol and Participant Profile
Groundbreaking research published in 2025 has demonstrated the feasibility of unsupervised voice activity detection (VAD) directly from ECoG signals, representing a critical advancement for BCIs targeting completely locked-in patients [34] [61]. This pilot study involved a single clinical trial participant (62-year-old male) with dysarthria due to ALS, who had been implanted with two ECoG arrays containing 64 electrodes each covering speech and upper-limb cortical areas. The participant retained some speaking capability, but his continuous speech was mostly unintelligible due to his neurological condition, scoring only 1 point out of 5 on the ALSFRS-R speech measure.
In the experimental paradigm, the participant was instructed to read single words presented on a monitor. Each target word was displayed for 2 seconds followed by an inter-trial interval of 3 seconds. The word pool consisted of 50 words, with each word repeated twice per session across 10 recording days over a 9-week period. This design ensured that the majority of recording time contained no speech activity, which researchers actively exploited to automatically assign identified segments as either speech or non-speech classes [34].
Signal Processing and Feature Extraction Pipeline
The research utilized a sophisticated signal processing workflow to extract speech-related features from raw ECoG signals:
- Preprocessing: Bad channels were removed based on visual inspection, followed by application of a common average referencing filter across each grid independently.
- Channel Selection: Researchers selected the top 64 channels with the strongest activation during overt speech production, identified in previous studies with the same participant.
- Filtering: A bandpass filter (IIR Butterworth, 4th order) extracted the broadband high-gamma (HG) band in the range of 70-170 Hz, with an additional notch filter (IIR Butterworth, 4th order) to attenuate the first harmonic of line noise in the range of 118-122 Hz.
- Feature Extraction: For each channel, logarithmic power features were computed using a window size of 50 ms and a frame shift of 10 ms.
- Normalization: All features were normalized to zero mean unit variance (z-score normalization) [34].
The high-gamma band was specifically targeted because it has been shown to track closely with the location and timing of speech production neural activation and has been successfully employed in previous studies for speech BCIs [34].
Unsupervised Learning Approach
The methodological innovation centered on a graph-based clustering approach to identify temporal segments of speech production from ECoG signals without using acoustic labels. This technique identified structural patterns with a fixed temporal context in high-gamma activity extracted from ECoG recordings. The estimated clustering labels were then used to train three different classification models previously employed in the literature for neural voice activity detection tasks: a recurrent neural network (RNN), a convolutional neural network (CNN), and logistic regression (LR) [34].
Performance Evaluation and Comparative Analysis
Quantitative Performance Metrics
The unsupervised VAD approach achieved remarkable performance despite the challenge of operating without acoustic ground truth for training:
- Timing Accuracy: The method achieved a median timing error of approximately 530 ms with respect to the actual spoken speech, demonstrating reasonable temporal alignment capability [34] [61].
- Real-time Latency: When embedded into a real-time BCI framework, the approach provided VAD results with a latency of only 10 ms, making it suitable for practical BCI applications where responsiveness is critical [34].
- Generalization: The model showed promising generalization capability when tested on unseen words from a larger word pool of 688 words that did not appear in the training data [34].
Table 2: Performance Comparison of ECoG-Based Voice Activity Detection Methods
| Method | Training Paradigm | Temporal Alignment Error | Inference Latency | Applicability to Locked-In Patients | Key Advantages |
|---|---|---|---|---|---|
| Unsupervised Graph-Based Clustering [34] [61] | Fully unsupervised; no acoustic labels required | ~530 ms median error | 10 ms | High - designed specifically for patients without voice | Does not require acoustic recordings; enables BCI for completely locked-in patients |
| Supervised VAD with Acoustic Labels [34] | Supervised; requires time-aligned acoustic recordings | Lower than unsupervised approach | Comparable (10-50 ms typically) | Low - requires patients to produce audible speech | Higher temporal precision when acoustic labels are available |
| ECoG-based Speech Decoding [62] | Various supervised approaches | Varies by method | Varies by method | Limited to patients with some preserved speech | Can decode speech content, not just detection |
Comparative Analysis with Alternative BCI Approaches
The search for effective BCIs has yielded multiple technological approaches, each with distinct advantages and limitations for clinical application:
- Neuralink: Utilizes an ultra-high-bandwidth implantable chip with thousands of micro-electrodes threaded into the cortex by a robotic surgeon. As of 2025, the company reported that five individuals with severe paralysis are using Neuralink to control digital and physical devices with their thoughts [62].
- Synchron: Employs a minimally invasive endovascular approach (Stentrode) delivered via blood vessels through the jugular vein, avoiding open-brain surgery. Clinical trials demonstrated that participants with paralysis could control computers for texting and other functions using thought alone [62].
- Precision Neuroscience: Developed an ultra-thin electrode array designed to be inserted between the skull and brain through a minimal dural opening. Their Layer 7 device received FDA 510(k) clearance in April 2025 for implantation durations of up to 30 days [62].
The unsupervised ECoG VAD approach distinguishes itself by specifically addressing the critical challenge of patients who have completely lost voice capability, whereas many existing BCI approaches still require some preserved speech or movement for system calibration and training.
Essential Research Reagents and Experimental Tools
Conducting research on unsupervised voice activity detection from ECoG requires specialized materials and methodological components. The following table summarizes key research reagents and their functions based on the methodologies employed in recent groundbreaking studies.
Table 3: Essential Research Reagents and Methodological Components for ECoG-based VAD Research
| Reagent/Component | Specification | Function in Research | Example from Literature |
|---|---|---|---|
| ECoG Electrode Arrays | 64-electrode arrays (4 mm center-to-center spacing, 2 mm diameter) [34] | Neural signal acquisition from cortical surface | Two 64-electrode arrays covering speech and upper-limb cortical areas [34] |
| Neural Signal Amplifier | NeuroPort System (Blackrock Neurotech) [34] | Signal digitization and initial processing | Sampling rate of 1 kHz for neural data acquisition [34] |
| High-Gamma Band Features | 70-170 Hz frequency range [34] | Tracking speech production neural activation | Extracted using IIR Butterworth bandpass filter (4th order) [34] |
| Graph-Based Clustering Algorithm | Custom implementation [34] | Identifying speech segments without labels | Structural pattern identification in HG activity with fixed temporal context [34] |
| Classification Models | RNN, CNN, Logistic Regression [34] | Voice activity detection from neural features | Three models trained on cluster-derived labels for performance comparison [34] |
| Microelectrode Arrays | Utah array, Neuralace [62] | Higher-resolution neural recording | Blackrock Neurotech's development of flexible lattice for less invasive coverage [62] |
The development of unsupervised voice activity detection from ECoG signals represents a pivotal advancement in brain-computer interface technology, directly addressing the critical challenge of restoring communication to completely locked-in patients who can no longer provide supervised training data. This approach leverages the superior signal-to-noise ratio and spatial resolution of ECoG compared to non-invasive alternatives like EEG, while demonstrating that fully unsupervised methods can achieve reasonable temporal alignment (median error ~530 ms) with exceptionally low real-time latency (10 ms).
The implications for clinical BCI applications are substantial. As noted in the research, "To the best of our knowledge, our results show for the first time that speech activity can be predicted purely from unlabeled ECoG signals, a crucial step toward individuals who cannot provide this information anymore due to their neurological condition, such as patients with locked-in syndrome" [34] [61]. This capability could fundamentally transform the landscape of communication BCIs, extending their potential to patient populations who were previously excluded from such technologies due to their inability to produce audible speech for system calibration.
Future research directions should focus on improving the temporal precision of unsupervised alignment, extending the approach to continuous speech decoding rather than isolated voice activity detection, and validating the methods across larger and more diverse patient populations. Additionally, integration of these unsupervised approaches with emerging minimally-invasive ECoG technologies, such as endovascular stents and thin-film cortical surfaces, promises to enhance the clinical viability and accessibility of BCIs for the patients who stand to benefit most from restored communication capabilities.
Strategies for SNR Enhancement and Overcoming Common Recording Challenges
Within the operating room, intraoperative neurophysiological monitoring (IONM) provides a critical window into the functional integrity of a patient's nervous system. The somatosensory evoked potential (SEP), elicited by electrical stimulation of a peripheral nerve and recorded over the somatosensory pathways, is a fundamental component of this monitoring. A primary technical challenge in acquiring robust SEPs is their extremely low amplitude, which is often dwarfed by background noise. The signal-to-noise ratio (SNR) is thus a paramount determinant of SEP quality and reliability. While multiple factors influence SNR, the repetition rate of the electrical stimulus is a powerful and readily adjustable parameter. Optimizing this rate can dramatically enhance SNR, enabling faster averaging and providing surgeons with more timely feedback to prevent iatrogenic neurological injury [63] [64]. This guide objectively compares the effects of different stimulation rates on SEP SNR, situating these findings within the broader research on signal optimization across neural recording modalities, from non-invasive EEG to invasive intracortical methods.
Comparative Analysis of Stimulation Rates on SEP SNR
The relationship between stimulation rate and SEP SNR is not linear and varies based on the monitored nerve and the required recording duration. The following analysis synthesizes key experimental findings to guide parameter selection.
Table 1: Impact of Stimulation Rate on Median Nerve SEP SNR
| Stimulation Rate (Hz) | Recording Duration | Median SNR (N20) | Key Physiological Findings |
|---|---|---|---|
| 12.7 Hz | ≤ 5 seconds | 22.9 (Highest) | Rapid noise reduction outweighs cortical amplitude decay [65] |
| 4.7 Hz | ≤ 5 seconds | Lower than 12.7 Hz (p = 1.5e-4) | Larger amplitude, but slower noise averaging [65] |
| Increasing Rate (2.7 - 28.7 Hz) | N/A | Correlates with latency increase & amplitude decay | Amplitude decay observed at cortical sites, but not at subcortical or peripheral sites [65] |
Table 2: Comparative Optimal Rates for Different Nerves
| Nerve / Procedure | Optimal Stimulation Rate | Key Contextual Factors |
|---|---|---|
| Medianus Nerve | 12.7 Hz (for short recordings) | For recording durations of ~5 seconds; ideal for rapid assessment [65] |
| Tibial Nerve | ~4.7 Hz (for all durations) | High-frequency optimization not effective; lower rates consistently superior [65] |
| Longer Medianus Recordings | ~4.7 Hz | For durations longer than ~5 seconds, the benefit of rapid averaging diminishes [65] |
| General SEP Monitoring | Patient-specific optimization recommended | Individual anatomy, pathology, and anesthetic effects necessitate a personalized approach [63] |
Experimental Protocols and Methodologies
The comparative data presented above are derived from rigorous intraoperative studies. This section details the standard methodologies employed to ensure reproducible and high-quality results.
Data Acquisition and Stimulation Protocol
In a typical optimization study, SEPs are recorded during surgeries (e.g., 22 cases as in [65]) using standard electrode montages. The key experimental manipulation involves varying the repetition rate of stimulus presentation across a wide range (e.g., 2.7 Hz to 28.7 Hz) while keeping other stimulation parameters (pulse width, intensity) constant. For median nerve SEPs, the primary signal of interest is often the N20 potential, a cortical component recorded over the contralateral scalp. The corresponding P37 potential is monitored for tibial nerve stimulation [65] [66].
SNR Calculation and Sampling
To assess the impact of recording duration, researchers use a random sampling approach. From the continuous data, a number of sweeps corresponding to short recording durations (e.g., up to 20 seconds) are randomly selected. The SNR is calculated for each of these short-duration epochs and then averaged across multiple trials to obtain a stable estimate for each stimulation rate and duration condition. This process allows for a systematic comparison of how quickly a reliable response can be obtained at different rates [65].
The Personalized Optimization Workflow
A generalized workflow for personalizing IONM parameters, including stimulation rate, can be summarized as follows:
This workflow highlights the iterative process of varying parameters like electrode location, filter settings, and stimulation rate to find the best SNR for an individual patient, a practice that is more flexible in IONM than in standard clinical neurophysiology [63] [64].
Physiological Basis and Signaling Pathways
The differential effect of stimulation rate on cortical versus subcortical structures has a clear neurophysiological basis. High-frequency stimulation induces a phenomenon known as attenuation, where the amplitude of the cortical response decreases with increasing rate, while subcortical and peripheral responses remain stable [65]. This is thought to be due to the synaptic properties of the cortical generators of the N20 potential, which are unable to follow high-frequency inputs as effectively as the axonal pathways that generate subcortical potentials.
This diagram illustrates the somatosensory pathway. The cortical response (N20) is susceptible to amplitude decay at high stimulation rates, whereas the responses from subcortical structures like the brainstem and thalamus remain stable [65].
The Scientist's Toolkit: Research Reagent Solutions
Optimizing SEP recordings relies on a suite of specialized tools and concepts, from physical hardware to signal processing techniques.
Table 3: Essential Tools for SEP Research and Clinical IONM
| Tool / Concept | Category | Function in SEP Optimization |
|---|---|---|
| Multi-electrode Array | Hardware | Enables personalized selection of active and reference electrode locations to maximize signal amplitude [63] [64] |
| Frequency Filtering | Signal Processing | "Aggressive" narrowing of passbands (e.g., high-pass filter >1Hz) to remove irrelevant brain activity and noise, boosting SNR [63] |
| Averaging Technique | Signal Processing | Classic method to improve SNR by summing time-locked responses, canceling random noise; required number of averages is reduced with higher SNR [65] [63] |
| Stimulation Rate | Stimulation Parameter | The key parameter being compared; directly influences cortical response amplitude and speed of noise reduction via averaging [65] |
| Personalized Approach | Methodological Paradigm | The overarching principle of tailoring parameters (electrodes, filter, rate) to the individual patient rather than relying solely on population norms [63] [64] |
Broader Context: SNR Across Neural Recording Modalities
The challenge of optimizing SNR is a universal constant in neural engineering, extending far beyond intraoperative SEPs. Research into brain-computer interfaces (BCIs), for instance, grapples with the same fundamental trade-offs. Invasive techniques like electrocorticography (ECoG) and intracortical recordings provide high-fidelity signals with excellent SNR, enabling complex decoding of tasks like handwriting [67]. However, their surgical requirements pose significant risks and limit scalability [67] [68].
Non-invasive alternatives, primarily electroencephalography (EEG), are safer and more accessible but suffer from a low SNR and spatial resolution due to signal attenuation by the skull [69] [67]. To overcome this, EEG-based research employs sophisticated feature extraction (e.g., Filter Bank Common Spatial Patterns, phase-locking values) and advanced deep-learning models to decode neural intent from noisy signals [68]. The quest for optimal SEP parameters is thus a specific manifestation of a universal challenge: extracting a meaningful neural signal from a noisy background across all recording modalities, where parameter tuning is always critical for success.
The drive towards high-density neural arrays is a cornerstone of modern neuroscience, enabling researchers to capture brain activity with unprecedented spatial resolution. However, this very density introduces a significant technical hurdle: crosstalk, the undesired electrical coupling between closely spaced signal pathways. This form of signal contamination can distort recordings, lead to data misrepresentation, and ultimately compromise the validity of scientific conclusions [70]. As the field pushes for implants with thousands of channels, the compact design necessary for high channel counts results in reduced line spacing and smaller safety margins within the device's interconnects, connectors, and cables [70]. This physical proximity creates an environment where the signal from one channel can "bleed" into its neighbors, effectively blurring the spatial resolution that high-density arrays were designed to achieve.
The risk of crosstalk is particularly acute in the context of signal-to-noise ratio (SNR) comparisons across different recording modalities. While non-invasive electroencephalography (EEG) is notoriously susceptible to artifacts from blinks and eye movements, even invasive techniques like electrocorticography (ECoG) are not immune to contamination, though they generally offer superior signal quality [15]. Intracortical recordings, which probe neural activity at the finest scale, face their own set of challenges, where factors like electrode-tissue interface impedance and the density of surrounding neurons significantly impact SNR [71]. Therefore, identifying and correcting for crosstalk is not merely a technical exercise but a critical step in data quality control, ensuring that recorded signals are a faithful representation of neural activity and not an artifact of the recording system itself [70].
Understanding Crosstalk: Mechanisms and Impact
The Physical Origins of Crosstalk
Crosstalk in high-density neural arrays arises from fundamental electrical principles. It occurs through two primary coupling mechanisms: resistive (or conductive) coupling and capacitive coupling [70]. Resistive coupling refers to a physical leakage current between adjacent interconnects, even when they are separated by an insulator. This type of coupling is generally independent of the signal's frequency.
In contrast, capacitive coupling is more complex and often more problematic. It arises from displacement currents generated by time-varying potentials along the transmission lines. A critical characteristic of capacitive crosstalk is that its magnitude increases with the frequency of the neural signal. This happens because the capacitive reactance (X_C = 1/ωC) decreases as frequency increases, providing a lower-impedance path for the signal to jump between lines [70]. This frequency dependence means that crosstalk contamination is not uniform across all neural signals; high-frequency components, such as those found in multi-unit activity (MUA), are far more susceptible to this form of interference than lower-frequency local field potentials (LFP) [70].
Consequences for Neural Data Interpretation
The impact of crosstalk extends beyond simple signal degradation; it can fundamentally alter the interpretation of neural data. A key analytical tool in neuroscience is signal coherence, which measures the linear relationship between two signals as a function of frequency. In a pristine recording, coherence between two channels should be determined solely by the biological relationship of the underlying neural populations and the physical distance between the electrodes on the cortex.
However, crosstalk introduces a non-biological confound. Research has shown that with crosstalk contamination, signal coherence can become highest for channels that are wired adjacently in the device's routing layout, even if the corresponding electrodes are placed far apart on the cortical surface [70]. This artificial inflation of coherence between physically distant brain regions can lead to erroneous conclusions about functional connectivity. The problem is most pronounced for high-frequency activity (above 300 Hz), where capacitive crosstalk is strongest, directly threatening the integrity of multi-unit activity analysis and the decoding of fine-scale neural patterns [70].
Experimental Comparison of Recording Modalities
A critical step in understanding signal quality is the direct comparison of different recording technologies. The following table summarizes key performance characteristics of EEG, ECoG, and intracortical microelectrodes, highlighting their inherent advantages and vulnerabilities to contamination.
Table 1: Performance Comparison of Neural Recording Modalities
| Recording Modality | Typical Spatial Resolution | Key Signal Contamination Risks | Typical SNR Considerations | Best Use Cases |
|---|---|---|---|---|
| EEG (Non-invasive) | ~1 cm | High vulnerability to blink, eye movement, and muscle artifacts [15]. | Lower SNR due to signal attenuation by skull and scalp. | Non-invasive brain monitoring, coarse-scale brain dynamics. |
| ECoG (Intracortical) | 1–10 mm | Less susceptible to biological artifacts than EEG, but still affected by blinks when recorded from prefrontal areas [15]. | Higher SNR than EEG; signal quality can be compromised by crosstalk in high-density arrays [70]. | Mapping of large-scale cortical networks, clinical epilepsy monitoring. |
| Micro-ECoG (μECoG) | Sub-millimeter to ~1.72 mm | Subject to crosstalk from densely routed interconnects; can resolve high-gamma activity with high fidelity [70] [72]. | Higher spatial resolution and SNR (e.g., 48% higher SNR than macro-ECoG) improve feature detection [72]. | Fine-scale mapping of functional areas, high-accuracy speech decoding [72]. |
| Intracortical Microelectrodes | Single Neuron | Biological noise from simultaneous firing of nearby neurons; thermal noise from electrode-tissue interface [71]. | SNR is more dependent on electrode impedance and neural density than contact size [71]. | Single-unit and multi-unit activity recording, studying neural spiking dynamics. |
Quantitative Insights from High-Density Arrays
The push for higher density is driven by the rich, fine-scale structure of neural information. Studies using micro-electrocorticography (μECoG) arrays with sub-millimeter to 1.72 mm inter-electrode spacing have demonstrated the tangible benefits of increased resolution. These arrays offer up to 57 times higher electrode density and a 48% higher signal-to-noise ratio (SNR) compared to conventional macro-ECoG arrays [72]. This enhanced signal quality directly translates to improved performance, enabling a 35% improvement in decoding accuracy for tasks like speech decoding from the sensorimotor cortex [72].
The superiority of high-density sampling is further confirmed by analyzing the distinctness of neural signals. The high-gamma band (70-150 Hz) activity, which correlates with local neural firing, shows low inter-electrode correlation (r = 0.1-0.3) at spacings of just 4 mm [72]. This indicates that articulatory and other neural properties are unique at a millimeter scale, meaning that coarse sampling inevitably misses or blurs critical information. Consequently, decoding models that leverage this high-density, high-SNR data, particularly non-linear models, consistently outperform those using data from standard-resolution arrays [72].
Methodologies for Identification and Correction
Experimental Protocols for Detecting Crosstalk
Identifying crosstalk requires systematic experimental design and analysis. A proven protocol involves the following key steps:
- In Vivo Recording with Layout Awareness: Record neural signals, such as somatosensory evoked potentials (SEPs) and multi-unit activity (MUA), using a state-of-the-art high-density array [70]. Throughout the experiment, meticulously track the physical routing layout of the electrodes—knowing which channels are adjacent in the implant's wiring is crucial for subsequent analysis.
- Compute Signal Coherence Maps: Calculate signal coherence between a reference channel and all other channels. Coherence is a frequency-domain measure of linear relationship between two time series [70].
- Analyze Coherence vs. Physical and Routing Distance: Compare the computed coherence against two different distance metrics: the inter-electrode distance on the cortex and the proximity in the routing layout [70].
- Identify Crosstalk Contamination: A clear indicator of crosstalk is elevated signal coherence between channels that are closely wired together in the routing layout, even when their corresponding electrodes are physically far apart on the cortical surface. This pattern is especially significant if the elevated coherence is observed in high-frequency bands ( >300 Hz), where capacitive crosstalk is most pronounced [70].
Table 2: Key Experimental Reagents and Tools for Crosstalk Research
| Research Reagent / Tool | Function in Experiment | Specific Example / Properties |
|---|---|---|
| Flexible Polyimide-Based Microelectrode Array | Chronic neural recording interface; conforms to cortical surface. | Arrays with 50 µm radius Pt electrodes; high-density configurations (e.g., 128-256 channels) [70] [72]. |
| Crosstalk Back-Correction Algorithm | Software tool to computationally estimate and remove crosstalk from recorded data. | Infers a "zero-crosstalk" scenario; validates by reduced coherence between closely-routed channels [70]. |
| Neuropixels Ultra Probe | High-density silicon probe for intracortical recording. | Ultra-high site density for improved cell detection and classification; better spatial precision for spike sorting [73]. |
| LCP-TF μECoG Array | High-density surface array for human intra-operative recording. | Liquid crystal polymer thin-film array; 1.33-1.72 mm inter-electrode distance; enables high-fidelity speech decoding [72]. |
| Computational Crosstalk Model | Simulates electrical coupling in the recording chain based on impedance measurements. | A six-block system model derived from active/passive transmission lines; used to predict crosstalk levels [70]. |
A Workflow for Crosstalk Correction
Once crosstalk is identified, researchers can apply a correction algorithm to mitigate its effects. The following diagram illustrates a generalized workflow for a crosstalk back-correction process, synthesized from contemporary research methods.
Diagram Title: Crosstalk Back-Correction Workflow
This workflow begins with the contaminated data and uses a model of the recording system to simulate the expected crosstalk. The core of the process is the application of a back-correction algorithm, which computationally infers what the signals would look like under a zero-crosstalk scenario [70]. The success of the correction is validated by observing a drop in the anomalous coherence between closely-routed channels after the algorithm is applied, corroborating that the contamination has been reduced [70].
Emerging Technologies and Future Directions
The challenge of crosstalk is catalyzing innovation in both hardware and signal processing. Next-generation neural probes are being designed specifically to overcome the limitations of density and signal quality. For instance, the Neuropixels Ultra probe features much smaller and denser recording sites than previous designs, functioning as an "implantable, voltage-sensing camera" [73]. This enhanced density allows it to more accurately capture the spatial electrical field footprint of individual neurons, improving the detection and classification of different brain cell types and increasing the yield of sortable neurons [73].
Concurrently, there is a growing emphasis on on-implant signal processing. With channel counts soaring into the thousands, transmitting all raw data wirelessly becomes a bottleneck due to power and bandwidth constraints [74]. The solution is to integrate efficient signal processing directly onto the implant. Techniques for spike detection, data compression, and spike sorting are being optimized for ultra-low-power operation, reducing the volume of data that must be transmitted without losing critical information [74]. This in-situ processing not only alleviates transmission problems but also opens the door to real-time crosstalk mitigation strategies in future brain-computer interfaces.
The pursuit of high-fidelity neural recordings is a delicate balance between increasing channel density and preserving signal integrity. Crosstalk represents a significant threat to this goal, capable of introducing spatially structured artifacts that can be misinterpreted as genuine neural phenomena. As the data and methodologies discussed herein demonstrate, combating crosstalk requires a multi-faceted approach: a thorough understanding of its electrical origins, rigorous experimental protocols for its detection, and robust computational methods for its correction.
The comparison of recording modalities clarifies that while no technology is entirely free from contamination risks, high-density arrays like μECoG offer substantial improvements in SNR and spatial resolution when crosstalk is properly managed. The ongoing development of ultra-dense probes and intelligent, on-implant processing circuits promises a future where researchers can simultaneously record from thousands of neurons with confidence in the veracity of their data. For now, a critical takeaway for every neuroscientist is that validating recorded data against the routing layout of the implant must become a standard and non-negotiable step in the data quality control pipeline [70].
Material and Fabrication Solutions for Chronic Recording Stability and Biocompatibility
The pursuit of stable, high-fidelity neural recordings over chronic timescales is a fundamental challenge in neuroscience and the development of clinical brain-computer interfaces (BCIs). The performance and longevity of neural interfaces are largely dictated by the foreign body response, a neuroinflammatory cascade initiated upon implantation. This response, characterized by glial scar formation, neuronal loss, and degradation of the electrode-tissue interface, leads to a progressive decline in recording quality [75] [76]. The core triggers of this failure include the significant mechanical mismatch between conventional rigid electrode materials and soft brain tissue, and the ensuing biological incompatibility [77] [76]. This guide objectively compares emerging material and fabrication solutions designed to mitigate these issues, framing the discussion within the critical context of achieving superior signal-to-noise ratio (SNR) for electrophysiological recording modalities.
Comparative SNR and Performance of Recording Modalities
The choice of recording modality and its material properties directly influences the quality of the acquired neural signal. The table below summarizes key performance characteristics of different approaches.
Table 1: Performance Comparison of Neural Recording Modalities
| Recording Modality | Spatial Resolution | Key Signal Components | Invasiveness & Stability | Representative SNR/Amplitude Findings |
|---|---|---|---|---|
| Electroencephalography (EEG) | Low (cm-scale) | Scalp-level potentials | Non-invasive; Stable but low signal quality | Susceptible to noise; suboptimal SNR [76]. |
| Electrocorticography (ECoG) | Medium (mm-scale) | Cortical surface potentials | Subdural; Stable over months [20] | SEP recordings showed similar SNR to intracortical MEAs in pigs [20]. |
| micro-Electrocorticography (µECoG) | High (sub-mm-scale) | High gamma (HG) power | Subdural; Chronic stability [72] [20] | 48% higher SNR vs. macro-ECoG/SEEG; 57x higher spatial density [72]. |
| Intracortical Microelectrodes (IME) | Very High (µm-scale) | Single-unit action potentials, LFPs | Penetrating; Chronic stability challenges [75] [20] | Higher amplitude SEPs and more high-frequency power vs. µECoG; similar SNR [20]. |
Material Solutions for Enhanced Biocompatibility and Stability
Innovative materials are being developed to address mechanical mismatch and the neuroinflammatory response. The following table compares several advanced solutions.
Table 2: Comparison of Material and Fabrication Solutions for Chronic Recording
| Solution Category | Core Mechanism of Action | Key Advantages | Experimental Evidence & Performance Data |
|---|---|---|---|
| Mechanically-Adaptive, Resveratrol-Eluting (MARE) Probes | Combines tissue-mimicking flexibility with local antioxidant delivery to mitigate oxidative stress [77]. | - Dry-state stiffness aids insertion- Post-implantation softening (E: ~10 MPa)- Sustained drug release (up to 60 days) [77] | - Significantly improved recording performance vs. rigid silicon controls- More stable impedance- Higher neuronal density and reduced glial scarring [77] |
| PEDOT:PSS-Based Bioelectronics | Conductive polymer offering high conductivity and mechanical compliance similar to neural tissue [78]. | - High conductivity and charge injection capacity- Low elastic modulus (0.1-10 MPa)- Solution-processable for flexible devices [78] | - Enhances signal-to-noise ratio (SNR)- Reduces interfacial impedance- Improves integration with cortical tissue [78] |
| Antimicrobial Coatings (TNA) | Nanostructured coating (TiO₂) to reduce microbial invasion and subsequent inflammation [79]. | - Reduces abundance of invasive microbes- Mitigates chronic neuroinflammation | - Correlated with fewer expressed neuroinflammatory markers at 4 and 12 weeks- Provides a platform for improved chronic recording [79] |
| Targeted Drug Delivery (DEXSPPIN) | Nanoparticles target vascular injury sites to deliver anti-inflammatory drugs (dexamethasone) locally [80]. | - Simultaneously augments hemostasis and drug delivery- Avoids systemic side effects- Biphasic drug release profile | - Significantly increased active electrode yield over 8 weeks- Reduced neuron degeneration and neuroinflammation [80] |
Detailed Experimental Protocols for Key Studies
Protocol: Evaluation of MARE Neural Probes
Objective: To compare the chronic recording performance and tissue response of MARE probes against industry-standard silicon probes [77].
Methodology:
- Device Implantation: MARE probes (50 µm thick, 8 recording sites) and rigid silicon NeuroNexus probes were implanted into the rat motor cortex.
- Neural Recording: Extracellular single-unit recordings were performed over acute (4-week) and chronic (12-week) time points. Metrics included active electrode yield (AEY), signal-to-noise ratio, and signal amplitude.
- Tissue Analysis: Post-mortem tissue was analyzed using a custom 146-gene panel to quantify expression related to neuroinflammation, oxidative stress, and neuronal health.
Key Findings: MARE probes demonstrated superior recording performance, more stable impedance, and a healing tissue response characterized by higher neuronal density and reduced expression of neuroinflammatory markers compared to rigid controls [77].
Protocol: Assessment of µECoG for Speech Decoding
Objective: To determine the benefit of high-density µECoG for recording and decoding speech-related neural signals [72].
Methodology:
- Electrode Arrays: High-density µECoG arrays (128- or 256-channel) with small inter-electrode distance (1.33-1.72 mm) were placed over the speech motor cortex (SMC) in human patients intra-operatively.
- Task Design: Patients performed a speech repetition task, listening to and repeating non-words.
- Signal Analysis: The high gamma band (70–150 Hz) power was extracted as a correlate of local neural activity. Evoked-signal-to-noise ratio (ESNR) was calculated and compared to simulated standard intracranial electrodes.
- Decoding Model: A non-linear decoding model was used to predict spoken phonemes from the neural activations.
Key Findings: µECoG provided neural signals with a 48% higher SNR and 57x higher spatial resolution than standard macro-ECoG. This enhanced signal quality improved speech decoding accuracy by 35% [72].
Protocol: Comparison of µECoG and Intracortical MEA
Objective: To directly compare signal characteristics of subdural µECoG and penetrating intracortical microelectrode arrays (MEAs) [20].
Methodology:
- Animal Model and Implantation: Twenty pigs were implanted with either a subdural µECoG array (32 electrodes) or a penetrating MEA (16 electrodes) in the primary somatosensory cortex (S1).
- Stimulation: Somatosensory evoked potentials (SEPs) were generated by electrically stimulating the ulnar nerve.
- Signal Analysis: The recorded SEPs were analyzed for peak-to-peak amplitude, signal-to-noise ratio (SNR), and power spectral density (PSD).
Key Findings: While SEPs from intracortical MEAs had higher amplitudes and contained significantly more spectral power at higher frequencies, the SNR was similar between the two interfaces, demonstrating µECoG's viability for capturing robust neural signals with less invasiveness [20].
Signaling Pathways and the Neuroinflammatory Response
The following diagram illustrates the primary signaling pathways involved in the neuroinflammatory response to intracortical microelectrodes and the points of intervention for the material solutions discussed.
Diagram Title: Neuroinflammatory Cascade and Intervention Points
The Scientist's Toolkit: Essential Research Reagents and Materials
This table details key materials and reagents used in the development of next-generation neural interfaces.
Table 3: Key Research Reagents and Materials for Neural Interface Development
| Item Name | Function/Description | Application in Research |
|---|---|---|
| Polyvinyl Acetate/Cellulose Nanocrystal Nanocomposite (NC) | A mechanically-adaptive polymer that is rigid for implantation but softens upon contact with physiological fluids to match brain's modulus [77]. | Substrate for MARE probes to reduce mechanical mismatch and enable drug elution [77]. |
| Resveratrol | A natural polyphenol antioxidant that mitigates oxidative stress by neutralizing reactive oxygen/nitrogen species [77]. | Loaded into NC probes (MARE) for local elution to reduce peri-implant inflammation and neuronal death [77]. |
| PEDOT:PSS | A conductive polymer (Poly(3,4-ethylenedioxythiophene):polystyrenesulfonate) with high conductivity, flexibility, and biocompatibility [78]. | Used as a coating or base material for flexible bioelectrodes to improve SNR, lower impedance, and enhance charge injection [78]. |
| Titania Nanotube Array (TNA) | A nanostructured titanium dioxide coating with antimicrobial properties [79]. | Coated onto silicon intracortical probes to reduce microbial invasion and the associated neuroinflammatory response at chronic time points [79]. |
| Dexamethasone Sodium Phosphate (DEXSP) | A potent synthetic anti-inflammatory corticosteroid. | The active pharmaceutical ingredient in DEXSPPIN nanoparticles, used to suppress the neuroinflammatory cascade locally [80]. |
| Platelet-Inspired Nanoparticles (PIN/DEXSPPIN) | Lipid-based nanoparticles functionalized with peptides (CBP, VBP, FMP) to target sites of vascular injury [80]. | Used as a systemic, targeted drug delivery vehicle to accumulate at the implant site, promoting hemostasis and releasing anti-inflammatory drugs [80]. |
| Liquid Crystal Polymer Thin-Film (LCP-TF) | A flexible, biocompatible substrate for high-density electrode arrays [72]. | Fabrication of µECoG arrays with high electrode density and conformal contact with the cortical surface [72]. |
In electrophysiological research, the quest for neural signals of unparalleled clarity is paramount. Electroencephalography (EEG), electrocorticography (ECoG), and intracortical recordings provide windows into brain activity, but the view is often obscured by noise and artifacts. Within this context, pre-processing techniques are the essential first step, tasked with improving the signal-to-noise ratio (SNR) to reveal the true neural correlates beneath. Common Average Referencing (CAR) and Independent Component Analysis (ICA) stand as two pillars in this pre-processing landscape. CAR operates as a spatial filter, subtracting the common signal across all electrodes to isolate local activity [81] [82]. In contrast, ICA is a blind source separation method that decomposes the recorded signals into statistically independent components, allowing for the identification and removal of artifactual sources [83] [84]. Framed within the broader thesis of SNR comparison across EEG, ECoG, and intracortical recordings, this guide provides an objective comparison of these two fundamental techniques. We will explore their underlying principles, summarize experimental data on their performance, and detail the protocols for their implementation, providing researchers with the information needed to make an informed choice for their specific recording modality and research goals.
Theoretical Foundations and Comparative Mechanisms
Common Average Referencing (CAR)
CAR functions on a relatively simple spatial filtering principle. It assumes that noise recorded by multiple electrodes, such as distant physiological activity or environmental interference, is "common" to all channels. The method calculates the average signal across all (or a selected subset of) electrodes at each time point and subtracts this average from each individual channel [81] [82]. The primary goal is to attenuate this shared noise, thereby enhancing the visibility of local, neurally generated signals. The operation can be represented as:
( V{i,CAR}(t) = V{i,raw}(t) - \frac{1}{N} \sum{j=1}^{N} V{j,raw}(t) )
where ( V{i,CAR}(t) ) is the re-referenced signal for channel *i* at time *t*, ( V{i,raw}(t) ) is the original signal, and N is the total number of channels used in the average.
A significant challenge with standard CAR is its susceptibility to bias from "responsive channels." If a channel with a very high-amplitude signal is included in the average, it can smear that signal into all other channels, distorting the true neural activity [82]. Advanced, data-driven variants like the Common Average Referencing by Least Anticorrelation (CARLA) have been developed to address this. CARLA iteratively selects a subset of channels with low cross-trial covariance to include in the common average, aiming to minimize the inclusion of highly responsive channels and thus reduce bias [82].
Independent Component Analysis (ICA)
ICA is a computational method for solving the blind source separation problem. It operates on the principle that the multichannel recorded data is a linear mixture of underlying sources that are statistically independent from one another. The goal of ICA is to find a "unmixing" matrix that separates these sources [83] [84]. The model is:
( \mathbf{X} = \mathbf{A}\mathbf{S} )
where ( \mathbf{X} ) is the matrix of recorded data (channels × time), ( \mathbf{S} ) is the matrix of independent components (sources), and ( \mathbf{A} is the "mixing" matrix that describes how each source projects to the sensors. ICA algorithms (e.g., Infomax, FastICA) estimate the unmixing matrix to yield ( \mathbf{S} ). Once computed, components can be inspected, and those identified as artifacts (e.g., eye blinks, muscle activity, line noise) can be removed by subtracting their contribution back from the original signal [83] [84]. A key advantage of ICA is its ability to isolate and remove non-neural artifacts that are not common across all electrodes, a task for which CAR is less suited.
Table 1: Core Conceptual Comparison between CAR and ICA.
| Feature | Common Average Referencing (CAR) | Independent Component Analysis (ICA) |
|---|---|---|
| Primary Function | Spatial filter for noise reduction | Blind source separation for artifact removal |
| Underlying Principle | Subtracts global average signal from each channel | Decomposes signals into statistically independent components |
| Assumption | Noise is common across all electrodes; neural signals are local | Recorded signals are a linear mixture of statistically independent sources |
| Primary Output | Re-referenced signal for each channel | Set of independent components (time courses & topographies) |
| Interpretability | Direct interpretation of processed channel data | Requires analysis of components (maps, spectra, time courses) |
Performance and Experimental Data Comparison
The effectiveness of CAR and ICA varies significantly depending on the recording modality, the nature of the artifacts, and the specific signal of interest. Quantitative comparisons reveal their distinct performance profiles.
Signal-to-Noise Ratio (SNR) and Signal Quality
In the context of intracortical recordings like cortico-cortical evoked potentials (CCEPs), the performance of CAR and its advanced variants can be quantified. In one study, the adjusted CARLA method was evaluated using the mean R² between all pairs of channels, where a lower value indicates less shared noise and thus better-referenced data. CARLA produced a significantly lower mean R² than standard CAR, demonstrating its superior ability to improve signal quality by reducing inter-channel dependency without introducing bias from responsive channels [82].
For EEG, the combination of ICA with spatial filtering techniques has shown remarkable efficacy. A study on dry EEG, which is particularly prone to movement artifacts, combined ICA-based cleaning (Fingerprint + ARCI) with a spatial harmonic analysis (SPHARA) method. This combined approach reduced the standard deviation of the signal from 9.76 µV to 6.15 µV and increased the SNR from 2.31 dB to 5.56 dB, outperforming either technique used in isolation [85]. This underscores the power of hybrid methods for challenging recording scenarios.
Spatial Resolution and Artifact Removal
ICA excels at isolating and removing specific physiological artifacts that have distinct spatial and temporal signatures. Research has shown it can effectively identify components corresponding to blinks, eye movements, muscle activity, heartbeats, and bad channels [83]. The success of this, however, relies heavily on expert visual inspection or semi-automated tools (like SASICA) to correctly label components, a process that can be subjective and time-consuming [83].
CAR, as a spatial filter, impacts spatial resolution by highlighting local activity. Its performance is optimal when the assumption of localized neural signals and widespread noise holds true. However, its performance can degrade if the neural signals of interest are themselves widely distributed across the electrode array, as they may be partially subtracted along with the noise [81].
Table 2: Summary of Quantitative Performance from Select Studies.
| Method | Recording Type | Key Performance Metric | Result | Citation |
|---|---|---|---|---|
| CARLA (Adjusted CAR) | CCEP (iEEG) | Reduction in mean R² (inter-channel dependency) | Significantly lower than standard CAR | [82] |
| ICA + SPHARA | Dry EEG | Standard Deviation (SD) | Improved from 9.76 µV to 6.15 µV | [85] |
| ICA + SPHARA | Dry EEG | Signal-to-Noise Ratio (SNR) | Improved from 2.31 dB to 5.56 dB | [85] |
| tEEG (for comparison) | Conventional vs. tEEG | Grasp decoding accuracy (Multiclass) | ~75.97% (tEEG) vs. ~61.27% (EEG) | [86] |
Experimental Protocols and Methodologies
Protocol for Common Average Referencing
A standard protocol for applying CAR, including the advanced CARLA method, is as follows [82]:
- Data Input: Begin with preprocessed, continuous, or epoched iEEG/EEG data.
- Channel Selection (for CARLA):
- Calculate the mean cross-trial covariance for each channel.
- Order the channels from lowest to highest mean covariance.
- Iteratively add channels from the ordered list to the common average. After each addition, calculate the mean anticorrelation between each single channel and all the currently re-referenced channels.
- The optimal subset of channels for the common average is the one that achieves the minimum mean anticorrelation. This subset is presumed to contain the fewest responsive channels and the most noise-correlated channels.
- Common Average Calculation: Compute the average signal across the selected subset of channels at every time point.
- Subtraction: Subtract this common average signal from the signal of every individual channel.
- Output: The result is the re-referenced dataset.
Protocol for Independent Component Analysis
A generalized protocol for using ICA for artifact removal, as implemented in tools like EEGLAB, is detailed below [83] [84]:
- Preprocessing: This is a critical step. Data should be filtered (e.g., high-pass at 1 Hz) and have bad channels and noisy data segments removed. The data should then be re-referenced (often to an average reference is suitable for ICA).
- Data Decomposition: Perform ICA on the preprocessed data. Common algorithms include Infomax ICA, FastICA, or SOBI. The input is the multichannel data matrix, and the output is an unmixing matrix and the independent components.
- Component Inspection: Visually inspect the resulting components using their:
- Topography: The spatial map of the component's projection onto the scalp.
- Time Course: The activation of the component over time.
- Power Spectrum: The frequency content of the component.
- This step is crucial for identifying artifacts. For example, eye blink components typically have a frontal topography and a low-frequency spectrum.
- Component Labeling & Rejection: Label components as 'neural' or 'artifactual' based on their features. Semi-automated tools like SASICA can assist by computing statistical features (e.g., focal topography, abnormal distribution) to flag potential artifacts [83].
- Data Reconstruction: Remove the artifactual components by projecting the data back to the sensor space using only the 'neural' components.
Table 3: Key Software Tools and Resources for CAR and ICA Implementation.
| Tool/Resource Name | Primary Function | Relevance to CAR/ICA | Citation / Source |
|---|---|---|---|
| EEGLAB | MATLAB toolbox for EEG processing | Provides a comprehensive environment for running ICA and visualizing components. Includes functions for CAR. | [84] |
| SASICA Plugin | Semi-automated selection of ICA components | Assists in the objective identification of artifactual components by computing multiple feature metrics. | [83] |
| FieldTrip Toolbox | MATLAB toolbox for MEG/EEG/iEEG analysis | Supports advanced analysis of iEEG/ECoG data, including re-referencing methods and source reconstruction. | [87] |
| CARLA Algorithm | Adjusted common average referencing | A specific, data-driven algorithm for optimizing channel selection in CAR for CCEP/iEEG data. | [82] |
The choice between Common Average Referencing and Independent Component Analysis is not a matter of declaring a universal winner but of selecting the right tool for the scientific question and recording modality. CAR is a computationally efficient, robust spatial filter highly effective for suppressing global noise in multi-electrode arrays, particularly in iEEG and ECoG studies. Its limitations lie in its potential to bias data if highly responsive channels are included and its relative inability to remove focal, non-neural artifacts. ICA is a powerful, versatile method for isolating and removing a wide array of artifacts, from eye blinks to muscle noise, making it indispensable for EEG and studies where such contaminants are prevalent. Its primary drawbacks are its computational demands and the expert supervision required for accurate component selection. The emerging trend of combining these methods—using ICA for artifact removal followed by CAR for further noise reduction—demonstrates a powerful hybrid approach [85]. Furthermore, the development of data-driven variants like CARLA [82] highlights a continued evolution in pre-processing techniques. For researchers focused on SNR optimization across different recording scales, a thoughtful, often sequential, application of these methods will yield the clearest and most interpretable window into brain dynamics.
Algorithmic Noise Reduction and Spatial Filtering Techniques for Cleaner Neural Data
In systems neuroscience, a fundamental challenge is disentangling true neural activity from noise to accurately interpret brain function. This pursuit is critical for both basic research and clinical applications, such as brain-computer interfaces (BCIs) and surgical planning. The signal-to-noise ratio (SNR) varies significantly across recording modalities, each with inherent trade-offs between spatial resolution, temporal resolution, and invasiveness. Electroencephalography (EEG) provides non-invasive recording but with limited spatial resolution and greater vulnerability to artifacts like blinks and eye movements [15]. Electrocorticography (ECoG), recorded directly from the cortical surface, offers higher spatial resolution and SNR, while intracortical microelectrodes capture neural activity at the finest spatial scale [72]. The development of advanced algorithmic techniques for noise reduction and spatial filtering is paramount for maximizing the information extracted from each modality. These methods range from traditional signal processing approaches to modern deep learning models, all aiming to suppress noise while preserving the integrity of neural signals. This guide provides a comparative analysis of these techniques, their experimental protocols, and their performance in enhancing neural data quality across different recording platforms.
Comparative SNR and Performance Across Neural Recording Modalities
The choice of recording modality fundamentally determines the baseline signal quality and the specific noise challenges that algorithms must address. The following table summarizes key characteristics and performance metrics.
Table 1: Signal Quality and Performance Comparison of Neural Recording Modalities
| Recording Modality | Spatial Resolution | Typical SNR Characteristics | Primary Noise Sources | Decoding Performance Examples |
|---|---|---|---|---|
| EEG (Non-invasive) | ~1-2 cm (scalp) | Lower; more susceptible to physiological artifacts [15] | Blinks, eye movements, muscle activity, line noise [15] | Varies with paradigm; e.g., SSVEP recognition up to 221.4 bits/min ITR with advanced filtering [88] |
| ECoG (Intracranial) | ~1 cm (standard macro) | Higher than EEG; artifacts from eye blinks still present in prefrontal areas [15] | Minor blink/saccade artifacts, environmental interference [15] | Foundation for speech decoding; performance improves with electrode density [72] |
| µECoG (Intracranial) | ~1.3-1.7 mm inter-electrode [72] | 48% higher SNR compared to macro-ECoG/SEEG [72] | Thermal noise, system noise | 35% improvement in speech decoding accuracy over standard intracranial signals [72] |
| Intracortical (Microelectrodes) | Micrometer scale | High for single units; susceptible to local field fluctuations | Micro-movements, biological encapsulation, thermal noise | N/A in provided results |
The quantitative advantages of high-resolution recordings are clear. Micro-electrocorticographic (µECoG) arrays demonstrate a 48% higher evoked-signal-to-noise ratio (ESNR) compared to standard macro-ECoG and stereo-EEG (SEEG) [72]. This enhanced signal quality directly translates to improved functional performance, yielding a 35% increase in speech decoding accuracy for phoneme prediction tasks [72]. The underlying reason is that neural information is highly localized; studies show low inter-electrode correlation (r=0.1-0.3) at 4 mm spacing, meaning critical articulatory features for speech are distinct at the millimeter scale [72].
Similarly, in non-invasive settings, advanced spatial filtering of EEG signals can dramatically improve performance for tasks like Steady-State Visual Evoked Potential (SSVEP) recognition. One study integrating graph-based spatial filters with lightweight convolutional neural networks achieved a maximum average information transfer rate (ITR) of 221.4 bits/min on a benchmark dataset, underscoring the value of optimized processing [88].
Experimental Protocols for Key Noise Reduction and Spatial Filtering Techniques
Deep Neural Network (DNN) for Adaptive Spatial Smoothing of fMRI Data
Objective: To overcome the spatial blurring artifacts introduced by conventional Gaussian smoothing in task-based fMRI, which compromises spatial specificity at the subject level [89].
Methodology Summary:
- Data Acquisition: Collect high-resolution (potentially sub-millimeter) task-based fMRI data where participants engage in a paradigm designed to evoke specific neural responses.
- Network Architecture and Training: Design a deep neural network, typically based on convolutional layers, to estimate optimal spatial smoothing parameters for each voxel. The network takes unsmoothed fMRI time series data as input. It is trained to maximize the correlation between the smoothed neural activity and the task design matrix, effectively learning tissue-dependent smoothing patterns that respect cortical anatomy [89].
- Comparison: The output of the DNN is compared against results from standard Gaussian smoothing and traditional adaptive methods like constrained Canonical Correlation Analysis (CCA). The comparison metrics typically include spatial specificity of activation maps, sensitivity to true signals, and quantitative model fit [89].
The Align-and-Filter Network (AFnet) for Multichannel Speech Enhancement
Objective: To improve the robustness of multichannel speech enhancement systems against target speaker location uncertainty by explicitly modeling spatial alignment [90].
Methodology Summary:
- Signal Model: Multichannel noisy signals
X_iare modeled in the time-frequency domain (STFT) asX_i = S_i + V_i, whereS_iis the speech component andV_iis noise at the i-th microphone [90]. - Network Architecture: The AFnet is a two-stage deep learning model.
- Align Net: The first module estimates a set of complex-valued "alignment masks"
A_i. These masks are supervised during training to approximate the Relative Transfer Functions (RTFs) of the target speech, which encode the spatial location of the speaker. Multiplying the input signals by these masks spatially aligns the speech components across all microphones [90]. - Filter Net: The second module estimates "filtering masks"
F_ithat are applied to the aligned signals to perform final noise suppression and recover the clean speech at a reference microphone [90].
- Align Net: The first module estimates a set of complex-valued "alignment masks"
- Training Scheme: A two-step training process is used: first, the Align Net is pre-trained using a loss function that minimizes the error between its output masks and the ground-truth RTFs. Subsequently, the entire AFnet is fine-tuned end-to-end using a reconstruction loss (e.g., a power-law compressed mean-squared error) to maximize the quality of the output speech [90].
Mutual Information for Spatiotemporal Mapping of ECoG in Speech
Objective: To capture the nonlinear neural dynamics of continuous speech production that are missed by traditional linear methods like cross-correlation [91].
Methodology Summary:
- Data Recording: ECoG signals are recorded from participants (e.g., epilepsy patients) as they engage in continuous speech production tasks.
- Signal Processing: A key innovation is the "masked analysis," where periods of silence in the speech recording are excluded from the computation. This prevents the analysis from being dominated by the simple contrast between silence and non-silence [91].
- Analysis: Mutual Information (MI), an information-theoretic measure capable of detecting both linear and nonlinear dependencies, is computed between the ECoG time series (e.g., high-gamma power) and the recorded speech envelope. This is compared against results from standard cross-correlation analysis [91].
- Validation: The method is validated by its ability to reveal earlier and more anatomically precise activations in speech-related brain areas (e.g., prefrontal and premotor cortex ~440 ms before speech onset) compared to linear methods [91].
Signaling Pathways and Experimental Workflows
The following diagram illustrates the core logical workflow for developing and validating a neural data processing algorithm, integrating steps common to the methodologies discussed above.
Neural Data Processing Workflow
The diagram below outlines the specific "align-then-filter" principle used in the AFnet architecture for multichannel speech enhancement, a strategy that improves robustness against spatial uncertainty [90].
Align-and-Filter Network Principle
The Scientist's Toolkit: Key Research Reagents and Materials
Table 2: Essential Materials and Tools for Advanced Neural Signal Processing Research
| Item Name | Function/Description | Example Use Case |
|---|---|---|
| LCP-TF µECoG Arrays | Thin-film micro-electrocorticography arrays with high electrode density (e.g., 1.33-1.72 mm spacing) for high-resolution neural recording [72]. | Enables recording of micro-scale neural features from the cortical surface, providing the high-SNR data required for improved speech decoding [72]. |
| Relative Transfer Function (RTF) | A mathematical representation of the cross-channel differences of a target source, used to encode its spatial location [90]. | Serves as a supervised learning target for training the spatial alignment module (Align Net) in multichannel speech enhancement networks [90]. |
| Mutual Information (MI) Metric | An information-theoretic measure that quantifies both linear and nonlinear statistical dependencies between variables [91]. | Used as a superior alternative to cross-correlation for mapping nonlinear neural dynamics in ECoG signals during continuous speech [91]. |
| Constrained CCA | A multivariate statistical method (Canonical Correlation Analysis) with constraints, used for adaptive spatial smoothing of fMRI data [89]. | Serves as a baseline or predecessor to more complex DNN models for enhancing detection of brain activation in task-based fMRI [89]. |
| Graph-Based Spatial Filter | A filter that leverages the physical topological relationships between EEG sensors to enhance feature quality [88]. | Used as a preprocessing step in EEG-based BCIs to suppress redundant information and improve SSVEP recognition performance [88]. |
Empirical Comparisons and Validation of SNR Across Recording Modalities
In the field of advanced neural interfacing, the trade-off between signal quality and invasiveness represents a central challenge for researchers and clinicians. Electrocorticography (ECoG) and intracortical microelectrode arrays (MEAs) occupy distinct positions on this spectrum, offering different balances of signal richness versus tissue disruption [92] [93]. This comparison guide objectively evaluates the performance of micro-electrocorticography (µECoG) against intracortical arrays, with specific focus on signal-to-noise ratio (SNR) and signal amplitude within the somatosensory cortex.
The somatosensory cortex serves as an ideal model system for this comparison due to its well-defined layered structure and the ability to generate reproducible evoked responses through controlled peripheral stimulation [1]. Understanding the precise performance characteristics of these neural interfaces is crucial for selecting appropriate technologies for basic neuroscience research, pharmaceutical development, and clinical brain-computer interface applications.
Neural Signal Fundamentals and Recording Technologies
Signal Characteristics Across Recording Modalities
Neural interfaces capture different aspects of brain electrical activity depending on their physical placement relative to neural tissue. Local field potentials (LFPs) represent the summed synaptic activity of neuronal populations, while action potentials (spikes) are brief, millisecond-scale events originating from individual neurons [1]. The recording technology fundamentally determines which of these signals can be captured and with what fidelity.
Micro-ECoG arrays are surface electrodes placed either epidurally (on top of the dura) or subdurally (beneath the dura but on the cortical surface). These devices typically feature electrode diameters <250 µm and inter-electrode spacing <1.5 mm, representing a significant miniaturization over conventional clinical ECoG [1]. Their primary advantage lies in providing cortical coverage without penetrating the brain parenchyma, thereby reducing tissue damage and inflammatory response [92] [33].
Intracortical microelectrode arrays are penetrating devices inserted directly into the cortical tissue, typically reaching depths of 1.5-2.0 mm [93] [1]. This positioning allows them to record both LFPs and action potentials from individual neurons, providing unparalleled spatial resolution and access to the full spectral content of neural activity [17] [1].
Table 1: Fundamental Characteristics of Neural Recording Technologies
| Parameter | Micro-ECoG | Intracortical Arrays |
|---|---|---|
| Spatial Resolution | 0.1-1.5 mm [92] [1] | 50-100 µm [17] |
| Recording Location | Cortical surface (epidural/subdural) [92] [1] | Within cortical layers [93] [1] |
| Signal Types | Local field potentials [93] [1] | Single-unit activity & local field potentials [17] [1] |
| Frequency Bandwidth | 0-500 Hz [17] | 0-7000 Hz [17] |
| Amplitude Range | Microvolts (µV) [17] | Millivolts (mV) for action potentials [17] |
Signal Acquisition Pathways
The following diagram illustrates the fundamental differences in how micro-ECoG and intracortical arrays capture neural signals from the somatosensory cortex:
Experimental Comparison Methodology
Controlled Somatosensory Evoked Potential Protocol
A recent direct comparison study implemented a controlled experimental paradigm to quantitatively assess both µECoG and intracortical array performance in the somatosensory cortex [1]. This methodology provides the foundation for the SNR and amplitude comparisons presented in this guide.
Animal Model and Surgical Preparation: The study utilized twenty healthy female Danish Landrace pigs (38.4 ± 10.7 kg). Following tranquilization and intubation, animals were maintained under anesthesia using sevoflurane, propofol, and fentanyl. A rectangular craniotomy (3 × 5 cm) centered around the bregma landmark exposed the primary somatosensory cortex (S1). The dura mater was carefully removed while preserving cortical vasculature [1].
Electrode Specifications and Implantation:
- Intracortical MEA: A 16-electrode (4 × 4) squared grid array with Pt/Ir electrodes, 75 μm tip diameter, 1.0 mm interelectrode spacing, and 0.6 MΩ impedance. The array was penetrated 1.6–1.8 mm perpendicularly into the cortex using a micromanipulator [1].
- µECoG Array: A 32-electrode array (8 × 4 configuration) with 200 μm diameter electrodes, 1 mm interelectrode distance, and impedance values of 10–40 kΩ. The array was implanted subdurally on the cortical surface [1].
Evoked Potential Generation: Somatosensory evoked potentials (SEPs) were generated by applying electrical stimulation to the ulnar nerve using cuff electrodes. The experimental protocol consisted of three stimulation trains delivering 50 pulses each (1 mA amplitude, 500 μs pulse duration) with a 2 s delay between pulses incorporating a pseudo-random Gaussian delay of ±250 ms to prevent habituation [1].
Signal Acquisition and Processing: Neural signals were acquired at 24 kHz using a Tucker-Davis Technologies electrophysiology system. Signals were bandpass filtered (0.1–5000 Hz) using a fourth-order Butterworth design. SEPs were windowed from -1.0 s to 1.5 s relative to stimulation onset, and analysis commenced one hour post-implantation to allow tissue responses and impedances to stabilize [1].
Experimental Workflow
The following diagram outlines the comprehensive experimental workflow used in the comparative study:
Quantitative Performance Comparison
Signal Amplitude and SNR Analysis
The controlled comparative study revealed significant differences in signal acquisition capabilities between the two technologies [1]. Quantitative measurements of somatosensory evoked potentials provide direct evidence of these performance characteristics.
Table 2: Direct Performance Comparison in Somatosensory Cortex
| Performance Metric | Micro-ECoG | Intracortical Arrays | Statistical Significance |
|---|---|---|---|
| SEP Amplitude | Lower amplitude responses [1] | Significantly higher amplitudes [1] | p < 0.05 |
| Signal-to-Noise Ratio | Similar SNR values [1] | Similar SNR values [1] | Not Significant |
| Spectral Power | Reduced power across spectrum [1] | Significantly more spectral power, especially at higher frequencies [1] | p < 0.05 |
| High-Frequency Content | Limited high-frequency information [1] | Preserved high-frequency components [1] | Qualitative Difference |
| Spatial Specificity | Millimeter-scale resolution [92] | Sub-millimeter resolution [17] | Fundamental Difference |
Technical Performance Data
Impedance characteristics and signal quality metrics provide additional context for interpreting the amplitude and SNR findings:
Table 3: Electrode Characteristics and Signal Specifications
| Parameter | Micro-ECoG | Intracortical Arrays |
|---|---|---|
| Electrode Diameter | 200 μm [1] | 75 μm [1] |
| Array Configuration | 8 × 4 (32 channels) [1] | 4 × 4 (16 channels) [1] |
| Inter-electrode Spacing | 1.0 mm [1] | 1.0 mm [1] |
| Impedance Range | 10-40 kΩ [1] | ~0.6 MΩ [1] |
| Primary Signal Contribution | Superficial cortical layers [1] | All cortical layers at implantation depth [1] |
| High-Frequency Cutoff | ~200-500 Hz [94] [17] | Up to 7000 Hz [17] |
The Scientist's Toolkit: Essential Research Materials
Table 4: Key Research Reagents and Experimental Solutions
| Item | Specification | Experimental Function |
|---|---|---|
| Intracortical MEA | 16-ch, 75 μm tips, 1.0 mm spacing (Microprobes) [1] | Records intracortical signals at 1.6-1.8 mm depth |
| µECoG Array | 32-ch, 200 μm contacts, 1 mm spacing (Neuronexus) [1] | Records cortical surface potentials |
| Nerve Cuff Electrodes | Custom design for ulnar nerve [1] | Delivers controlled peripheral stimulation |
| Physiological Saline | Isotonic solution [1] | Maintains cortical moisture during recording |
| Signal Acquisition System | TDT RZ2 with ZIF-Clip headstage [1] | High-fidelity neural signal acquisition |
| Stimulation System | STG4008 Programmable Stimulator [1] | Generates precise peripheral nerve stimuli |
Discussion and Research Implications
Interpretation of Comparative Performance Data
The finding that intracortical arrays yield significantly higher signal amplitudes while maintaining similar SNR values to µECoG arrays has important implications for neural interface selection [1]. The amplitude advantage stems from the proximity of intracortical electrodes to active neural sources, particularly in middle cortical layers where synchronous synaptic activity generates strong local field potentials during evoked responses [1].
The preservation of SNR despite higher amplitudes suggests that intracortical recordings may maintain favorable noise characteristics despite their smaller electrode sites and higher impedances. This may be attributed to the tighter coupling between electrode and neural tissue, reducing the impact of environmental noise sources [1]. Additionally, the significantly enhanced spectral power in intracortical recordings, particularly at higher frequencies, indicates better preservation of fast neural dynamics critical for information encoding in the somatosensory system [1].
Technology Selection Guidelines
The choice between µECoG and intracortical arrays depends heavily on research objectives and practical constraints:
Micro-ECoG is preferable for large-scale cortical mapping, chronic implantation studies where signal stability is prioritized, and when minimizing tissue damage is a primary concern [92] [33]. The technology provides adequate performance for decoding broader brain states and basic somatosensory responses without penetrating the cortex [17].
Intracortical arrays are indicated when high spatial resolution is required, for studying laminar-specific processing, when single-neuron activity is essential, and for capturing high-frequency neural dynamics [17] [1]. The technology is particularly valuable for detailed circuit-level analysis of somatosensory processing and when maximum information content per channel is needed [93].
Recent advancements in µECoG technology, including higher density arrays (up to 1024 channels) and minimally invasive implantation techniques, continue to narrow the performance gap while maintaining the advantages of surface recording [33]. Similarly, developments in flexible intracortical electrodes aim to reduce the foreign body response and improve chronic recording stability [33] [95].
Multivariate Pattern Analysis: Relating EEG, fMRI, and ECoG Signals to Underlying Neural Codes
Understanding the complex neural dynamics that underpin human cognitive function remains a central challenge in neuroscience. A significant hurdle in this pursuit is that no single neuroimaging technique can simultaneously capture brain activity with both high spatial and high temporal resolution. Instead, the field relies on complementary methods: functional magnetic resonance imaging (fMRI) provides high spatial resolution but poor temporal resolution, while electroencephalography (EEG) offers millisecond-scale temporal precision at the cost of limited spatial specificity. To interpret the signals from these non-invasive methods, they are often compared with electrocorticography (ECoG), an invasive technique that records electrical activity directly from the cortical surface, offering a more direct measurement of neural population activity with high spatiotemporal resolution [9] [96].
Historically, the relationships between fMRI, EEG, and ECoG signals were established using univariate analysis approaches, which treat each measurement point (e.g., a voxel or electrode) independently. However, the brain represents information through distributed population codes that are inherently multivariate. Multivariate Pattern Analysis (MVPA) represents a paradigm shift, as it is sensitive to these distributed patterns of activity and can decode stimulus information with higher sensitivity than univariate methods [9] [97]. This guide objectively compares the performance of EEG, fMRI, and ECoG through the lens of MVPA, synthesizing recent experimental evidence to elucidate how these signals relate to the underlying neural codes, with a particular focus on their signal quality and information content.
Fundamental Characteristics of EEG, fMRI, and ECoG
The following table summarizes the core technical characteristics and performance metrics of EEG, fMRI, and ECoG, highlighting their inherent trade-offs.
Table 1: Fundamental characteristics and performance metrics of EEG, fMRI, and ECoG.
| Feature | EEG (Non-invasive) | fMRI (Non-invasive) | ECoG (Invasive) |
|---|---|---|---|
| Primary Signal Measured | Scalp electrical potentials | Blood Oxygenation Level Dependent (BOLD) response | Cortical surface electrical potentials |
| Spatial Resolution | Limited (centimeters) [9] | High (millimeters) [9] | High (millimeters) [96] |
| Temporal Resolution | High (milliseconds) [9] | Low (seconds) [9] | High (milliseconds) [9] |
| Typical Signal-to-Noise Ratio (SNR) | Lower; susceptible to biological artifacts [15] | Variable; benefits from grand-averaging [96] | Higher; less susceptible to scalp artifacts [15] [72] |
| Invasiveness | Non-invasive | Non-invasive | Invasive (requires surgery) |
| Key Strength in MVPA | Millisecond-resolution tracking of neural dynamics | Fine-grained spatial localization of information | High spatiotemporal resolution; ground truth for neural population codes |
A Representative Experimental Protocol for Multimodal Comparison
To objectively compare these modalities, experiments must record data using a consistent stimulus set. The following workflow and methodological details are drawn from a seminal 2022 study that provides a direct, content-sensitive comparison [9] [98] [97].
Experimental Workflow for Multimodal Comparison
The diagram below outlines the core experimental workflow for acquiring and comparing EEG, fMRI, and ECoG data using identical visual stimuli.
Detailed Methodological Components
- Stimulus Set: The study used 125 grayscale images of everyday objects from five categories (animals, chairs, faces, fruits, vehicles). Each object was presented under five different viewing conditions, varying in size (1.5°, 3°, or 6° visual angle) and depth rotation (0°, 45°, or 90°). This design allowed researchers to test not only category decoding but also the tolerance of neural representations to identity-preserving transformations [9] [98].
- EEG Protocol:
- Design: Participants completed about 32 runs, with each of the 125 images presented once per run in random order. Each trial consisted of a 200 ms image presentation followed by an 800 ms blank screen.
- Task: A one-back task on object identity ensured participant attention.
- Acquisition: Data were recorded from 64 electrodes at a 1100 Hz sampling rate.
- Preprocessing: Data were low-pass filtered at 40 Hz, resampled to 1000 Hz, and epoched from -100 ms to +600 ms around stimulus onset [9] [98].
- fMRI Protocol: fMRI data were recorded separately from the same participants using an event-related design while they viewed the same stimulus set. The specific acquisition parameters (e.g., TR, TE) were aligned to maximize the detection of fine-grained spatial patterns in the BOLD signal [9] [97].
- ECoG Protocol: The analysis leveraged a previously published ECoG dataset recorded from epileptic patients using the same stimulus set [9]. This allowed for a direct comparison of non-invasive signals to a more direct measure of cortical population activity.
- Multivariate Analysis: The core analysis involved training pattern classifiers to decode object category information from the activation patterns in each modality (EEG, fMRI, ECoG) at successive time points. The temporal dynamics of decoding accuracy and the correlation between representational patterns across modalities were the key metrics of comparison [9] [97].
Comparative Performance in Visual Object Representation
The application of MVPA to data from the above protocol reveals distinct and complementary profiles for each modality.
Table 2: MVPA performance comparison in decoding visual object information.
| Aspect | EEG | fMRI | ECoG |
|---|---|---|---|
| Temporal Dynamics of Decoding | Object category signals emerge rapidly and can be tracked with millisecond precision [9] [97]. | Provides a single, sluggish measure integrated over seconds, obscuring fast dynamics. | Matches EEG in tracking the swift emergence of category signals at a similar temporal scale [9] [97]. |
| Spatial Specificity | Limited; source localization is challenging. | High; can localize information to specific visual regions (e.g., occipital vs. temporal cortex) [9] [97]. | High; can resolve information at the millimeter scale, revealing distinct articulatory features for speech [72]. |
| Representational Content | Correlates with ECoG, but correlation drops for size- and orientation-invariant representations [9]. | Shows a tighter relationship to ECoG in occipital than temporal regions, linked to fMRI SNR differences [9]. | Serves as a benchmark; directly captures complex, high-dimensional population codes for objects [9]. |
| Signal Quality & SNR | Lower SNR; more susceptible to artifacts like blinks and saccades, though these can be mitigated [15]. | SNR can be low in certain regions (e.g., temporal cortex); benefits significantly from grand-averaging across subjects [9] [96]. | Inherently higher signal quality and SNR; less contaminated by non-neural biological artifacts [15] [72]. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful execution of multivariate comparison studies requires a suite of specialized tools and software.
Table 3: Key research reagents and solutions for multivariate neuroimaging research.
| Tool / Solution | Primary Function | Example Use Case |
|---|---|---|
| High-Density EEG Systems (e.g., 64+ channel setups) | Records scalp electrical potentials with high temporal resolution. | Core apparatus for acquiring non-invasive neural dynamics data [9] [98]. |
| fMRI Scanner (3T and above) | Measures the hemodynamic (BOLD) response associated with neural activity. | Core apparatus for acquiring high-spatial-resolution brain maps [9] [96]. |
| ECoG Grids & Arrays | Records electrical activity directly from the cortical surface. | Provides the ground-truth invasive signal; high-density µECoG boosts spatial resolution and decoding accuracy [72]. |
| Stimulus Presentation Software (e.g., MATLAB Psychtoolbox) | Precisely controls the timing and presentation of experimental stimuli. | Critical for ensuring temporal synchronization between stimulus events and neural recordings [9] [98]. |
| Data Analysis Environments (e.g., EEGLAB, SPM, FSL, Custom Python/MATLAB scripts) | Provides a framework for preprocessing, analyzing, and visualizing complex neuroimaging data. | Essential for implementing MVPA pipelines, including feature extraction, classification, and statistical testing [9]. |
Signaling Pathways: Relating Electrical and Hemodynamic Signals
The relationship between non-invasive signals and underlying neural activity is complex. The following diagram synthesizes established findings on how EEG and fMRI signals correlate with invasive ECoG measurements.
Key relationships illustrated include:
- fMRI and ECoG: The fMRI BOLD signal shows a consistent positive correlation with high-frequency ECoG power (the gamma band), which is considered a marker of local neural processing [96]. Its relationship with low-frequency power is more complex and can vary, showing either positive or negative correlations depending on the brain region and context [96].
- EEG and ECoG: The correlation between EEG and ECoG is not straightforward. It is influenced by the specific frequency components examined and the cortical region from which the ECoG signal is recorded [9]. Furthermore, this relationship is affected by the nature of the neural representation, such as its tolerance to changes in stimulus features like orientation and size [9] [97].
Multivariate Pattern Analysis provides a powerful framework for relating the signals from EEG, fMRI, and ECoG to the underlying neural codes. The evidence clearly demonstrates that each modality offers a distinct and complementary window into brain function. EEG excels at tracking the rapid temporal evolution of information, fMRI at pinpointing its spatial location, and ECoG serves as a high-fidelity benchmark for cortical population codes. The relationship between these signals is not unitary but is instead shaped by timing, brain region, and the specific content of the neural representation.
Future progress in this field hinges on technological and analytical advancements. Promising directions include the development of high-density, flexible microelectrode arrays for improved ECoG recording and stimulation [99], the use of more complex non-linear decoding models to leverage the rich information in high-resolution data [72], and the continued refinement of methods for fusing non-invasive data to create more accurate estimates of fast, spatially precise neural dynamics. These advances will be crucial for building a more complete and mechanistic understanding of human cognition.
Motor disorders such as amyotrophic lateral sclerosis (ALS) and locked-in syndrome can severely impair or eliminate verbal communication, dramatically affecting quality of life for affected individuals. Neural speech prostheses offer a promising solution by decoding speech directly from brain signals, potentially restoring natural communication capabilities. The performance of these prostheses fundamentally depends on the quality of neural recordings, which has driven research toward higher-resolution interfaces. Within this context, micro-electrocorticography (µECoG) has emerged as a technology balancing minimal invasiveness with high signal fidelity, occupying a crucial middle ground between non-invasive electroencephalography (EEG), surface electrocorticography (ECoG), and penetrating intracortical microelectrodes.
This review objectively compares the performance characteristics of µECoG against alternative neural recording methodologies, with a specific focus on quantifying the 35% improvement in speech decoding accuracy demonstrated in recent research. We examine the experimental protocols, signal characteristics, and material technologies that enable this enhanced performance, providing researchers with a comprehensive comparison guide grounded in empirical data. The analysis situates µECoG within the broader landscape of neural interface technologies, highlighting its specific advantages for speech decoding applications while acknowledging its limitations relative to more invasive approaches.
Performance Comparison: Quantifying the µECoG Advantage
Key Metrics Across Neural Recording Modalities
Table 1: Performance comparison of neural recording technologies for speech decoding applications
| Technology | Spatial Resolution | Signal-to-Noise Ratio | Invasiveness | Temporal Resolution | Speech Decoding Accuracy | Chronic Stability |
|---|---|---|---|---|---|---|
| EEG | ~1-2 cm | Low | Non-invasive | ~1-100 Hz | Limited | Stable |
| ECoG/SEEG | 4-10 mm | Moderate | High (craniotomy) | 0-200 Hz | Moderate | Weeks to months |
| µECoG | 0.3-1.72 mm | 48% higher than ECoG [72] | Moderate (cranial micro-slit) [33] | 0-500 Hz | 35% improvement over ECoG [72] | Months [92] [33] |
| Intracortical | ~50-400 μm | Highest | High (tissue penetration) | 0-8,000 Hz | Highest (97.5% accuracy demonstrated) [93] | Months to years (with inflammation challenges) [79] |
Quantitative Signal Quality Improvements
Table 2: Specific signal improvements with high-density µECoG for speech decoding
| Parameter | Standard ECoG/SEEG | High-Density µECoG | Improvement |
|---|---|---|---|
| Spatial Resolution | 4-10 mm spacing | 1.33-1.72 mm spacing | 57× higher electrode density [72] |
| Signal-to-Noise Ratio | Baseline reference | 48% higher ESNR [72] | Significant improvement in evoked signal detection |
| Phoneme Decoding | Lower accuracy | 35% improvement [72] | Enhanced articulatory feature resolution |
| High Gamma (70-150 Hz) Capture | Limited spatial specificity | Excellent spatial specificity [72] | Better correlation with neural firing patterns |
| Inter-electrode Correlation | Higher correlation at 4mm spacing [72] | Low inter-electrode correlation (r=0.1-0.3 at 4mm) [72] | Reduced signal redundancy |
Experimental Protocols: Methodologies for Validating µECoG Performance
Intra-operative Speech Production Paradigm
The foundational study demonstrating the 35% speech decoding improvement utilized an intra-operative experimental design with speech-abled patients [72]. The protocol involved implantation of liquid crystal polymer thin-film (LCP-TF) µECoG arrays during deep brain stimulator implantation or tumor resection procedures. Researchers employed two array configurations: a 128-channel subdural array (8×16 configuration with 1.33 mm inter-electrode distance) and a 256-channel subdural array (12×22 configuration with 1.72 mm inter-electrode distance). These arrays provided up to 57× higher electrode density compared to conventional macro-ECoG arrays [72].
During recording sessions, patients performed a speech repetition task involving consonant-vowel-consonant (CVC) or vowel-consonant-vowel (VCV) non-word tokens. The stimulus set contained a fixed inventory of 9 phonemes (4 vowels and 5 consonants) in each position within the token. Participants listened to each auditory stimulus and repeated it aloud while neural signals were recorded. The experimental design included three task blocks with 52 unique tokens per block, with three repetitions of each token overall. Patients correctly repeated non-words with high accuracy (>95%), confirming task engagement despite the intraoperative setting [72].
Neural data analysis focused on high gamma band (HG: 70-150 Hz) activity, which has been shown to correlate with local neural firing and exhibit high spatial specificity. Researchers employed non-parametric permutation tests with FDR correction to identify electrodes with statistically significant HG power increases during speech production compared to pre-stimulus baseline periods. The analysis revealed that 77.4% of significant electrodes were active before speech utterance onset, capturing motor planning activity preceding vocalization [72].
Signal Processing and Decoding Methodology
The recorded neural signals underwent specific processing pipelines to enable accurate speech decoding. After acquisition, signals were re-referenced using techniques such as common average referencing (CAR) to reduce global noise and enhance local neural activity [100]. Researchers then computed the evoked signal-to-noise ratio (ESNR) of HG power during speech production epochs (-500 ms to +500 ms relative to speech onset) and compared these metrics to those obtained from standard intracranial recordings [72].
For decoding analysis, researchers employed both linear and non-linear models to predict spoken phonemes from HG neural activation patterns. The comparison demonstrated that non-linear decoding models designed to exploit the enhanced spatio-temporal information from µECoG recordings outperformed traditional linear techniques. This approach leveraged the rich feature set captured by high-density sampling to achieve the reported 35% improvement in decoding accuracy compared to conventional ECoG and stereoelectroencephalography (SEEG) methods [72].
Comparative Experimental Framework
To validate the superiority of µECoG for speech decoding, researchers implemented a direct comparison framework against standard intracranial recording technologies. This involved quantifying key signal parameters including spatial resolution, signal-to-noise ratio, and inter-electrode correlation structure. The experimental design specifically assessed the ability of each technology to resolve articulatory features of speech, which form the fundamental building blocks for accurate decoding [72].
The evaluation measured the information content available from µECoG's dense spatial sampling compared to the coarser sampling of conventional approaches. By analyzing the distinctness of neural representations for different phonemes across the electrode array, researchers demonstrated that µECoG's increased spatial resolution directly enabled more accurate classification of speech elements. This experimental approach provided the empirical foundation for quantifying the 35% decoding improvement [72].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key materials and technologies for high-density µECoG research
| Component | Specifications | Function | Research Considerations |
|---|---|---|---|
| LCP-TF µECoG Arrays | 128-256 channels, 1.33-1.72 mm spacing, 200 μm electrodes [72] | High-resolution neural recording | 57× higher density than macro-ECoG; 48% higher SNR [72] |
| Platinum Electrodes | Traditional biocompatible metal [92] | Neural signal transduction | Corrosion-resistant; long-term biocompatibility; established fabrication [92] |
| Polyimide/Graphene Substrates | Flexible, biocompatible materials [100] [92] | Electrode array substrate | Conformal cortical contact; reduced immune response [92] |
| Common Average Referencing (CAR) | Signal processing technique [100] | Noise reduction in neural signals | Improves evoked signal-to-noise ratio (ESNR) [100] |
| High Gamma Band Analysis | 70-150 Hz frequency range [72] | Index of local neural activity | Correlates with multi-unit firing; high spatial specificity [72] |
| Non-linear Decoding Models | Machine learning algorithms [72] | Speech feature classification | Leverages spatio-temporal neural patterns; outperforms linear models [72] |
| Cranial Micro-slit Technique | 500-900 μm wide skull incisions [33] | Minimally invasive implantation | Avoids craniotomy; reversible; minimal tissue damage [33] |
Signaling Pathways and Neural Information Flow in Speech Production
The exceptional performance of µECoG in speech decoding applications stems from its ability to capture critical neural signaling pathways at appropriate spatial and temporal scales. During speech production, articulatory commands originate in speech motor cortex (SMC) and involve complex spatiotemporal patterns across cortical layers. µECoG electrodes positioned on the cortical surface primarily capture local field potentials (LFPs) generated by synchronized postsynaptic potentials in pyramidal neurons [92].
The high gamma band (70-150 Hz) activity that proves most informative for speech decoding reflects the ensemble firing of neuronal populations within a few millimeters of each electrode [72]. This high-frequency activity exhibits greater spatial specificity than lower-frequency bands, with studies showing low inter-electrode correlation (r=0.1-0.3 at 4mm spacing) during speech articulation [72]. The rich spatiotemporal information encoded in these signals enables discrimination of subtle articulatory features corresponding to different phonemes.
The signaling pathway begins with motor planning activity in prefrontal and premotor regions, progresses to articulatory programming in ventral sensorimotor cortex, and finally executes through coordinated activity in laryngeal, lingual, and facial motor areas. µECoG's spatial resolution enables tracking of this cascade at the level of individual articulatory features, which form the building blocks for phonemes, words, and ultimately complete sentences [72]. The technology's balance of spatial specificity and broad coverage makes it particularly suited to capturing this distributed yet fine-grained process.
Comparative Analysis: µECoG in the Neural Recording Landscape
Advantages Relative to Alternative Technologies
µECoG occupies a strategic position in the trade-space between signal quality and invasiveness in neural recording technologies. Compared to scalp EEG, µECoG provides dramatically superior spatial resolution and signal-to-noise ratio by bypassing the signal-blurring effects of the skull and scalp [92]. This enables detection of high-frequency neural activity (including the critical high-gamma band) that is essential for detailed speech decoding but is largely absent in EEG recordings.
Relative to conventional ECoG with its typical 4-10 mm electrode spacing, µECoG provides 57× higher spatial resolution with 48% higher signal-to-noise ratio [72]. This enhanced signal quality directly translates to the documented 35% improvement in speech decoding accuracy, as the denser sampling better captures the fine-grained spatial patterns of articulatory representations in speech motor cortex.
Compared to intracortical microelectrodes, µECoG offers reduced invasiveness while maintaining strong decoding performance. Penetrating electrodes, while providing access to single-neuron activity, trigger more substantial neuroinflammatory responses that can compromise long-term signal stability [79]. µECoG's surface placement elicits less tissue damage and immune response, contributing to better chronic recording stability [92] [33].
Limitations and Performance Ceilings
Despite its advantages for speech decoding, µECoG does have inherent limitations. As a surface recording technology, it primarily captures activity from superficial cortical layers, potentially missing information encoded in deeper layers [1]. While µECoG signals approximate those of penetrating electrodes in some contexts, they cannot resolve single-neuron spiking activity, which may contain additional information valuable for decoding [93].
For the most demanding applications requiring the highest information transfer rates, intracortical arrays have demonstrated superior performance, with one study achieving 97.5% accuracy with a 125,000-word vocabulary [93]. However, this comes with increased surgical invasiveness and potential long-term stability challenges due to the more aggressive tissue response to penetrating electrodes [79].
The quantified 35% improvement in speech decoding accuracy with high-resolution µECoG represents a significant advancement in neural interface technology. This performance enhancement, coupled with the technique's favorable invasiveness profile, positions µECoG as a compelling platform for next-generation speech prostheses. The experimental data demonstrate that increased spatial sampling density directly enhances the ability to resolve articulatory features, enabling more accurate phoneme classification and ultimately more naturalistic speech synthesis.
Future research directions include further increasing electrode densities while maintaining biocompatibility, developing wireless implementations for chronic use, and advancing non-linear decoding algorithms to more fully exploit the rich spatiotemporal patterns captured by µECoG. Additionally, hybrid approaches combining µECoG with limited intracortical sampling may offer pathways to capture both broad network dynamics and highly localized neural activity. As these technologies mature, high-density µECoG appears poised to play a central role in restoring communication capabilities for individuals with severe speech impairments.
Electroencephalography (EEG) and magnetoencephalography (MEG) are fundamental, non-invasive techniques for investigating brain activity with millisecond temporal resolution, yet they exhibit distinct and complementary sensitivity profiles to neural sources [101] [102]. Understanding these differences is crucial for selecting the appropriate modality for specific neuroscientific inquiries or clinical diagnostics, particularly within research focused on comparing the signal-to-noise ratios (SNR) of various neural recording techniques. This guide provides an objective, data-driven comparison of EEG and MEG sensitivity for cortical and subcortical sources, summarizing key quantitative findings and the experimental protocols that underpin them. The central thesis, supported by recent computational studies, is that while MEG demonstrates superior SNR for superficial, tangentially oriented cortical sources, EEG retains a significant advantage for probing radial and deep brain sources, including those located subcortically [101] [103] [19].
The differential sensitivity of EEG and MEG originates from the fundamental physics of bioelectric currents and the properties of the head as a volume conductor. EEG records the electrical potential distribution generated by intracranial current sources on the scalp surface. In contrast, MEG measures the minute magnetic fields induced by these same currents [101] [104].
A critical difference, often simplified by the spherical head model, is MEG's pronounced insensitivity to radially oriented sources—those pointing directly toward or away from the scalp surface. In an idealized spherical conductor, a radial source produces no detectable magnetic field outside the head [102]. Although the human head is not a perfect sphere, this property largely holds in realistic head models, where MEG sensitivity remains highly dependent on source orientation. Computational studies show that for most cortical locations, there exists a source orientation to which MEG is nearly insensitive, with the median signal magnitude for the lowest-sensitivity orientation being only 6% of that for the highest-sensitivity orientation. For EEG, this ratio is substantially higher at 63%, indicating its more uniform sensitivity across different source orientations [102].
Table 1: Fundamental Characteristics of EEG and MEG
| Feature | EEG (Electroencephalography) | MEG (Magnetoencephalography) |
|---|---|---|
| Measured Quantity | Electrical potential on scalp (µV) | Magnetic field outside head (fT) |
| Primary Source | Primarily postsynaptic potentials | Primarily postsynaptic potentials |
| Sensitivity to Radial Sources | High | Very Low |
| Sensitivity to Tangential Sources | High | High |
| Impact of Skull & CSF | High (Skull strongly attenuates signal) | Low (Magnetic fields are less distorted) |
| Typical Sensor Count | 64 - 256 electrodes | 100 - 300 magnetometers |
Advanced finite element method (FEM) head models have enabled precise quantification of EEG and MEG SNR across the cortex. The choice of head model complexity significantly impacts the accuracy of these predictions, especially for EEG.
The Critical Role of Head Modeling
The accuracy of sensitivity maps is contingent on the fidelity of the head volume conductor model. A key finding is that ignoring the cerebrospinal fluid (CSF) compartment leads to an overestimation of EEG SNR values [101] [103]. The CSF, with its high conductivity, acts as a shunt for electrical currents, and its omission in simpler three-compartment models (scalp, skull, brain) results in inaccurate forward solutions. Highly detailed, six-compartment anisotropic head models (including scalp, skull compacta, skull spongiosa, CSF, gray matter, and anisotropic white matter) are now considered the gold standard for generating reliable sensitivity maps [101].
Key SNR Findings on the Cortical Surface
Computational studies using such detailed models reveal consistent patterns:
- MEG's Superiority for Tangential Sources: The majority of sources located in the sulcal walls (gyri) are tangentially oriented relative to the scalp. For these sources, MEG typically exhibits a higher SNR than EEG [101] [19].
- EEG's Advantage for Radial and Deep Sources: EEG sensitivity is higher for radially oriented sources, often found at the crowns of gyri, and for deeper cortical sources [101]. The sensitivity of MEG for superficial sources shows a strong depth dependency, decaying rapidly with increasing source-to-sensor distance [102] [19].
- Overall SNR Uniformity: When considering the entire cortex, the spatial distribution of SNR is generally more uniform for EEG than for MEG [19].
Table 2: SNR Comparison for Cortical Sources
| Parameter | EEG | MEG | Notes |
|---|---|---|---|
| Median Relative Sensitivity (Rλ) | 0.63 | 0.06 | Ratio of signal magnitude for lowest vs. highest sensitivity orientation [102]. |
| Deep Source Sensitivity | Higher | Lower | EEG maintains better SNR for sources deeper within sulci or fissures [101] [19]. |
| Impact of Head Model | High (CSF critical) | Moderate | Realistic modeling of skull and CSF is essential for accurate EEG maps [101]. |
| Spatial SNR Distribution | More Uniform | Less Uniform | MEG SNR is more focal, concentrated over superficial cortical areas [19]. |
A long-held assumption has been that MEG is incapable of detecting subcortical activity. However, recent comprehensive studies challenge this notion. While EEG is unequivocally more sensitive to deep sources overall, MEG is not fully insensitive to subcortical sources [101] [103].
The critical factor for MEG's ability to record subcortical activity is the orientation of the source. Deep sources that possess a sufficient tangential component can generate a measurable magnetic field outside the head [101]. This finding expands the potential applications of MEG in investigating deep brain structures, such as the thalamus, hippocampus, or basal ganglia, although detecting such signals remains challenging due to their low amplitude and distance from sensors.
Experimental Protocols for Sensitivity Mapping
The findings discussed above are derived from sophisticated computational simulations. The following workflow outlines the standard methodology for generating and comparing EEG/MEG sensitivity maps.
Workflow for Sensitivity Map Generation
The process of creating SNR maps involves acquiring anatomical data, constructing a computational head model, and calculating forward solutions for a distribution of putative sources.
Key Methodological Components
- MRI Data Acquisition: High-resolution structural MRIs (T1-weighted and T2-weighted) are acquired to define head tissue geometry. Diffusion Tensor Imaging (DTI) may be used to model the anisotropic conductivity of white matter [101].
- Head Model Generation: Tissue compartments (e.g., scalp, skull, CSF, brain) are segmented from the MRI data. The Finite Element Method (FEM) is often preferred for its ability to model complex geometries and anisotropic tissue properties accurately [101]. Studies compare models of varying complexity, from isotropic three-compartment (3CI) to anisotropic six-compartment (6CA) models [101] [103].
- Forward Solution Calculation: The forward problem—predicting sensor measurements for a given neural source—is solved for thousands of candidate source locations on the cortical surface and in subcortical structures. Sources are typically modeled as current dipoles, oriented perpendicular to the cortical surface [102] [19].
- Noise Estimation and SNR Calculation: The SNR for a source at location k is calculated as ( \text{SNR}k = 10 \log{10} \frac{\sum{i=1}^N (a \cdot b{k, i})^2}{\sum{i=1}^N \sigmai^2} ), where a is the source amplitude, ( b{k, i} ) is the forward solution for sensor *i*, and ( \sigmai^2 ) is the noise variance at sensor i [19]. Noise can be estimated from empty-room recordings (MEG) or resting-state data, or modeled as originating from a uniform distribution of background brain sources [19].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Essential Tools for EEG/MEG Sensitivity Research
| Tool / Solution | Function in Research | Examples / Specifications |
|---|---|---|
| High-Density EEG System | Records electrical potential from scalp with high spatial sampling. | 64-256 channel systems with Ag/AgCl electrodes; Amplifiers with high input impedance. |
| Whole-Head MEG System | Measures femtotesla-scale magnetic fields generated by neural currents. | System with 100+ magnetometers or planar gradiometers housed in a magnetically shielded room. |
| MRI Scanner | Provides anatomical data for individual head model construction. | 3T scanners; T1w and T2w sequences; DTI for anisotropy. |
| FEM/BEM Software | Computes accurate forward solutions in realistic head geometries. | SimNIBS, MNE-Python, FieldTrip. |
| Cortical Segmentation Tool | Reconstructs the cortical surface from MRI. | FreeSurfer, FSL, SPM. |
| Anisotropic Conductivity Model | Models the direction-dependent electrical conductivity of white matter. | Derived from DTI data; critical for model accuracy [101]. |
EEG and MEG offer complementary windows into brain dynamics. The choice between them—or the decision to use them simultaneously—should be informed by the specific neural targets of interest. MEG is the superior tool for mapping superficial, tangentially oriented cortical activity, which constitutes a large portion of brain signals. EEG, however, provides a more comprehensive view of brain activity, including contributions from radial and deep sources, both cortical and subcortical. Future advancements in wearable MEG technology, such as optically-pumped magnetometers (OPMs), and sophisticated multimodal integration with fMRI and DOT, promise to further enhance our ability to map brain function with high spatiotemporal resolution across the entire brain [105] [106]. For researchers focused on signal-to-noise ratio comparisons across neurorecording techniques, this analysis underscores that neither EEG nor MEG is universally superior; their efficacy is intrinsically linked to the neuroanatomical and neurophysiological context of the research question.
The advancement of Brain-Computer Interfaces (BCIs) from laboratory demonstrations to real-world applications creates a pressing need for robust benchmarking standards. Performance evaluation enables researchers to compare different approaches, identify effective strategies, and drive the field forward [107]. Historically, BCI benchmarking focused on offline analysis of previously recorded datasets, allowing comparison of signal processing algorithms using identical raw data [107]. However, this approach fails to capture the complexities of online performance, where users learn to compensate for systematic errors and dynamically interact with controlled devices [107] [108]. Real-world BCI applications demand benchmarking paradigms that quantify performance under realistic conditions while accounting for the critical engineering trade-offs between speed, accuracy, and latency [109].
The selection of neural recording modality—non-invasive electroencephalography (EEG), invasive electrocorticography (ECoG), or intracortical interfaces—fundamentally influences this performance by determining the available signal-to-noise ratio (SNR) and information content. Each modality presents a different set of capabilities and limitations for communication and control tasks. This comparison guide objectively evaluates the performance of these principal BCI modalities, framing the analysis within the context of SNR characteristics and their implications for real-world application.
Performance Metrics and Quantitative Comparison
Benchmarking BCI performance requires multiple complementary metrics, as no single measure fully captures system capabilities. Key metrics include information transfer rate (ITR), measured in bits per second (bps), which quantifies how much information is communicated per unit time; classification accuracy, which measures the correct classification of intended commands; task completion time, which reflects practical performance in applied settings; and system latency, which measures the delay between brain activity and system response and is critical for real-time interaction [107] [109]. The SNR of the recorded neural signal underlies all these metrics, directly influencing decoding accuracy and ultimately the feasibility of different BCI applications [10].
Table 1: Comparative Performance Metrics Across BCI Modalities
| Recording Modality | Information Transfer Rate (ITR) | Typical Applications | Key Advantages | Primary Limitations |
|---|---|---|---|---|
| Non-invasive (EEG) | Low (Highly variable by paradigm) | SSVEP, P300, Motor Imagery [110] [111] | Completely non-invasive, safe, low-cost | Low spatial resolution, low SNR, vulnerable to artifacts [9] [15] |
| ECoG | Medium | Motor decoding, Epilepsy focus localization [10] | High SNR, good spatial resolution | Requires craniotomy, limited cortical coverage [10] |
| Intracortical | High (e.g., 200+ bps [109]) | High-performance communication, complex control [109] | Very high SNR, high spatial and temporal resolution | Most invasive, surgical risk, signal stability over time |
Table 2: Signal Quality Comparison Across Invasive Modalities
| Metric | Endovascular (Stentrode) | Subdural (SD) | Epidural (ED) |
|---|---|---|---|
| Signal Bandwidth | Comparable to SD and ED [10] | Conventional standard [10] | Comparable to SD [10] |
| Signal-to-Noise Ratio (SNR) | Not significantly different from SD and ED [10] | Conventional standard [10] | Not significantly different from SD [10] |
| Spatial Resolution | Location and frequency dependent [10] | High [10] | Lower than SD [10] |
| Decoding Accuracy | Comparable to SD and ED [10] | High [10] | Comparable to SD [10] |
| Implantation Risk | Minimally invasive (no craniotomy) [10] | High (requires craniotomy) [10] | High (requires craniotomy) [10] |
Quantitative data reveals distinct performance tiers. Modern intracortical interfaces, such as the Paradromics Connexus BCI, have demonstrated ITRs exceeding 200 bps with minimal latency (56ms), a rate that surpasses the information content of transcribed human speech [109]. ECoG provides intermediate performance with SNRs substantially higher than non-invasive EEG [15] [10]. Recent innovations in non-invasive approaches, such as the steady-state motion visual evoked potential (SSMVEP) paradigm, have achieved accuracies of 83.81% ± 6.52% by integrating motion and color stimuli to enhance attention and reduce fatigue [110].
Benchmarking Methodologies and Experimental Protocols
Real-World Task Performance: The Cybathlon Competition
The Cybathlon competition established a pioneering benchmarking approach using a computer game that mimics real-world assistive technology tasks. The primary metric was task completion time in a competitive racing game, where pilots with severe physical disabilities used BCIs to control avatars. This method emphasized practical performance under realistic conditions over idealized laboratory metrics [107]. The rules governed acceptable hardware, software, and the essential inclusion of human pilots, ensuring fair comparison across different BCI approaches. This benchmarking strategy successfully highlighted how BCI performance depends on the entire system, including the user's ability to learn and adapt [107].
Cybathlon Benchmarking Workflow
Standardized Algorithm Evaluation: The MOABB Framework
For reproducible comparison of signal processing algorithms, the MOABB (Mother of All BCI Benchmarks) open-source framework was created. MOABB addresses the reproducibility crisis in BCI research by providing a standardized platform for evaluating machine learning models across multiple EEG datasets [111]. It supports major BCI paradigms (Motor Imagery, P300, SSVEP) and enables both within-subject and cross-subject evaluations. The framework incorporates state-of-the-art deep learning and machine learning models, offers hyperparameter tuning via Grid Search, and even evaluates the computational carbon footprint of different algorithms [111].
Engineering-Focused Benchmarks: The SONIC Standard
Paradromics introduced the SONIC (Standard for Optimizing Neural Interface Capacity) benchmark to rigorously measure BCI performance using mutual information between presented and decoded stimuli. In preclinical experiments, controlled sound sequences are presented, and the BCI decodes which sounds were presented based on recorded neural activity [109]. SONIC specifically accounts for both ITR and latency, preventing systems from gaming results with long delays. This benchmark has demonstrated the capability of intracortical interfaces to achieve over 200 bps with 56ms latency, setting a new industry standard for high-performance neural interfaces [109].
SONIC Benchmarking Methodology
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Tools for BCI Benchmarking
| Tool Category | Specific Examples | Research Function |
|---|---|---|
| Recording Hardware | g.USBamp (g.tec), Intan RHD, Blackrock Microsystems, Custom implants [110] [109] [10] | Acquires raw neural signals with specific noise and resolution characteristics. |
| Electrode Types | Wet/dry EEG electrodes, ECoG grid electrodes, Utah arrays, Stentrode endovascular arrays [107] [10] | Interface with neural tissue, determining signal source and quality. |
| Stimulation Paradigms | SSVEP, SSMVEP, P300, Motor Imagery tasks [110] [111] | Elicit reproducible, classifiable brain responses for BCI control. |
| Software Frameworks | MOABB, EEGLAB, BCILAB, Psychophysics Toolbox [9] [111] | Provide standardized data processing, analysis, and benchmarking pipelines. |
| Decoding Algorithms | EEGNet, CSP, Riemannian Geometry, Filter Bank CCA, TRCA [110] [111] | Translate neural signals into device commands with varying efficiency. |
| Performance Metrics | SONIC benchmark, Information Transfer Rate, Classification Accuracy, Task Completion Time [107] [109] | Quantify and compare BCI performance across systems and paradigms. |
Signaling Pathways and Neural Basis of BCI Control
The effectiveness of different BCI paradigms can be understood through their engagement of specific neural pathways. Visual evoked potentials, such as SSVEP and the newer SSMVEP, primarily activate the visual pathways from retina to visual cortex [110]. The dorsal stream (M-pathway) is crucial for motion detection, while the ventral stream (P-pathway) processes color and object identification [110]. Bimodal stimulation that engages both pathways simultaneously can enhance response intensity and improve SNR [110].
Neurofeedback and BCI control engage higher-order cognitive networks. Research using real-time fMRI has demonstrated that BCI control increases whole-brain SNR compared to passive task performance [108]. This enhancement involves a positive network including dorsal parietal and frontal regions and the anterior insula of the right hemisphere, along with decreased activity in default mode network regions [108]. This network regulation likely supports the improved classification accuracy observed during active BCI control.
Neural Pathways in BCI Control
Benchmarking real-world BCI performance requires a multifaceted approach that considers the interplay between recording modality, signal processing algorithms, and user learning. The SNR advantages of invasive modalities like ECoG and intracortical interfaces enable higher ITRs and more complex control schemes, but must be balanced against their increased invasiveness and surgical risks [10]. Emerging standards like the Cybathlon tasks, MOABB framework, and SONIC benchmark provide complementary tools for rigorous, reproducible performance evaluation across different BCI approaches [107] [109] [111]. Future progress will depend on continued refinement of these benchmarking standards alongside technological innovations that enhance SNR while minimizing invasiveness, ultimately expanding the practical applications of BCIs for communication and control.
Conclusion
The comparative analysis of EEG, ECoG, and intracortical recordings reveals a clear, fundamental trade-off: increases in signal-to-noise ratio and spatial resolution are intrinsically linked to greater invasiveness. While intracortical microelectrodes offer unparalleled access to single-neuron activity, recent advancements in high-density µECoG provide a compelling compromise, delivering a significant boost in SNR and spatial resolution for superior speech and sensory decoding with a reduced chronic immune response compared to penetrating electrodes. EEG remains indispensable for non-invasive monitoring, though its low SNR necessitates sophisticated processing techniques. The future of neural interfaces lies in the continued development of high-density, biocompatible arrays, the integration of nonlinear decoding models that fully leverage high-SNR data, and the rigorous validation of signals against artifacts like crosstalk. These directions will be crucial for translating high-fidelity neural recordings into robust clinical neuroprosthetics and refined tools for drug development and basic neuroscience research.
References
- 1. Comparison of Subdural and Intracortical Recordings ... [https://www.mdpi.com/1424-8220/24/21/6847]
- 2. - Signal - to - Wikipedia noise ratio [https://en.wikipedia.org/wiki/Signal-to-noise_ratio]
- 3. Measuring the signal-to-noise ratio of a neuron [https://pmc.ncbi.nlm.nih.gov/articles/PMC4466709/]
- 4. How is the signal - to - noise of an event-related-potential measured? ratio [https://psychology.stackexchange.com/questions/8930/how-is-the-signal-to-noise-ratio-of-an-event-related-potential-measured]
- 5. Signal Amplification and Noise Reduction in ... [https://www.labx.com/resources/signal-amplification-and-noise-reduction-in-electrophysiology-data/5624]
- 6. Correlation between scalp-recorded ... [https://www.sciencedirect.com/science/article/pii/S1059131106002482]
- 7. Cortical Electrocorticogram (ECoG) Is a Local Signal - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6538865/]
- 8. Simultaneous recording of ECoG and intracortical neuronal ... [https://www.sciencedirect.com/science/article/abs/pii/S1053811910010700]
- 9. A multivariate comparison of electroencephalogram and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9624066/]
- 10. Signal quality of simultaneously recorded endovascular, ... [https://www.nature.com/articles/s41598-018-26457-7]
- 11. Depth-to-scalp spatiotemporal dynamics for stereo-EEG [https://pmc.ncbi.nlm.nih.gov/articles/PMC12399257/]
- 12. Noninvasive source localization of interictal EEG spikes [https://pubmed.ncbi.nlm.nih.gov/17143137/]
- 13. Dataset of Speech Production in intracranial ... [https://www.nature.com/articles/s41597-022-01542-9]
- 14. The origin of extracellular fields and currents — EEG, ECoG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4907333/]
- 15. Signal Quality of Simultaneously Recorded ECoG and Non ... [https://www.sciencedirect.com/science/article/pii/S105381190971211X]
- 16. Predicting Movement from Multiunit Activity - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6673077/]
- 17. 5.3 Comparison of ECoG and intracortical signals [https://fiveable.me/brain-computer-interfaces/unit-5/comparison-ecog-intracortical-signals/study-guide/ahyTx7o9wtbS31R2]
- 18. Elucidating relations between fMRI, ECoG, and EEG ... [https://www.sciencedirect.com/science/article/abs/pii/S1053811918305238]
- 19. Mapping the signal‐to‐noise‐ratios of cortical sources in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2882168/]
- 20. Comparison of Subdural and Intracortical Recordings ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11548369/]
- 21. Decoding Movement From Electrocorticographic Activity [https://www.frontiersin.org/journals/neuroinformatics/articles/10.3389/fninf.2019.00074/full]
- 22. Two-Dimensional Movement Control Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2744037/]
- 23. The history, current state and future possibilities of the non- ... [https://www.sciencedirect.com/science/article/pii/S2590093525000049]
- 24. Long-term evaluation and feasibility study of the insulated ... [https://www.sciencedirect.com/science/article/abs/pii/S016502701830205X]
- 25. Advances in flexible high-density microelectrode arrays for ... [https://www.sciencedirect.com/science/article/pii/S0956566325009790]
- 26. EEG-based brain-computer interface enables real-time ... [https://www.nature.com/articles/s41467-025-61064-x]
- 27. Impact of Brain Surface Boundary Conditions on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7438758/]
- 28. Evaluating the Impact of Tissue Layers on Signal Quality ... [https://arxiv.org/html/2506.03452v1]
- 29. Electrocorticography (ECoG) | Thesen Lab [https://med.nyu.edu/thesenlab/research-0/intracranial-eeg/]
- 30. Materials and devices for high‐density, high‐throughput ... [https://www.sciencedirect.com/science/article/pii/S2667325824000402]
- 31. High-resolution neural recordings improve the accuracy of ... [https://pubmed.ncbi.nlm.nih.gov/37932250/]
- 32. High-density electrocorticography for speech decoding and ... [https://www.news-medical.net/news/20231108/High-density-electrocorticography-for-speech-decoding-and-improved-neural-speech-prostheses.aspx]
- 33. Minimally invasive implantation of scalable high-density ... [https://www.nature.com/articles/s41551-025-01501-w]
- 34. Real-time detection of spoken speech from unlabeled ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12498269/]
- 35. A high-performance neuroprosthesis for speech decoding and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10826467/]
- 36. Guidewire-driven deployment of high density ECoG arrays ... [https://arxiv.org/abs/2511.12907]
- 37. A highly stable electrode with low electrode-skin ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566322007965]
- 38. Quantification of Signal-to-Noise Ratio in Cerebral Cortex ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6282047/]
- 39. Brain Computer Interfaces 2025-2045: Technologies, ... [https://www.idtechex.com/en/research-report/brain-computer-interfaces/1024]
- 40. Electrode modifications to lower electrode impedance and ... [https://pubmed.ncbi.nlm.nih.gov/26394650/]
- 41. Quantifying physical degradation alongside recording and ... [https://www.sciencedirect.com/science/article/pii/S1742706125001151]
- 42. Modification of electrodes with self-assembled, close- ... [https://www.sciencedirect.com/science/article/pii/S1388248121002459]
- 43. ECG signal quality in intermittent long-term dry electrode ... [https://www.nature.com/articles/s41598-024-56595-0]
- 44. Characterization of High-Gamma Activity in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10440607/]
- 45. Characterization of High-Gamma Activity in ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2023.1206120/full]
- 46. Comparison of subdural and subgaleal recordings of cortical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4600028/]
- 47. Including Measures of High Gamma Power Can Improve ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2020.00130/full]
- 48. Stability of ECoG high gamma signals during speech and ... [https://pubmed.ncbi.nlm.nih.gov/38925110/]
- 49. Comparison of tuning properties of gamma and high ... [https://www.nature.com/articles/s41598-020-61961-9]
- 50. Intracortical recordings reveal the neuronal selectivity for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11665852/]
- 51. Enhancing motor imagery EEG signal decoding through ... [https://pubmed.ncbi.nlm.nih.gov/39672015/]
- 52. Enhancing motor imagery EEG signal decoding through ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482524016196]
- 53. Comparison metrics and power trade-offs for BCI motor ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1547074/full]
- 54. Motor imagery EEG signal classification using novel deep ... [https://www.nature.com/articles/s41598-025-00824-7]
- 55. Feature reweighting for EEG-based motor imagery ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809425007268]
- 56. A study of motor imagery EEG classification based on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12307489/]
- 57. A hybrid approach for EEG motor imagery classification ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482525010261]
- 58. Towards decoding motor imagery from EEG signal using ... [https://www.nature.com/articles/s41598-025-05423-0]
- 59. EEG Signal Prediction for Motor Imagery Classification in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11991189/]
- 60. Artificial Intelligence Approaches for EEG Signal Acquisition ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12390113/]
- 61. Real-time detection of spoken speech from unlabeled ... [https://pubmed.ncbi.nlm.nih.gov/40972658/]
- 62. BCIs in 2025: Trials, Progress, and Challenges [https://andersenlab.com/blueprint/bci-challenges-and-opportunities]
- 63. Intraoperative Monitoring of Sensory Evoked Potentials in ... [https://www.mdpi.com/2075-4426/15/1/26]
- 64. Intraoperative Monitoring of Sensory Evoked Potentials in ... [https://www.preprints.org/manuscript/202411.1858/v1]
- 65. Optimization of signal-to-noise ratio in short-duration SEP ... [https://www.sciencedirect.com/science/article/pii/S1388245723002365]
- 66. Multimodal intraoperative neurophysiological monitoring ... [https://www.sciencedirect.com/science/article/pii/S1388245724003122]
- 67. A low-latency neural inference framework for real-time ... [https://www.nature.com/articles/s41598-025-24972-y]
- 68. EEG-Driven Arm Movement Decoding - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC12189900/]
- 69. A systematic review of EEG-based Imagined Speech ... [https://www.sciencedirect.com/science/article/abs/pii/S1568494625008749]
- 70. Bridging circuit modeling and signal analysis to understand ... [https://www.nature.com/articles/s41467-025-59391-0]
- 71. Theoretical analysis of intracortical microelectrode recordings [https://pmc.ncbi.nlm.nih.gov/articles/PMC3196618/]
- 72. High-resolution neural recordings improve the accuracy of ... [https://www.nature.com/articles/s41467-023-42555-1]
- 73. Probe powerfully records neural circuits during behavior [https://newsroom.uw.edu/news-releases/probe-powerfully-records-neural-circuits-during-behavior]
- 74. Real-time, neural signal processing for high-density brain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12275325/]
- 75. techniques and electrode types | Fiveable Intracortical recording [https://fiveable.me/brain-computer-interfaces/unit-5/intracortical-recording-techniques-electrode-types/study-guide/SrzCQggHd83D0coS]
- 76. Revolutionizing brain‒computer interfaces - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12243264/]
- 77. Mechanically-adaptive, resveratrol-eluting neural probes ... [https://www.nature.com/articles/s41528-025-00440-5]
- 78. PEDOT:PSS-based bioelectronics for brain monitoring and modulation [https://www.nature.com/articles/s41378-025-00948-w?error=cookies_not_supported&code=1bb2e90b-5fc5-42bd-b76e-e3c7454bfa6b]
- 79. Antimicrobial coated intracortical probes reduce invading ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961225007537]
- 80. Dexamethasone-loaded platelet-inspired nanoparticles ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12480985/]
- 81. Enhancing oscillations in intracranial electrophysiological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8407590/]
- 82. CARLA: Adjusted common average referencing for cortico ... [https://www.sciencedirect.com/science/article/abs/pii/S0165027024000980]
- 83. A practical guide to the selection of independent ... [https://www.sciencedirect.com/science/article/abs/pii/S0165027015000928]
- 84. Independent Component Analysis for artifact removal [https://eeglab.org/tutorials/06_RejectArtifacts/RunICA.html]
- 85. Frontiers | Combination of spatial and temporal de- noising and artifact... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1576954/full]
- 86. A Comparative Study of Conventional and Tripolar EEG for... [https://arxiv.org/html/2402.09448v2]
- 87. Analysis of human ECoG and sEEG recordings [https://www.fieldtriptoolbox.org/tutorial/intracranial/human_ecog/]
- 88. A synergistic framework integrating graph-based spatial ... [https://www.sciencedirect.com/science/article/pii/S0925231225022337]
- 89. A deep neural network for adaptive spatial smoothing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12070436/]
- 90. Better Exploiting Spatial Separability in Multichannel ... [https://research.samsung.com/blog/Better-Exploiting-Spatial-Separability-in-Multichannel-Speech-Enhancement-with-an-Align-and-Filter-Network]
- 91. Revealing Spatiotemporal Neural Activation Patterns in ... [https://www.sciencedirect.com/science/article/pii/S2772528625000470]
- 92. Progress in the Field of Micro-Electrocorticography - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6356841/]
- 93. Electrocorticography (ECoG) surveys the landscape [https://www.paradromics.com/blog/electrocorticography]
- 94. The size of via holes influence the amplitude and selectivity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8935835/]
- 95. Advances in large-scale electrophysiology with high-density ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12394932/]
- 96. Elucidating relations between fMRI, ECoG, and EEG through a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6063527/]
- 97. A multivariate comparison of electroencephalogram and ... [https://pubmed.ncbi.nlm.nih.gov/36330341/]
- 98. A multivariate comparison of electroencephalogram and ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2022.983602/full]
- 99. Towards precise synthetic neural codes: high-dimensional ... [https://www.nature.com/articles/s41528-025-00447-y]
- 100. A Novel μECoG Electrode Interface for Comparison of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7478123/]
- 101. A comprehensive study on electroencephalography and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7856654/]
- 102. Sensitivity of MEG and EEG to Source Orientation [https://link.springer.com/article/10.1007/s10548-010-0154-x]
- 103. A comprehensive study on electroencephalography and ... [https://pubmed.ncbi.nlm.nih.gov/33156569/]
- 104. Magnetoencephalography - Latest research and news [https://www.nature.com/subjects/magnetoencephalography]
- 105. Highlights from MEG-TREC 2025 [https://fixel.ufhealth.org/2025/05/01/highlights-from-meg-trec-2025/]
- 106. Simultaneously Acquired Magnetoencephalography and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12301506/]
- 107. Benchmarking Brain-Computer Interfaces Outside the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5768650/]
- 108. Brain–computer interfaces increase whole-brain signal to noise [https://pmc.ncbi.nlm.nih.gov/articles/PMC3746889/]
- 109. Think Fast | Setting new standards and new records for ... [https://www.paradromics.com/blog/bci-benchmarking]
- 110. Frontiers | Improvement of BCI with bimodal SSMVEPs... performance [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1506104/full]
- 111. How to Compare BCI Models? [https://medium.com/neurotechx/how-to-compare-bci-models-d227bd401b16]