Invasive vs. Non-Invasive Neural Recordings: A Comprehensive Analysis of Spatial and Temporal Resolution for Biomedical Research
This article provides a detailed comparative analysis of the spatial and temporal resolution capabilities of invasive and non-invasive neural recording techniques, tailored for researchers, scientists, and drug development professionals.
Invasive vs. Non-Invasive Neural Recordings: A Comprehensive Analysis of Spatial and Temporal Resolution for Biomedical Research
Abstract
This article provides a detailed comparative analysis of the spatial and temporal resolution capabilities of invasive and non-invasive neural recording techniques, tailored for researchers, scientists, and drug development professionals. It explores the fundamental biophysical principles governing signal fidelity, reviews current methodologies from intracortical arrays to EEG and fMRI, and analyzes performance limitations and optimization strategies. The content critically evaluates real-world application suitability across clinical and research scenarios, offering evidence-based insights to guide method selection for specific biomedical objectives, from high-precision neuroprosthetics to large-scale brain monitoring.
Fundamental Principles of Neural Signal Acquisition: From Biophysics to Measurement
Defining Spatial and Temporal Resolution in Neural Recording Contexts
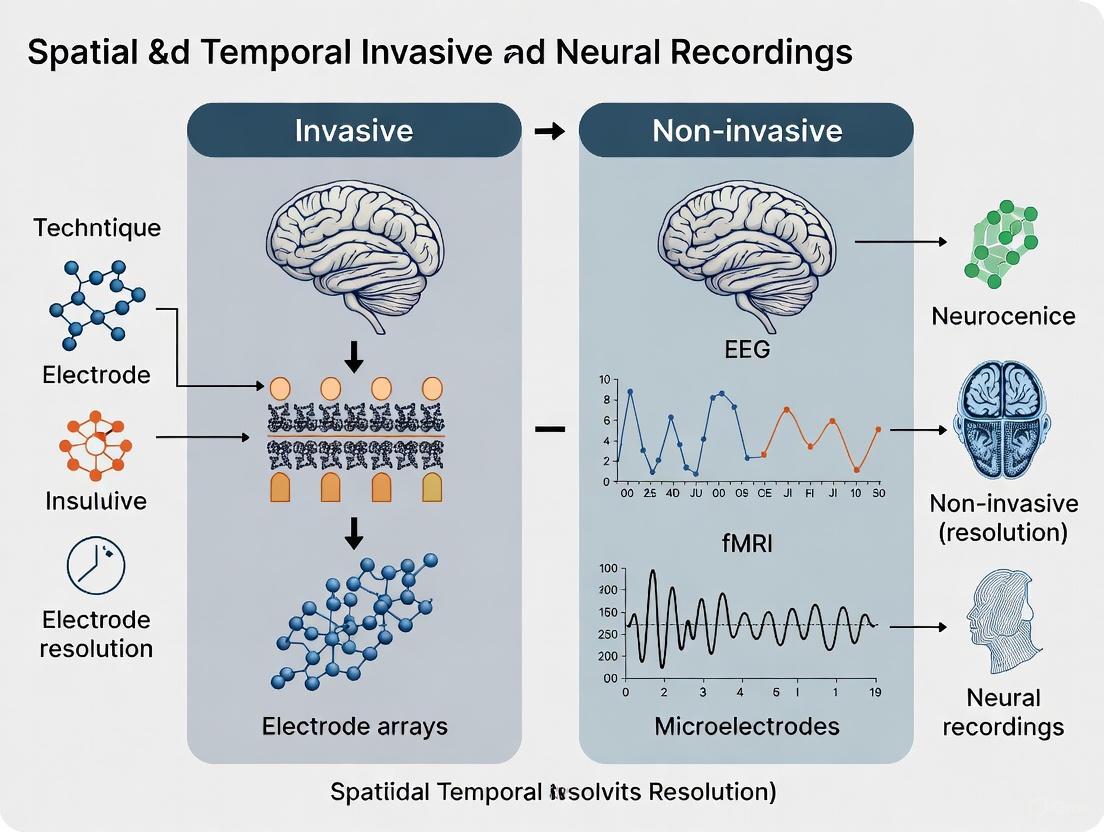
Understanding the capabilities and limitations of neural recording technologies is fundamental to neuroscience research and neurotechnology development. Spatial and temporal resolution represent two core characteristics that define the fidelity with which these tools can capture brain activity. Spatial resolution refers to the smallest physical distance between distinct neural sources that can be discriminated, while temporal resolution refers to the precision in measuring when neural events occur over time. These parameters form a critical trade-off landscape that fundamentally distinguishes invasive techniques, which record directly from neural tissue, from non-invasive approaches, which measure through the skull and other biological barriers [1]. This technical guide examines the defining parameters of current neural recording methodologies, providing a framework for selecting appropriate technologies based on research requirements in the context of invasive versus non-invasive approaches.
Quantitative Comparison of Neural Recording Technologies
The following tables summarize key spatial and temporal resolution parameters for major neural recording techniques, highlighting the fundamental divide between invasive and non-invasive methodologies.
Table 1: Spatial and Temporal Resolution Characteristics of Neural Recording Techniques
| Technique | Spatial Resolution | Temporal Resolution | Invasiveness |
|---|---|---|---|
| Neuropixels Ultra | ~6 μm electrode spacing [2] | >1 kHz (millisecond) [3] | Invasive |
| 2P Calcium Imaging | Subcellular (can resolve individual components) [3] | 5-25 Hz (limited by calcium dynamics) [3] | Invasive |
| 2P Voltage Imaging | Subcellular (can resolve individual components) [3] | 0.2-4 kHz (millisecond) [3] | Invasive |
| ECoG | Millimeter scale (cortical surface) | Millisecond | Invasive (surface) |
| Digital Holographic Imaging | Not specified (through scalp) [4] | Not specified (neural tissue deformation) [4] | Non-invasive |
| EEG | Centimeter scale (limited by skull) [1] | Millisecond [5] | Non-invasive |
| MEG | Millimeter to centimeter [5] | Millisecond [5] | Non-invasive |
| fNIRS | ~1-2 cm (limited by light scattering) | Seconds (hemodynamic response) | Non-invasive |
| fMRI | Millimeter scale [5] | Seconds (hemodynamic response) [5] | Non-invasive |
Table 2: Performance Characteristics of Invasive Recording Methods in Rodent Models
| Method | Typical # of Neurons | Temporal Resolution | Subcellular Resolution | Freely Moving? |
|---|---|---|---|---|
| 1P-Ca Head Mounted | 100-500 [3] | 5-25 Hz [3] | No [3] | Yes [3] |
| 2P-Ca Head Mounted | 10-100 [3] | 5-25 Hz [3] | Yes [3] | Yes [3] |
| Microwire Electrodes | ~20 per probe [3] | >1 kHz [3] | No [3] | Yes [3] |
| Silicon Electrodes | ~100 per probe [3] | >1 kHz [3] | No [3] | Yes [3] |
| Next-gen Probes | ~300 per probe [3] | >1 kHz [3] | No [3] | Yes [3] |
Experimental Protocols for High-Resolution Neural Recording
Large-Scale Electrophysiology with Neuropixels
The International Brain Laboratory collaborative study established a standardized protocol for brain-wide neural activity mapping in awake, behaving mice [6]. This methodology enables simultaneous recording from hundreds of brain regions with single-neuron resolution.
Experimental Workflow:
- Animal Preparation: 139 mice (94 male, 45 female) trained on a visual decision-making task with sensory, motor, and cognitive components
- Probe Implementation: 699 Neuropixels probes implanted following a standardized grid covering the left forebrain/midbrain and right hindbrain/cerebellum
- Signal Acquisition: Simultaneous recordings from multiple brain regions using 2-probe insertions per session
- Spike Sorting: Kilosort algorithm with custom additions applied to raw data
- Anatomical Registration: Probe tracks reconstructed using serial-section two-photon microscopy with neuron assignment to Allen Common Coordinate Framework regions
- Quality Control: Stringent metrics applied to identify 75,708 well-isolated neurons from 621,733 total units
This protocol yielded unprecedented spatial coverage (279 brain areas) while maintaining temporal resolution sufficient to capture single action potentials (>1 kHz), enabling analysis of neural correlates across sensory, decision, and motor processing stages [6].
Advanced Non-Invasive Neural Signal Detection
Johns Hopkins APL developed a novel protocol for detecting neural activity through the scalp using digital holographic imaging (DHI), representing a potential advancement for non-invasive spatial resolution [4].
Experimental Workflow:
- System Configuration: Digital holographic imaging system with nanometer-scale sensitivity
- Signal Targeting: Focus on neural tissue deformation (tens of nanometers in height) as novel signal source
- Illumination: Active laser illumination of neural tissue with recording of scattered light on specialized camera
- Clutter Mitigation: Signal processing to distinguish neural deformations from physiological noise (blood flow, heart rate, respiration)
- Validation: Extensive fundamental tests conducted over several years to confirm correlation with neural firing
This methodology aims to overcome the spatial resolution limitations of traditional non-invasive approaches like EEG by detecting mechanical rather than electrical manifestations of neural activity [4].
Mutual Information Analysis for ECoG Speech Decoding
A refined analytical protocol for electrocorticography (ECoG) demonstrates how computational methods can enhance information extraction from existing recording technologies [7].
Experimental Workflow:
- Data Acquisition: ECoG recordings during continuous speech production from participants undergoing intracranial monitoring for epilepsy
- Signal Processing: Comparison of traditional correlation coefficient (CC) analysis versus mutual information (MI) measure
- Masking Implementation: Exclusion of silence periods to refine detection of speech-related neural activity
- Temporal Mapping: Identification of neural activation patterns relative to speech onset
- Validation: Assessment of spatiotemporal activation patterns against known speech neuroanatomy
This protocol revealed that masked MI analysis detected earlier prefrontal and premotor activations (~440 ms before speech onset) with sharper anatomical coherence compared to traditional methods, improving spatial localization within the constraints of ECoG electrode spacing [7].
Signaling Pathways and Experimental Workflows
Neural Recording Experimental Pipeline
Neural Signal Detection Pathways
Research Reagent Solutions Toolkit
Table 3: Essential Research Tools for Neural Recording Experiments
| Tool/Technology | Function/Purpose | Example Implementation |
|---|---|---|
| Neuropixels Probes | High-density electrophysiology | NP Ultra: 6μm site spacing for increased neuronal yield [2] |
| Genetically Encoded Calcium Indicators (GECIs) | Optical recording of neural activity via calcium dynamics | Cell type-specific expression for longitudinal recordings [3] |
| Genetically Encoded Voltage Indicators (GEVIs) | Direct optical recording of membrane potential | More direct neural activity measurement vs. calcium imaging [3] |
| Kilosort | Spike sorting algorithm | Identification of individual neurons from raw extracellular data [6] |
| Allen Common Coordinate Framework (CCF) | Standardized brain atlas registration | Anatomical localization of recorded neurons [6] |
| Digital Holographic Imaging System | Non-invasive detection of neural tissue deformation | nanometer-scale detection through scalp [4] |
| Mutual Information Analysis | Nonlinear neural signal analysis | Enhanced speech decoding from ECoG signals [7] |
Discussion and Future Directions
The fundamental trade-off between spatial and temporal resolution continues to define the application landscape of neural recording technologies. Invasive methods maintain superiority for studying microcircuit dynamics with single-neuron and millisecond precision, as evidenced by brain-wide Neuropixels recordings capturing 621,733 neurons across 279 brain areas [6]. Non-invasive approaches are rapidly evolving, with innovations like digital holographic imaging potentially offering new pathways to overcome current spatial limitations [4].
Future progress will likely emerge from several complementary approaches: continued miniaturization and density scaling of electrode arrays [2] [8], development of faster and more sensitive molecular sensors [3], advanced signal processing techniques that extract more information from existing data streams [7] [8], and novel physics-based approaches to non-invasive detection [4]. These developments will progressively blur the distinctions between invasive and non-invasive paradigms, potentially enabling unprecedented access to brain dynamics across spatial and temporal scales for both basic neuroscience and clinical applications.
Brain function manifests across multiple spatiotemporal scales, producing distinct but interconnected physiological signals. Understanding the biophysical origins and recording capabilities of these signals—from the rapid, single-neuron action potentials to the slow, metabolic hemodynamic responses—is fundamental to neuroscience research and its clinical applications. This technical guide details the core principles of these signals, with a specific focus on the implications of their measurement for the spatial and temporal resolution of invasive versus non-invasive neural recordings. A precise grasp of these relationships is critical for selecting appropriate methodologies in basic research and drug development.
Core Biophysical Signals of Neural Activity
Action Potentials (APs)
Action potentials are the all-or-none electrical impulses conducted along a neuron's axon. They are generated by the rapid, voltage-dependent flux of ions (primarily Na⁺ and K⁺) across the neuronal membrane. APs represent the primary output of neurons and are the fundamental unit of fast communication in the nervous system. Their duration is typically on the order of 1-2 milliseconds, providing a very high temporal signature of neural discharge [3].
- Invasive Recording: The gold standard for AP recording is the patch clamp technique, which involves forming a tight seal with a cell's membrane using a glass micropipette to obtain intracellular recordings. While highly accurate, it is invasive, low-throughput, and leads to post-recording cell death [9]. For extracellular recording, microelectrode arrays (MEAs) can detect APs (spikes) from single neurons (single-unit activity, SUA) or small groups (multi-unit activity, MUA) within a radius of approximately ~200 μm from the electrode tip [10] [11].
- Non-Invasive Recording: True non-invasive recording of APs with patch-clamp quality from outside the cell membrane has been a major challenge. Recent advances in electrolyte-gated organic field-effect transistors (EGOFETs) show promise, leveraging a high-resistance cell/transistor seal to transduce APs from cardiomyocytes without poration, achieving signals with high fidelity to patch-clamp recordings [9]. Conventional non-invasive tools like EEG are incapable of resolving single APs.
Local Field Potentials (LFPs)
Local Field Potentials represent the ensemble electrical activity from a local population of neurons. They are recorded extracellularly and obtained by low-pass filtering the raw signal (typically with a cutoff of ~100-300 Hz) [10] [12]. Unlike APs, LFPs primarily reflect synaptic and dendritic processes, specifically the summed postsynaptic currents (both excitatory and inhibitory) from a large number of neurons [10] [13] [12]. The spatial reach of an LFP signal is estimated to be one to several millimeters around the recording electrode, capturing activity from tens of thousands to millions of neurons [10] [12].
A key concept is the relationship between LFPs and spiking activity. Research has shown that a linear filter operation on the spiking activity of one or a few neurons can estimate a significant fraction of the LFP time course, suggesting a predictable relationship between local output and circuit-level properties [10]. Furthermore, LFPs have been demonstrated to be a better predictor of the hemodynamic BOLD signal used in fMRI than multi-unit spiking activity, linking electrical circuit activity to metabolic responses [12].
Hemodynamic Responses
Hemodynamic responses are the secondary, metabolic consequences of neural activity. The primary electrical activity of neurons triggers a complex cascade of events that increases local cerebral blood flow, blood volume, and oxygen consumption. This neurovascular coupling is the basis for functional Magnetic Resonance Imaging (fMRI), which typically measures the Blood-Oxygen-Level-Dependent (BOLD) signal [13]. These metabolic changes unfold over a much slower time scale, with a delay of 2-6 seconds post-stimulus and a duration of over 10 seconds, resulting in very low temporal resolution compared to electrical signals [13].
Table 1: Characteristics of Core Neural Signals
| Signal | Biophysical Origin | Temporal Resolution | Spatial Resolution (Typical Measurement) |
|---|---|---|---|
| Action Potential (AP) | Voltage-gated ion channels; neuronal output [3] | ~1-2 ms [3] | Single neuron (Invasive: MEA) [11] |
| Local Field Potential (LFP) | Synaptic & dendritic postsynaptic currents; local network input/processing [10] [12] | ~200 ms for significant estimation from spikes [10] | ~1-3 mm (Invasive: depth probes) [10] [12] |
| Hemodynamic Response | Neurovascular coupling; metabolic demand [13] | ~Seconds (e.g., BOLD signal) [13] | ~1-5 mm (Non-invasive: fMRI) [13] |
Spatial and Temporal Resolution in Recording Technologies
The choice of recording technology imposes fundamental constraints on the observable dimensions of neural activity. The trade-off between spatial and temporal resolution is a central theme in systems neuroscience.
Invasive Recording Modalities
Invasive techniques involve the surgical implantation of electrodes directly into or onto brain tissue, granting access to high-frequency signals like APs and LFPs with a high signal-to-noise ratio (SNR) [11].
- Microelectrode Arrays (MEAs): These are typically implanted in the gray matter and can record action potentials (spikes) and LFPs. They offer the highest spatial and temporal resolution for invasive electrophysiology, allowing the recording of hundreds of neurons simultaneously with sub-millisecond temporal precision [3] [11].
- Electrocorticography (ECoG) and Stereo-EEG (sEEG): ECoG electrodes are placed on the surface of the cortex (subdural or epidural), while sEEG electrodes are depth probes inserted into brain tissue. Both record LFPs from the cortical surface or deeper structures with a higher spatial resolution and SNR than scalp EEG, but they cannot typically resolve single-unit activity [11].
- Deep Brain Stimulation (DBS) Electrodes: While primarily used for therapeutic stimulation, these macro-electrodes can also record LFPs from deep brain structures, such as the subthalamic nucleus, providing insights into pathological network oscillations in disorders like Parkinson's disease [12] [11].
Non-Invasive Recording Modalities
Non-invasive techniques record signals from outside the skull, ensuring safety and ease of use but at the cost of signal specificity and resolution due to the blurring effects of the skull, scalp, and other tissues [13] [14].
- Electroencephalography (EEG): EEG measures the electrical potential on the scalp generated by the synchronized postsynaptic currents of cortical pyramidal neurons. Its temporal resolution is excellent (millisecond range), but its spatial resolution is poor (centimeters) because of volume conduction, where signals spread and blur through the skull and scalp [13] [15] [14].
- Magnetoencephalography (MEG): MEG detects the minute magnetic fields produced by intracellular neuronal currents. It shares a similar temporal resolution with EEG but offers better spatial resolution, as magnetic fields are less distorted by the skull and scalp [13].
- Functional Magnetic Resonance Imaging (fMRI): fMRI measures the hemodynamic response (BOLD signal) indirectly. It provides high spatial resolution (millimeters) but very low temporal resolution (seconds) due to the slow nature of the blood-flow response [13].
- Emerging Optical Techniques: Digital holographic imaging (DHI) is an emerging non-invasive method that detects neural tissue deformations at the nanometer scale occurring during neural activity. This represents a novel signal source with the potential for high-resolution non-invasive recording, though it is still in the research and development phase [4].
Table 2: Comparison of Neural Recording Technologies
| Technology | Invasiveness | Typical Signals | Temporal Resolution | Spatial Resolution |
|---|---|---|---|---|
| Microelectrode Array (MEA) | Invasive | SUA, MUA, LFP | >1 kHz [3] | ~100s of neurons [11] |
| ECoG/sEEG | Invasive | LFP | >1 kHz | ~1 cm (surface/deep structures) [11] |
| Scalp EEG | Non-invasive | Scalp potentials | ~1 ms [14] | ~1-10 cm [13] |
| MEG | Non-invasive | Magnetic fields | ~1 ms [13] | ~5-10 mm [13] |
| fMRI | Non-invasive | BOLD (hemodynamic) | ~1-3 seconds [13] | ~1-5 mm [13] |
| Digital Holographic Imaging | Non-invasive | Tissue deformation | Under Investigation [4] | Under Investigation [4] |
Experimental Protocols for Key Investigations
Protocol 1: Linear Estimation of LFPs from Spiking Activity
This protocol, based on the work of Rasch et al. (2009), details how to estimate LFP time courses from simultaneously recorded spike trains, illustrating the functional link between neuronal output and local circuit dynamics [10].
- Electrophysiological Recording: Perform simultaneous extracellular recordings in the region of interest (e.g., primary visual cortex) using a matrix of electrodes. Record both the wideband signal and spike times.
- Signal Preprocessing:
- LFP Extraction: Low-pass filter the raw wideband signal with a cutoff frequency of ~220 Hz and resample at 500 Hz to obtain the LFP [10].
- Spike Train Generation: High-pass filter the raw signal (e.g., cutoff 500 Hz) and apply a threshold detection (e.g., 5 SDs of the noise) to extract spike times. The spike train, ( x(t) ), is generated from these times with the mean firing rate subtracted.
- Wiener-Kolmogorov Filter Estimation:
- Divide the data into training and test segments.
- Using the training segment, compute the Fourier transform of the cross-correlation between the LFP and spike train, ( P{Lx}(f) ), and the Fourier transform of the spike train autocorrelation, ( P{xx}(f) ).
- Calculate the optimal linear filter in the frequency domain: ( H(f) = P{Lx}(f) / (P{xx}(f) + \theta) ), where ( \theta ) is a small regularization constant to prevent division by zero [10].
- LFP Estimation and Validation:
- Convolve the spike train from the test segment, ( x{test}(t) ), with the derived filter, ( h(t) ) (the inverse Fourier transform of ( H(f) )), to produce the linear estimate ( L{est}(t) ).
- Quantify the estimation accuracy by computing the mean squared error between the estimated LFP, ( L_{est}(t) ), and the actual recorded LFP, ( L(t) ), in the test segment.
Diagram 1: LFP from Spikes Estimation Workflow.
Protocol 2: Non-Invasive AP Recording with EGOFETs
This protocol outlines the use of inkjet-printed electrolyte-gated organic field-effect transistors (EGOFETs) for non-invasive, high-fidelity AP recording, a promising alternative to patch clamping [9].
- Device Fabrication: Inkjet-print a thin film (~30 nm) of the organic semiconducting polymer P3HT to form the FET channel. Characterize the film for smooth topography (r.m.s. roughness ~1 nm) to facilitate a tight cell seal.
- Cell Culture: Seed a monolayer of human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) onto the fibronectin-coated EGOFET device. The culture medium acts as the electrolyte for the transistor.
- Electrical Characterization:
- Obtain the transfer characteristic curve (( I{DS} ) vs ( V{GS} )) of the EGOFET with the cell monolayer present.
- Compute the transconductance (( g_m )) to identify the optimal bias point for recording.
- AP Recording:
- Bias the transistor in its full accumulation mode (e.g., ( V{GS} = -0.8 V ), ( V{DS} = -0.5 V )), where transconductance is high.
- Record the modulated drain-source current (( \Delta I_{DS}(t) )) over time. A successful recording will show a waveform with a characteristic cardiac AP morphology and a high signal-to-noise ratio (e.g., ~60 dB).
- Validation: Benchmark the EGOFET recordings against simultaneous or matched-batch recordings from patch clamp (for AP morphology) and MEAs (for field potential morphology) to validate signal quality and information content.
Research Reagent and Material Solutions
The following table details key materials and reagents essential for the experiments described in this guide.
Table 3: Essential Research Reagents and Materials
| Item | Function / Description | Example Application |
|---|---|---|
| Microelectrode Arrays (MEAs) | Grids of micro-scale electrodes for simultaneous recording from multiple neurons. | Invasive recording of SUA, MUA, and LFP in animal models or ex vivo preparations [3] [11]. |
| ECoG / sEEG Electrodes | Electrodes placed on the cortical surface or within deep brain structures to record LFPs. | Pre-surgical epilepsy monitoring in humans; research on network oscillations [11] [16]. |
| High-Density EEG (hd-EEG) Systems | Scalp electrode nets with 64+ channels for improved spatial sampling. | Non-invasive brain mapping and electrophysiological source imaging (ESI) [13] [15]. |
| Genetically Encoded Calcium Indicators (GECIs) | Fluorescent proteins that change intensity upon binding calcium ions, acting as a proxy for neural activity. | Wide-field or two-photon calcium imaging to monitor population activity in genetically targeted cell types [3]. |
| P3HT-based EGOFETs | Inkjet-printed polymer transistors that transduce membrane potential changes via a high-resistance cell/transistor seal. | Non-invasive recording of action potentials with patch-clamp-like quality from cultured cells (e.g., hiPSC-CMs) [9]. |
| Wiener-Kolmogorov Filter | An optimal linear filter used to estimate a signal (LFP) from another related signal (spike train). | Quantifying the relationship between spiking activity and local field potentials in electrophysiological data [10]. |
Synthesis and Future Perspectives
The relationship between the biophysical origin of neural signals and the capabilities of our recording technologies defines the current landscape of neuroscience investigation. Invasive methods provide unparalleled access to the fast, microscopic world of action potentials and localized circuit dynamics (LFPs), but their clinical application is limited. Non-invasive methods, particularly EEG and fMRI, offer safe and scalable platforms for human research and clinical diagnostics, but they infer underlying neural activity from signals that are either spatially blurred (EEG) or temporally slow (fMRI) [13] [11] [14].
Future progress hinges on technological convergence. The combination of simultaneous invasive and non-invasive recordings (e.g., iEEG with EEG/MEG) in human patients acts as a "Rosetta stone," providing ground truth to validate and enhance the resolution of non-invasive source imaging techniques [16]. Furthermore, the development of closed-loop, bidirectional BCIs that both decode neural signals and encode information via electrical stimulation (ICMS, DBS) represents a powerful frontier for therapeutic intervention, requiring a deep integration of the principles outlined in this guide [11]. Finally, emerging technologies like digital holographic imaging and novel organic electronics promise to open new windows into brain function by measuring previously untapped signal sources, potentially reshaping the existing trade-offs between invasiveness and resolution [4] [9].
Diagram 2: Neural Signal Hierarchy & Measurement.
The fidelity of recorded neural signals is fundamentally constrained by the biological materials through which they must pass before reaching a sensor. In the context of invasive versus non-invasive neural recording technologies, understanding these signal degradation pathways is crucial for selecting appropriate methodologies and interpreting recorded data accurately. The scalp, skull, and various tissue layers act as successive filters that progressively degrade both the spatial and temporal resolution of underlying neural activity [17] [18]. This technical guide examines the biophysical basis of these filtering effects, quantifies their impact on different recording modalities, and explores methodological approaches for mitigating signal degradation in neural recording applications across research and clinical domains.
The electrical signals generated by cortical neurons undergo substantial transformation as they propagate through volume conductors with differing electrical properties. These filtering effects pose particular challenges for non-invasive techniques like electroencephalography (EEG), where signals must traverse the cerebrospinal fluid, skull, scalp, and skin before reaching recording electrodes [17] [19]. In contrast, invasive methods such as electrocorticography (ECoG) and intracortical microelectrodes bypass some of these barriers, placing sensors in closer proximity to neural sources and consequently capturing higher-frequency components with greater spatial precision [17] [18].
Biophysical Basis of Neural Signal Degradation
Fundamental Signal Degradation Mechanisms
The passage of neural signals through biological tissues involves three primary degradation mechanisms: distance-dependent attenuation, spatial filtering, and frequency-dependent filtering. Electric fields produced by neurons decay exponentially with distance from their source, with the number of simultaneously active neurons required for detection increasing dramatically with sensor distance [17]. For scalp EEG, this means that small neuronal clusters are often undetectable or recorded at significantly lower signal-to-noise ratios compared to invasive methods [17].
The spatial filtering properties of biological tissues arise from their resistive and capacitive characteristics. The skull represents a particularly significant barrier due to its low electrical conductivity compared to other intracranial tissues [19]. This conductivity mismatch causes both signal attenuation and spatial blurring, as current lines spread through the resistive skull layer before reaching scalp electrodes. The result is that EEG signals represent spatially averaged activity over approximately 1-3 cm² of cortical territory [18].
Table 1: Electrical Properties of Biological Tissues in Neural Signal Propagation
| Tissue Type | Relative Conductivity | Impact on Signal Propagation | Primary Filtering Effect |
|---|---|---|---|
| Cortical Gray Matter | Moderate | Source of neural signals | N/A |
| Cerebrospinal Fluid | High | Current spreading | Spatial low-pass filtering |
| Skull | Low | Significant signal attenuation | Strong spatial low-pass filtering |
| Scalp/Skin | Moderate | Additional signal attenuation | Mild spatial low-pass filtering |
Tissue-Specific Filtering Characteristics
Different tissue layers contribute uniquely to the overall filtering of neural signals. The skull possesses particularly low electrical conductivity compared to other intracranial tissues, creating a significant conductivity mismatch that dissipates and spreads neural currents [19]. This property makes the skull the primary contributor to spatial blurring in scalp EEG recordings. The thickness and composition of both compact and spongy bone layers further influence the extent of signal degradation, with variations across individuals and skull regions adding to the complexity of accurate source localization [20].
The scalp and skin layers introduce additional signal degradation through their resistive properties and the presence of non-neural bioelectric signals from muscle activity, eye movements, and other sources [21]. These tissues act as secondary spatial filters and represent a significant source of physiological noise that further complicates the detection of underlying neural activity in non-invasive recordings.
Quantitative Comparison of Recording Modalities
The cumulative effect of signal degradation pathways varies significantly across different neural recording approaches. The distance between recording electrodes and target neural populations fundamentally determines both the spatial resolution and frequency content of acquired signals [18].
Table 2: Signal Characteristics Across Neural Recording Modalities
| Recording Method | Spatial Resolution | Typical Signal Amplitude | Frequency Range | Primary Applications |
|---|---|---|---|---|
| Scalp EEG | 1-3 cm | ~100 μV | 0.5-100 Hz | Clinical monitoring, cognitive studies |
| ECoG (Subdural) | 0.5-5 mm | 50-100 μV | 0-200 Hz | Epilepsy monitoring, BCIs |
| Intracortical Microelectrodes | 200 μm | 50-500 μV | 0-7,000 Hz | Single-unit recording, neuroprosthetics |
| Endovascular Electrodes | 1-2.4 mm | Similar to ECoG | 0-200 Hz | Minimally invasive monitoring |
Invasive recordings provide access to a broader frequency spectrum, including high-frequency components (up to several kHz) that are typically attenuated below noise levels in non-invasive approaches [17]. The spatial resolution of invasive methods is substantially higher, with intracortical microelectrodes capable of resolving single-unit activity from individual neurons, while scalp EEG records averaged activity over large neuronal populations (1-3 cm diameter regions) [18].
The signal degradation pathways directly impact the information content available from each recording modality. Non-invasive EEG primarily captures synchronized post-synaptic potentials from pyramidal neurons arranged in parallel orientation [19]. These signals are dominated by low-frequency components (<90 Hz for dry EEG electrodes) due to the low-pass filtering properties of intervening tissues [17]. In contrast, invasive methods can detect action potentials, local field potentials, and higher-frequency oscillations that provide more detailed information about local neural processing [17].
Experimental Methodologies for Studying Signal Degradation
Simultaneous Recording Paradigms
Research into signal degradation pathways has been advanced through simultaneous recording setups that capture neural activity at multiple spatial scales concurrently. The combination of scalp EEG with intracerebral electrodes in epileptic patients provides particularly valuable insights, as it allows direct comparison of how the same neural events appear at different recording distances [19] [16].
These simultaneous recording paradigms have demonstrated that the cortical area involved in generating detectable scalp EEG signals is rather large, with estimates suggesting several square centimeters of synchronously active cortex are required to produce measurable scalp potentials [19]. The relative location of neural sources with respect to recording electrodes strongly influences signal properties, with source geometry representing a critical parameter in signal transmission to the scalp [19].
Figure 1: Neural Signal Degradation Pathways from Cortex to Recording Electrodes
Modeling Approaches
Computational modeling represents another important methodology for investigating signal degradation pathways. Spatio-temporal extended source models combine realistic anatomical descriptions with physiologically relevant temporal dynamics to simulate how neural activity propagates to recording electrodes [19]. These models incorporate:
- Anatomically accurate head models derived from MRI segmentation
- Distributed dipole sources representing populations of pyramidal neurons
- Electrical properties of different tissue types
- Electrode-specific forward models predicting recorded signals
Such modeling approaches have been particularly valuable for understanding the relationship between synchronization degree in neuronal populations and the resulting EEG signals. Interestingly, intracerebral EEG can reflect epileptic activities corresponding to weak synchronization between neuronal populations, while scalp EEG requires much stronger synchronization to detect the same events [19].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Methods for Neural Signal Degradation Research
| Research Tool | Function/Purpose | Technical Specifications |
|---|---|---|
| High-Density EEG Systems | Improves spatial resolution through dense electrode arrays | 128-256 electrodes, precise anatomical localization |
| μECoG Arrays | Enables high-resolution cortical surface recording | 25μm thickness, 150-250μm electrode diameter, transparent substrates |
| Computational Modeling Platforms | Simulates signal propagation through head tissues | Finite-element analysis, realistic head models, dipole source modeling |
| Thinned Skull Preparation | Intermediate approach between invasive and non-invasive | ~100-200μm skull thickness, maintains biological barrier with reduced signal degradation |
| Simultaneous Recording Setups | Direct comparison of signals across different recording distances | Synchronized scalp and intracranial EEG systems |
Advanced Signal Processing Approaches
Modern signal processing techniques play a crucial role in mitigating the effects of signal degradation in neural recordings. For non-invasive EEG, methods such as Independent Component Analysis (ICA) help separate neural signals from contaminating artifacts arising from eye movements, muscle activity, and other non-neural sources [21]. Source localization algorithms including beamforming and dipole modeling attempt to reconstruct the origins of neural activity despite the spatial filtering effects of intervening tissues [21].
The combination of EEG with other neuroimaging modalities such as functional MRI (fMRI) represents another strategy for overcoming the limitations of individual techniques. This multimodal approach leverages the excellent temporal resolution of EEG (milliseconds) with the superior spatial resolution of fMRI (millimeters), providing a more comprehensive picture of neural dynamics [21]. However, the integration of these modalities requires careful consideration of their different physiological origins and spatiotemporal characteristics.
Figure 2: Signal Processing Workflow for Mitigating Neural Signal Degradation
Emerging Technologies and Future Directions
Recent technological developments offer promising avenues for overcoming traditional limitations in neural signal recording. Minimally invasive approaches such as endovascular electrodes represent a compromise between the fidelity of fully invasive methods and the safety of non-invasive approaches [18]. These devices can be delivered through standard catheterization procedures into blood vessels adjacent to target brain regions, recording local field potentials with resolution comparable to ECoG but without requiring open brain surgery [18].
Advanced electrode materials and designs are also contributing to improved signal quality. Transparent μECoG grids enable simultaneous electrophysiological recording and optical imaging or optogenetic manipulation through thinned skull preparations [20]. These arrays maintain stable impedances for extended periods (at least one month in chronic studies) and allow spatially distinct electrophysiological recordings with electrode separations of 500-750μm [20].
The growing field of brain-to-brain synchrony research using hyperscanning techniques further demonstrates the importance of understanding signal degradation pathways. Studies measuring neural synchronization between interacting individuals rely on sophisticated signal processing to distinguish true neural coupling from spurious correlations introduced by shared environments or task structures [22]. The most applied approaches for determining neural connectivity in these studies include phase-locking value (PLV) in EEG research and wavelet transform coherence (WTC) in fNIRS studies [22].
The skull, scalp, and tissue properties collectively form a complex filtering system that significantly shapes the neural information accessible to recording technologies. These signal degradation pathways impose fundamental constraints on the spatial and temporal resolution of all neural recording methods, with particularly pronounced effects on non-invasive approaches like EEG. Understanding these biological filters is essential for selecting appropriate methodologies, interpreting experimental results, and developing new technologies that overcome current limitations.
The trade-offs between signal fidelity and invasiveness continue to drive innovation in neural recording technologies, with emerging approaches seeking optimal balances for specific applications. Future advances will likely combine improved electrode designs, sophisticated computational modeling, and multimodal integration to extract increasingly precise information about brain function from both invasive and non-invasive recordings. For researchers and clinicians working with neural data, a thorough understanding of signal degradation pathways remains indispensable for drawing meaningful conclusions about the neural processes underlying recorded signals.
The interpretation of brain activity through electrical signals is fundamentally shaped by the recording methodology. A central tenet in neuroscience is that non-invasive and invasive techniques provide different windows into neural computation, largely due to their differential sensitivity to contributions from various neuronal populations. Non-invasive recordings, such as electroencephalography (EEG), predominantly capture summed activity from large, synchronously active populations with specific geometrical arrangements—primarily cortical pyramidal cells [17]. In contrast, invasive recordings, including intracortical electrodes, can access a more diverse neural repertoire, including action potentials (APs) and local field potentials (LFPs) from various neuron types such as pyramidal cells and interneurons, offering a richer, more localized picture of circuit dynamics [17] [23]. This whitepaper explores the biophysical and technical foundations of this dichotomy, its implications for signal interpretation, and details the experimental protocols that enable these insights.
Fundamental Principles of Signal Generation and Acquisition
The Biophysical Basis of Extracellular Signals
The electrical signals measured both on the scalp and within the brain originate from the same fundamental source: extracellular currents generated by neuronal activity. Specifically, the primary contributors are post-synaptic currents, which create dipoles as ions flow across neuronal membranes [17]. However, the path these signals take to the recording electrode and the medium they traverse drastically alter what is measured.
- Pyramidal Cell Dominance in EEG: The electric fields produced by individual neurons decay exponentially with distance. For a signal to be detectable on the scalp, the electric fields from a vast number of neurons must superimpose coherently. This requires not only synchronous activity but also a specific cellular morphology. Pyramidal cells, with their long, parallelly aligned apical dendrites perpendicular to the cortical surface, constitute an ideal geometrical arrangement for the summation of their dipoles. Their abundance in the cortex further ensures that their activity dominates the EEG signal. In contrast, the fields from interneurons, which often have stellate, non-polarized morphologies, tend to cancel out over distance and are therefore undetectable at the scalp [17].
- Diverse Sources in Invasive Recordings: Intracortical recordings, such as those from multi-electrode arrays (MEAs), are placed directly in or near the neural tissue. This proximity bypasses the signal-attenuating effects of the skull and scalp. Consequently, they can record a broader superposition of electrophysiological processes, including those underlying EEG, plus contributions from local interneurons, axonal action potentials, and other sources [17]. Invasive Local Field Potentials (LFPs) reflect input to, local processing, and output of cortical areas [17].
Technical Limitations: Spatial and Temporal Resolution
The physical characteristics of signal propagation impose intrinsic limitations on what each recording modality can resolve.
- Spatial Filtering: Tissue between the brain and scalp electrodes (cerebrospinal fluid, skull, scalp) acts as a volume conductor, causing significant spatial blurring. This "low-pass filter" effect means that EEG cannot resolve the activity of small, discrete neuronal clusters. The spatial resolution of EEG is on the order of centimeters, making it impossible to distinguish two spatially close neuronal sources activated sequentially with small temporal separation [24]. Invasive signals, being recorded directly from the source, suffer minimal spatial distortion and can resolve activity at micron-scale resolution, allowing for the identification of individual neurons and their spatial relationships [23].
- Temporal Filtering: The same tissues that cause spatial blurring also act as a temporal low-pass filter, preferentially attenuating high-frequency signals. With some exceptions, non-invasive signals are generally limited to analyzing activity below approximately 90 Hz, where they are buried in background noise [17]. Invasive recordings, conversely, can capture information-rich signals up to several kHz, encompassing the full spectrum of LFP oscillations and individual action potentials [17].
Table 1: Quantitative Comparison of Neural Recording Modalities
| Feature | Non-Invasive (EEG) | Invasive (LFP) | Invasive (Spikes) |
|---|---|---|---|
| Dominant Neural Sources | Pyramidal neurons | Mixed populations (Pyramidal & Interneurons) | Individual, identifiable neurons |
| Spatial Resolution | Centimeters (cm) [24] | Micrometers to millimeters (µm-mm) | Single neuron (µm) |
| Temporal Resolution | Millisecond (ms) scale, but limited to low frequencies (<~90 Hz) [17] | Millisecond (ms) scale, up to several kHz [17] | Sub-millisecond (ms) scale |
| Key Limitation | Spatial distortion & blurring; cannot access high-frequency data [24] | Cannot track individual spikes over long periods | Limited spatial coverage; surgical intervention required |
Experimental Evidence for Distinct Population Contributions
Cell-Type-Specific Spatial Signatures in Invasive Recordings
Recent studies using high-density electrode arrays have provided direct experimental evidence that different neuron types produce distinct spatial signatures in their extracellular potentials, which can be used for classification.
In a key study, researchers combined high-density neurophysiological recordings in freely moving mice with optogenetic tagging of parvalbumin-positive (PV) interneurons [23]. This provided a ground-truth dataset of labeled pyramidal cells (PYR) and PV cells. The investigation went beyond traditional single-channel waveform analysis by applying an event-based delta-transformation to the multichannel waveforms. This transformation removed all single-channel waveform information, conserving only the purely spatial information—the distribution of the signal across the electrode array at a single point in time [23].
- Findings: Machine learning models trained solely on these spatial features successfully differentiated PYR from PV cells with high accuracy. The analysis revealed that the spatial distribution of the initial depolarization phase was the most contributory feature. PYR spikes exhibited higher spatial synchrony at the beginning of the extracellular spike compared to PV spikes, a finding consistent with the large, polarized structure of pyramidal neurons [23].
- Implication: This work demonstrates that the morphology, connectivity, and ion channel distribution of a neuron are imprinted on the spatial pattern of its extracellular electrical footprint. Invasive high-density recordings can capture these signatures, enabling cell-type-specific analysis without optical tagging, a capability completely absent in non-invasive methods.
Functional Diversity Among Pyramidal Cell Subtypes
Even within the broad category of pyramidal cells, invasive techniques and targeted imaging reveal significant functional diversity that is obscured in non-invasive signals.
Research using cell-type-specific widefield calcium imaging in mice has shown that different pyramidal neuron projection types, such as pyramidal tract (PT) and intratelencephalic (IT) neurons, drive functionally distinct, cortex-wide activity patterns during a decision-making task [25]. Dimensionality reduction and clustering analyses of cortex-wide activity revealed unique spatiotemporal components for each pyramidal cell type. Furthermore, optogenetic inactivation experiments demonstrated that in parietal cortex, PT neurons had the largest causal role in sensory perception, whereas in frontal cortex, all pyramidal types were required for accurate choices but showed distinct choice tuning [25]. This shows that invasive and cell-type-specific methods can uncover specialized subcircuits within the pyramidal cell population that contribute to different aspects of a cognitive process.
Experimental Protocols for Cell-Type-Specific Neural Recording
Protocol 1: Cell-Type Classification Using High-Density Extracellular Recordings
This protocol is adapted from the study that classified pyramidal and PV cells based on spatiotemporal waveforms [23].
1. Probe Implantation and Animal Preparation:
- Subjects: PV-Cre mice crossed with Ai32 reporter mice to enable optogenetic identification of PV cells.
- Probe: Implant a high-density, multi-shank silicon probe (e.g., 32 sites per shank) coupled with an optical fiber for photostimulation.
- Surgery: Under anesthesia, implant the probe in the target region (e.g., hippocampal CA1 or neocortex). Allow a recovery period of at least 48 hours.
2. Data Acquisition:
- Recording: Record wide-band neural signals (e.g., 1-5000 Hz) from the freely moving animal during spontaneous behavior and during photostimulation sessions.
- Photostimulation: Deliver light pulses (e.g., 50-70 ms, 35 µW) to optogenetically drive ChR2-expressing PV cells. This creates ground-truth labels for PV units.
- Spike Sorting: Extract spike waveforms and sort them into single units offline using automated algorithms (e.g., KlustaKwik) followed by manual curation. Apply quality criteria: amplitude >50 µV, L-ratio <0.05, isolation distance >20.
3. Feature Extraction and Classification:
- Single-Channel Features: For a baseline, extract standard waveform features (trough-to-peak duration, peak asymmetry, etc.) from the channel with the largest spike amplitude for each unit.
- Spatiotemporal Features: To isolate purely spatial information, apply an event-based delta-transformation. For each spike, subtract the mean waveform across all channels, leaving only the spatial distribution of the signal at each time point. From this, derive features like the spatial spread of the spike peak or trough.
- Model Training: Train a binary classifier (e.g., support vector machine or random forest) using a dataset of labeled PYR and PV units. Validate model performance using cross-validation.
Protocol 2: Multimodal Integration of EEG and Diffuse Optical Tomography (DOT)
This protocol addresses the limitations of single-modal non-invasive imaging by combining EEG's temporal resolution with DOT's spatial resolution to improve source localization [24].
1. Experimental Setup:
- System: Use a integrated, portable EEG-DOT system. A 32-channel EEG setup is a minimum, but higher densities (64-channel) improve results.
- Optode Placement: Arrange DOT source and detector optodes in a regular-density grid (e.g., source-detector distance of ~3 cm) over the region of interest, co-located with EEG electrodes.
- Head Model: Generate a forward model using a segmented brain atlas (e.g., ICBM152) with tetrahedral meshing to define the scalp, skull, CSF, and brain compartments.
2. Data Collection and Forward Modeling:
- Task: Have participants perform a task designed to activate sequential, spatially close sources (e.g., finger tapping).
- Recording: Acquire EEG and fNIRS/DOT data simultaneously.
- Leadfield Calculation: Calculate the leadfield matrix for EEG, defining how a current source at each brain location would project to each EEG electrode.
- Photon Diffusion Modeling: Calculate the forward model for DOT, defining how absorption changes in the brain volume would affect light intensity at each detector.
3. Joint Inverse Problem Solution:
- DOT Reconstruction: First, reconstruct the hemodynamic activation image from the fNIRS data by solving the DOT inverse problem.
- Spatial Prior for EEG: Use the DOT reconstruction as an empirical, spatially informed prior to constrain the highly ill-posed EEG inverse problem. This can be done within a Restricted Maximum Likelihood (ReML) framework.
- Validation: The combined EEG-DOT reconstruction should successfully resolve neuronal sources that are both spatially (e.g., 2-3 cm apart) and temporally (e.g., 50 ms separation) close, a feat not achievable by either modality alone [24].
Table 2: Key Research Reagent Solutions
| Reagent / Resource | Function and Application in Research |
|---|---|
| Genetically Encoded Calcium Indicators (GECIs, e.g., GCaMP6/7) | Enables cell-type-specific optical recording of calcium fluctuations as a proxy for neuronal activity in vivo, allowing longitudinal studies [3]. |
| Cre-Driver Mouse Lines (e.g., Fezf2-CreER, PlexinD1-CreER, PV-Cre) | Provides genetic access to specific neuronal populations (e.g., PT, IT, or PV neurons) for targeted expression of sensors or actuators [25]. |
| High-Density Multi-Electrode Arrays (e.g., Neuropixels) | Allows for large-scale, single-neuron resolution electrophysiology across multiple brain structures, capturing spatiotemporal spike waveforms [23]. |
| Optogenetic Actuators (e.g., Channelrhodopsin-2/ChR2) | Used for precise, millisecond-timescale control of specific neuron populations and for providing ground-truth labels during spike sorting [23]. |
| Modular Brain Signal Decoding Platform (e.g., py_neuromodulation) | An open-source software platform for standardized extraction of features (oscillatory power, waveform shape, coherence) from invasive brain signals for machine learning [26]. |
Visualizing Signaling Pathways and Workflows
Neural Signal Propagation from Cortex to Scalp
Diagram 1: Signal propagation from brain to scalp.
Joint EEG-DOT Source Reconstruction Workflow
Diagram 2: Joint reconstruction workflow.
The divergence in neuronal population contributions between non-invasive and invasive recordings is not an artifact but a direct consequence of fundamental biophysical principles. Non-invasive EEG provides a unique, macro-scale summary of coherent pyramidal cell activity, invaluable for studying large-scale brain dynamics and states. Invasive methods, by residing within the neural tissue, unlock the micro-scale diversity of the circuit, revealing the distinct roles of pyramidal subtypes and interneurons. This dichotomy necessitates a careful, methodology-aware interpretation of neural data. The future of systems neuroscience lies in leveraging the strengths of both approaches, potentially through multimodal integration [24] [16] and advanced computational models [27] [26], to bridge the gap between macroscopic brain signals and their microscopic, cell-type-specific generators.
Methodological Landscape: From Intracortical Arrays to Scalp-Based Systems
Brain-computer interfaces (BCIs) have evolved from early neurophysiological studies to sophisticated systems capable of converting neural activity into executable commands for external devices [14]. Within this domain, high-resolution invasive neural interfaces represent a critical technological frontier, offering unprecedented access to neural signals with exceptional spatial and temporal fidelity. These technologies are primarily distinguished from their non-invasive counterparts by their physical proximity to neural tissue, enabling them to bypass the signal-filtering effects of the skull and scalp [28]. This technical guide focuses on two principal high-resolution invasive approaches: intracortical microelectrode arrays and electrocorticography (ECoG), examining their fundamental operating principles, technical capabilities, experimental methodologies, and applications within neuroscience research and therapeutic development.
The fundamental advantage of invasive recordings stems from their access to rich neurophysiological information. While non-invasive electroencephalography (EEG) captures post-synaptic extracellular currents from large, synchronized populations of pyramidal neurons after significant signal attenuation and spatial blurring, invasive techniques can record a broader spectrum of signals closer to their source [28]. Intracortical microelectrode arrays provide the highest resolution, enabling detection of extracellular action potentials (APs) from individual neurons or small groups, as well as local field potentials (LFPs) reflecting integrated synaptic activity within a localized region [29] [28]. ECoG electrodes, positioned on the cortical surface (subdural or epidural), capture signals with higher spatial resolution and signal-to-noise ratio than EEG, including high-frequency broadband activity that is inaccessible non-invasively [29]. For researchers and drug development professionals, understanding the capabilities and limitations of these technologies is crucial for designing studies that require precise monitoring of neural circuit activity, assessing therapeutic efficacy, or developing next-generation neurotherapeutics.
Technical Foundations and Signal Characteristics
Intracortical Microelectrode Arrays
Intracortical microelectrode arrays are implanted directly into the gray matter, enabling recording and stimulation at the level of individual neurons. These systems are designed to sample neural activity at spatial and temporal scales sufficient to resolve action potentials (typically < 1 ms duration) and fine-scale local field potentials.
- Utah Array: A widely used commercial intracortical array consisting of 100 silicon-based microelectrodes arranged in a 10×10 grid. Each electrode tip records action potentials from one or a few nearby neurons, providing unparalleled access to the neural code [29].
- Neuropixels Probes: Representing the next generation of recording technology, these CMOS-based devices pack nearly 1,000 recording sites onto a single, slender shank. Their high channel count allows simultaneous recording from hundreds to thousands of individual neurons across multiple cortical layers and brain structures [29].
- High-Density Microelectrode Arrays (HD-MEAs): Advanced CMOS-based systems now feature dramatically increased electrode densities (>3,000 per mm²) and channel counts, with recent devices incorporating up to 236,880 electrodes with subcellular spatial resolution [30].
These intracortical devices face significant challenges related to biological integration. The mechanical mismatch between rigid implant materials and soft, pulsating brain tissue can cause chronic inflammation, glial scarring, and eventual signal degradation [29]. Emerging solutions focus on flexible, polymer-based substrates and "bio-integrative" electrodes designed to form more stable interfaces with neural tissue through anti-inflammatory coatings or injectable mesh electronics [29].
Electrocorticography (ECoG)
ECoG occupies an intermediate position between intracortical arrays and non-invasive EEG, offering higher signal quality than surface EEG without penetrating brain tissue. Conventional ECoG uses electrode grids placed surgically on the cortical surface, either below (subdural) or above (epidural) the dura mater.
- Standard ECoG Grids: Typically feature electrode diameters of 2-5 mm with inter-electrode spacing of 5-10 mm, providing coverage over larger cortical areas while capturing neural population signals with higher spatial resolution than EEG [29].
- Micro-Electrocorticography (µECoG): Recent advances have led to the development of high-density thin-film microelectrode arrays that significantly increase spatial resolution. State-of-the-art systems now feature 1,024-channel arrays with electrode diameters as small as 50 µm and inter-electrode pitches of 300-400 µm [31].
- Minimally Invasive Implantation: Innovative surgical techniques such as the "cranial micro-slit" approach allow implantation of high-density arrays through 500-900 μm wide skull incisions without full craniotomy, reducing surgical risk and improving translational potential [31].
µECoG represents a promising combination of minimal tissue damage and improved signal quality, capturing signals from the cortical surface at spatial resolutions approaching those of some intracortical methods [31]. ECoG signals are generally more stable over time compared to intracortical recordings, with less susceptibility to signal drift or the progressive degradation that can affect penetrating electrodes [29].
Comparative Technical Specifications
Table 1: Quantitative Comparison of Invasive Neural Recording Technologies
| Parameter | Intracortical Microelectrodes | ECoG/µECoG |
|---|---|---|
| Spatial Resolution | Single neurons (50-200 μm recording radius) [29] | Neural populations (50 μm - 5 mm, depending on electrode size) [31] [29] |
| Temporal Resolution | <1 ms (suitable for action potentials) [28] | <1 ms (suitable for high-frequency oscillations) [29] |
| Typical Electrode Density | 100 electrodes/16 mm² (Utah Array) to 1,000 electrodes/shaft (Neuropixels) [29] | 1,024 electrodes over several cm² (modern µECoG) [31] |
| Signal Types Captured | Action potentials (spikes), Local Field Potentials (LFPs) [28] | Mainly LFPs and cortical surface potentials, including high-frequency broadband [29] |
| Frequency Range | DC to >7,000 Hz [28] | DC to ~500 Hz (effectively; hardware may support higher) [29] [28] |
| Signal Attenuation by Tissue | Minimal | Moderate (filtered by meningeal layers) |
| Typical Implantation Duration | Months to years (with potential signal decline) [29] | Long-term (more stable signal profile) [31] |
| Surgical Invasiveness | High (penetrates parenchyma) | Moderate (surface implantation; minimally invasive approaches available) [31] |
Table 2: Signal Content and Information Capacity Comparison
| Signal Characteristic | Intracortical Arrays | ECoG | Non-Invasive EEG |
|---|---|---|---|
| Primary Neural Sources | Local neurons (spikes), integrated synaptic activity (LFP) [28] | Superficial cortical layers, synchronized populations [29] | Large synchronized pyramidal cell populations [28] |
| Dominant Signal Components | Multi-unit activity, LFP (all frequency bands) [28] | LFP, high-gamma activity, cortical slow potentials [29] | Alpha, beta rhythms; event-related potentials [14] |
| Access to High-Frequency Activity | Full spectrum (>500 Hz) [28] | Limited high-frequency content (effectively <200 Hz) [28] | Very limited (<80 Hz, severely attenuated) [28] |
| Information Transfer Rate Potential | Very high | High | Moderate to low [28] |
| Spatial Specificity | Very high (individual neurons) [29] | Moderate to high (neural populations) [31] | Low (smeared by volume conduction) [28] |
Experimental Design and Methodological Approaches
System Configuration and Implantation
The design of experiments using high-resolution invasive techniques requires careful consideration of surgical approach, array configuration, and data acquisition infrastructure.
For intracortical microelectrode arrays, implantation typically requires a craniotomy and penetration of the pial surface. The Utah Array is commonly implanted using a pneumatic inserter, with electrode lengths tailored to target specific cortical layers (e.g., 1.0-1.5 mm for motor cortex) [29]. The emerging Neuropixels probes are typically implanted using slow insertion techniques to minimize tissue damage, with their high channel count enabling sampling across multiple cortical layers and brain regions simultaneously [29]. Surgical planning for these procedures often incorporates pre-operative MRI and neuronavigation to target specific functional regions.
For ECoG, less invasive approaches are possible. Recent innovations include minimally invasive implantation of thin-film µECoG arrays through cranial micro-slit techniques using precision sagittal saw blades to create 500-900 μm wide openings, avoiding full craniotomy [31]. This approach has been demonstrated in both porcine models and human cadavers, with the entire surgical procedure requiring approximately 20 minutes per array [31]. Intraoperative fluoroscopy or computed tomographic image guidance, combined with neuroendoscopy, enables precise placement of these flexible arrays on the cortical surface.
System architecture for both approaches requires addressing the challenge of managing high-channel-count data streams. Modern systems incorporate miniaturized headstages containing electronics for analog-to-digital conversion and signal conditioning, which stream data to real-time processing systems [31]. Fully implantable, wireless systems are in development to reduce infection risk and improve usability [29].
Signal Processing and Neural Decoding
The massive data streams generated by high-resolution invasive interfaces necessitate sophisticated processing pipelines to extract behaviorally relevant signals.
Figure 1: Neural Signal Processing Workflow for high-resolution invasive brain-computer interfaces, showing the transformation from raw data to device commands through a modular processing pipeline.
A standardized framework for invasive brain signal decoding has been developed through platforms such as py_neuromodulation, which provides modularized feature estimation chains for oscillatory dynamics, waveform shape, interregional coherence, and other relevant features [26]. For movement decoding, researchers typically formulate the problem as a two-class classification task (rest versus movement) using fast Fourier transform features computed from streamed raw data in specific frequency bands [26].
Recent advances in decoding have incorporated connectomic approaches that account for individual variations in electrode localization. These methods use functional or structural connectivity fingerprints extracted from recording locations in normative space to identify optimal decoding networks that generalize across patients [26]. This approach addresses a critical limitation in clinical translation by reducing or eliminating the need for patient-specific training sessions.
For intracortical arrays, spike sorting represents an additional processing step to isolate single-unit activity from multi-electrode recordings. This typically involves bandpass filtering (300-3000 Hz), threshold detection, and clustering based on waveform features using algorithms such as K-means or Gaussian mixture models [29]. The high channel counts of modern Neuropixels probes enable a "cluster-free" approach to spike identification through template matching across many channels [29].
Multimodal Integration and Stimulation Capabilities
Modern high-resolution invasive interfaces increasingly incorporate multimodal capabilities, combining recording with stimulation and other measurement modalities.
Bidirectional interfaces enable both recording and stimulation through the same electrodes, creating closed-loop systems that can adapt in real-time to neural activity. For example, recent µECoG arrays incorporate both recording electrodes (50 μm) and larger stimulation electrodes (380 μm) on the same flexible substrate [31]. This enables focal neuromodulation at sub-millimeter scales while monitoring neural responses simultaneously.
Integration with brain connectomics represents another frontier, combining invasive neurophysiology with whole-brain structural and functional connectivity maps derived from diffusion tensor imaging and functional MRI [26]. This integration enables network-based decoding approaches that leverage knowledge of large-scale brain circuits to improve decoding performance and generalization across patients.
Additional multimodal combinations include interfaces that incorporate drug delivery channels, optical stimulation capabilities, or chemical sensing alongside electrophysiological recording [32]. These advanced systems require sophisticated material science approaches to create flexible, multifunctional neural interfaces that minimize tissue damage while maximizing functionality.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Materials for High-Resolution Invasive Neural Interfaces
| Item | Function/Application | Technical Specifications | Representative Examples/Alternatives |
|---|---|---|---|
| High-Density Microelectrode Arrays | Neural signal acquisition at cellular resolution | 236,880 electrodes on 5.51 × 5.91 mm² area; 33,840 simultaneous channels at 70 kHz [30] | CMOS-based HD-MEAs [30] |
| Thin-Film µECoG Arrays | Cortical surface recording with minimal invasiveness | 1,024 channels; 50 μm recording electrodes; 400 μm pitch; >91% yield [31] | Flexible polyimide-based arrays [31] |
| Neuropixels Probes | Large-scale single-neuron recording across brain regions | ~1,000 recording sites per shank; simultaneous recording from thousands of neurons [29] | Neuropixels 1.0 and 2.0 [29] |
| Utah Arrays | Intracortical recording for BCIs | 100 silicon microelectrodes in 10×10 grid; 1-1.5 mm electrode length [29] | Blackrock Microsystems Utah Array [29] |
| Flexible Polymer Substrates | Reduced tissue damage and improved biocompatibility | Polyimide or parylene-C substrates; conformable to cortical surface [29] | Various custom formulations [29] |
| Anti-inflammatory Coatings | Mitigation of foreign body response | Drug-eluting coatings (e.g., dexamethasone); surface modifications [29] | Biodegradable polymer coatings [29] |
| py_neuromodulation Platform | Standardized neural signal decoding | Modular feature extraction; connectomic decoding; machine learning integration [26] | Open-source Python platform [26] |
| Minimally Invasive Delivery Tools | Surgical implantation with reduced trauma | Cranial micro-slit technique: 500-900 μm wide incisions [31] | Precision sagittal saw blades; neuroendoscopes [31] |
Applications in Research and Therapeutic Development
High-resolution invasive neural interfaces have enabled significant advances across multiple domains of neuroscience research and therapeutic development.
Fundamental Neuroscience and Neural Decoding
In basic neuroscience, these technologies have enabled unprecedented investigation into the neural basis of behavior, revealing how information is encoded and processed across distributed neural circuits. Intracortical arrays have been particularly valuable for understanding motor control at the level of individual neurons and small populations, revealing how movement parameters such as direction, speed, and force are represented in motor and premotor cortices [28]. ECoG has provided insights into large-scale cortical dynamics underlying higher cognitive functions, with its broader coverage enabling investigation of distributed processing across multiple brain regions [29].
Movement decoding performance from invasive signals substantially exceeds what is possible with non-invasive approaches. Studies using ECoG have demonstrated movement detection rates of 0.88 ± 0.17 with balanced accuracy of 0.79 ± 0.13, even during concurrent deep brain stimulation [26]. Importantly, decoding performance has been shown to correlate with clinical measures, exhibiting negative correlations with Parkinson's disease symptom severity (Unified Parkinson's Disease Rating Scale Part III) [26].
Clinical Applications and Therapeutic Development
For drug development professionals and clinical researchers, high-resolution invasive interfaces offer powerful tools for assessing therapeutic efficacy and developing novel intervention strategies.
In neurological disorders, these technologies enable precise monitoring of pathological neural activity and response to interventions. For Parkinson's disease, invasive recordings have revealed characteristic beta-band oscillations in the subthalamic nucleus that can be modulated by dopaminergic medication and deep brain stimulation [26]. In epilepsy, ECoG grids are routinely used clinically to localize seizure foci prior to surgical resection, with high-density arrays providing improved spatial precision for identifying pathological networks [31].
The development of closed-loop neuromodulation systems represents one of the most promising therapeutic applications. These systems use real-time neural signals to guide therapeutic stimulation, enabling precise temporal targeting of interventions. For example, responsive neurostimulation systems for epilepsy deliver stimulation only when abnormal activity is detected, potentially improving efficacy while reducing side effects compared to continuous stimulation [26].
For pharmaceutical development, invasive interfaces provide sensitive measures of drug effects on neural circuit function. The high temporal resolution of these technologies enables detection of acute changes in neural activity following drug administration, while chronic implantation allows longitudinal assessment of treatment effects over relevant timeframes.
Emerging Directions and Future Prospects
The field of high-resolution invasive neural interfaces continues to evolve rapidly, with several emerging trends likely to shape future research and applications:
- Bio-integrative electrodes designed to form functional interfaces with neural tissue through surface coatings that release anti-inflammatory drugs, scaffolds that promote neuronal ingrowth, or injectable mesh electronics [29].
- Wireless, fully implantable systems that eliminate transcutaneous connectors, reducing infection risk and improving quality of life for chronic users [29].
- Connectomic decoding frameworks that leverage individual brain connectivity patterns to improve decoding performance and generalization across patients [26].
- Miniaturization of electrode features and increasing channel counts, with electrode densities now exceeding 3,000 per mm² [30].
- Multimodal integration combining electrophysiology with optical stimulation, drug delivery, and chemical sensing in unified platform technologies [32].
These advances promise to enhance both the scientific utility and clinical applicability of high-resolution invasive neural interfaces, potentially expanding their use beyond severe neurological conditions to broader therapeutic applications.
High-resolution invasive neural interfaces, particularly intracortical microelectrode arrays and electrocorticography, provide unparalleled access to neural signals with spatial and temporal fidelity far exceeding non-invasive alternatives. While each technology presents distinct trade-offs in terms of invasiveness, spatial resolution, and stability, both enable neuroscientists and drug development professionals to investigate neural function and dysfunction with unprecedented precision. Continued advances in electrode design, signal processing, and multimodal integration promise to further enhance the capabilities of these powerful technologies, driving progress in both basic neuroscience and therapeutic development for neurological and psychiatric disorders.
Electroencephalography (EEG) and Magnetoencephalography (MEG) are non-invasive neurophysiological techniques that measure the electromagnetic fields generated by synchronous postsynaptic currents in pyramidal neurons. Although rooted in the same biophysics, they capture distinct signal manifestations: EEG measures electrical potential differences on the scalp, while MEG measures the weak magnetic fields perpendicular to the head surface generated by intracellular currents [33]. These complementary modalities provide a window into neural dynamics with millisecond-scale temporal resolution, capturing brain activity at the speed of thought, which is critical for understanding fast cognitive processes and network interactions [34] [35].
Their non-invasive nature and high temporal resolution position EEG and MEG as essential bridges in the study of neural recording resolution. They offer a unique compromise, providing direct measurements of neuralelectrical activity with finer temporal resolution than functional MRI, though with spatially inferior resolution to invasive methods like electrocorticography (ECoG). This whitepaper details the technical capabilities, methodological advances, and practical applications of these workhorse technologies for a research audience focused on characterizing brain function in health and disease.
Technical Comparison and Spatial Resolution Limits
The fundamental difference in what EEG and MEG measure—electric potentials versus magnetic fields—leads to their distinct performance characteristics and spatial resolution profiles. EEG signals are affected by volume conduction through the skull, scalp, and other tissues, which smears and blurs the electrical potential distribution. MEG signals, being magnetic, are less distorted by the skull and scalp, generally resulting in superior spatial resolution [33].
Recent theoretical work explores the ultimate limits of MEG spatial resolution, describing a two-regime model governed by sensor density and magnetic field smoothness [36]. In the low-density regime, resolution increases with the square root of the number of sensors and is improved by using multi-component sensors. In the asymptotically high-density regime, the inherent smoothness of the magnetic field constrains resolution to a slow, logarithmic divergence as more sensors are added. For scalp MEG using Optically Pumped Magnetometers (OPMs), an interplay of these two regimes controls how resolution saturates as sensors approach the neural sources [36]. The following table summarizes the key quantitative performance metrics for both modalities.
Table 1: Quantitative Performance Comparison of EEG and MEG
| Feature | Electroencephalography (EEG) | Magnetoencephalography (MEG) |
|---|---|---|
| Spatial Resolution | ~10-20 mm (approximate) | A few millimeters to ~10 mm [36] |
| Temporal Resolution | Millisecond (ms) scale | Millisecond (ms) scale [34] [35] |
| Signal Origin | Extracellular volume currents (primarily) | Intracellular primary currents (primarily) |
| Sensitivity | Tangential & radial sources; sensitive to deep sources | Primarily tangential sources; less sensitive to deep sources |
| Typical Channel Count | 64-128 (research); 3-9 (wearable/BCI) [37] | 100-300 (SQUID systems) |
| Classification Accuracy | 61-69% (varies with channel count) [37] | ~73% (with 204 gradiometers) [37] |
A critical technological advancement is the emergence of Optically Pumped Magnetometers (OPMs). A 2025 comparative study of the auditory Mismatch Negativity (MMN) response found that a prototype Helium-OPM system performed equally well or better than EEG in detecting significant individual responses, despite having only five sensors [38]. This demonstrates the potential for wearable MEG systems that combine the benefits of MEG signal acquisition with the practicality of EEG.
Methodological Advances and Experimental Protocols
Core Analytical Workflows
Modern analysis of EEG and MEG data extends beyond simple spectral power analysis to include complex measures of network dynamics and cross-frequency interactions. Key methodologies include:
- Phase-Amplitude Coupling (PAC): This analysis quantifies how the phase of a lower-frequency oscillation (e.g., beta band, 13-30 Hz) modulates the amplitude of a higher-frequency oscillation (e.g., gamma band, 30-100 Hz). In Parkinson's disease (PD), MEG studies have identified pathologically enhanced beta-gamma PAC in the sensorimotor cortex, which correlates with motor symptom severity (akinesia) and is reduced by dopaminergic medication (L-DOPA) [34].
- Dynamic Functional Connectivity (dFC) & Meta-State Analysis: This approach models brain activity as a sequence of rapidly transitioning global brain states ("meta-states"). The temporal structure of these sequences reveals a recurrent, cyclical organization during rest. This pattern is disrupted in neurodegenerative conditions like Alzheimer's disease, showing increased randomness in Mild Cognitive Impairment (MCI) and AD cohorts across both EEG and MEG datasets [39].
- Electromagnetic Source Imaging: This is a robust framework for interpolating poor-quality or missing sensor data. It involves estimating underlying brain source activity using signals from good-quality sensors and the leadfield matrix, then forwarding this reconstructed activity to estimate the data at bad sensor locations. This method is particularly valuable for dealing with noisy sensors or the partial brain coverage of some OPM arrays [40].
- Multimodal Integration with Encoding Models: Deep learning-based encoding models represent a frontier for integrating MEG and fMRI. One novel transformer-based model uses naturalistic stimuli (e.g., narrative stories) to learn a latent cortical source space that simultaneously predicts both MEG and fMRI signals. This approach effectively estimates brain activity with high spatiotemporal resolution, validated by its ability to generalize and predict held-out ECoG data [35].
Detailed Experimental Protocol: Auditory Attention BCI
A 2025 study provides a clear protocol for using MEG/EEG to decode selective auditory attention, a common brain-computer interface (BCI) paradigm [37].
- Objective: To investigate the decrease in classification accuracy when moving from whole-scalp MEG to low-channel-count EEG, and when training classifiers only from the initial part of a recording.
- Stimuli & Task: Participants are presented with two concurrent auditory streams of spoken words ("Yes" by a female speaker, "No" by a male speaker), one to each ear (±40° azimuth). They are instructed to attend to one stream while ignoring the other. The word sequences are presented at 1 Hz, with occasional deviants (5% probability) to enhance attention modulation.
- Data Acquisition: Simultaneous MEG (306 channels) and EEG (64 channels) are recorded.
- Data Analysis: Support Vector Machine (SVM) classifiers are trained on unaveraged trials (1-second duration) to predict the target of attention. Key manipulations include:
- Training Data Sampling: Trials are extracted either randomly from the entire recording or only from the beginning.
- Channel Count: Classifiers are trained on 204 MEG gradiometers or EEG with 64, 30, 9, or 3 channels.
- Key Findings: The highest accuracy (73.2%) was achieved with MEG using randomly sampled training data. EEG accuracy decreased with channel count (69% to 61%). Using only beginning-of-recording data for training caused an average 11% drop in accuracy, highlighting a major challenge for real-time BCI calibration [37].
Applications in Clinical Research and Drug Development
EEG and MEG play increasingly important roles in de-risking drug development and improving clinical outcomes in psychiatry and neurology, primarily through two pathways [41].
Pharmacodynamic Measures and Target Engagement
Functional neuroimaging with EEG/MEG can critically answer key questions early in clinical drug development:
- Brain Penetration: Does the drug affect clinically relevant brain systems?
- Functional Target Engagement: What is the impact on brain oscillations or evoked potentials?
- Dose Selection: What is the dose-response relationship for functional brain effects?
- Indication Selection: How do the brain effects inform the choice of clinical indication?
For example, in developing Phosphodiesterase 4 inhibitors (PDE4i's) for cognitive impairment associated with schizophrenia (CIAS), EEG/ERP measures revealed pro-cognitive effects at ~30% target occupancy—a dose range that would have been missed using PET imaging alone, which typically argues for higher, less-tolerated doses [41].
Biomarkers for Patient Stratification and Enrichment
MEG and EEG provide quantitative biomarkers that can identify patient subgroups most likely to respond to a therapy, thereby enriching clinical trials. In Parkinson's disease, specific MEG signatures show promise for clinical application:
- Cortical Beta Power: Increases in sensorimotor beta power following L-DOPA administration correlate with motor symptom improvement, potentially predicting treatment response [34].
- Beta-Gamma Phase-Amplitude Coupling: Pathologically elevated beta-gamma PAC in PD is distributed broadly across the cortex but linked to motor symptoms specifically in the sensorimotor areas, offering a quantifiable target for intervention [34].
- Temporal Dynamics: Measures of cortical beta bursts and the reduced functional repertoire in PD provide new insights into disease pathophysiology and its modulation by treatments like Deep Brain Stimulation (DBS) [34].
Table 2: Key Neuroimaging Biomarkers in Parkinson's Disease (MEG)
| Biomarker | Physiological Correlation | Modulation by Therapy | Clinical Application Potential |
|---|---|---|---|
| Sensorimotor Beta Power | Decreased in advanced PD "OFF" state | Increased by L-DOPA; correlates with motor improvement [34] | Predict L-DOPA response [34] |
| Beta-Gamma Phase-Amplitude Coupling | Correlates with akinesia severity [34] | Diminished by L-DOPA [34] | Optimize DBS targeting [34] |
| Frontal Alpha Eigenvalue Centrality | Elevated network influence; correlates with UPDRS-III score [34] | Modulated by therapies | Patient stratification & tracking progression |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Technologies and Analytical Tools for Modern EEG/MEG Research
| Tool Category | Specific Example | Function & Explanation |
|---|---|---|
| MEG Sensor Technology | SQUID-MEG (Cryogenic) | Traditional systems with sensors 2-4 cm from scalp; high sensitivity but bulky [38] [36]. |
| MEG Sensor Technology | Optically Pumped Magnetometers (OPMs) | Next-generation, wearable sensors operating at room temperature; enable "scalp MEG" [38] [36]. |
| EEG Sensor Technology | Dry Electrode Headsets | Enable rapid setup without conductive gel; suitable for home-based monitoring and long recordings [42]. |
| EEG Sensor Technology | Ear-EEG Systems | Discreet form factor for recordings within the ear canal; improves user comfort for long-term use [42]. |
| Computational Framework | Electromagnetic Source Imaging | A robust method for interpolating bad sensor data or enhancing spatial resolution by estimating underlying brain sources [40]. |
| Multimodal Fusion Model | MEG-fMRI Encoding Model (e.g., [35]) | Deep learning model that combines MEG and fMRI data to estimate latent cortical sources with high spatiotemporal resolution. |
| Foundation Model | BrainOmni [33] | A foundation model performing unified pretraining on both EEG and MEG signals, enabling cross-modality generalization. |
EEG and MEG remain indispensable non-invasive workhorses for cognitive neuroscience and clinical neuropharmacology. The field is undergoing a significant transformation, driven by wearable technologies like OPM-MEG and dry-EEG, and sophisticated computational methods such as foundation models and multimodal fusion. These advances are steadily overcoming traditional limitations, particularly in spatial resolution and real-world applicability. For researchers and drug development professionals, leveraging these tools to quantify target engagement and identify patient subgroups through defined electrophysiological biomarkers offers a promising path to de-risking clinical trials and advancing precision medicine in neurology and psychiatry.
In the field of non-invasive neural recording, the trade-off between spatial and temporal resolution presents a fundamental challenge. Functional Magnetic Resonance Imaging (fMRI) and Functional Near-Infrared Spectroscopy (fNIRS) have emerged as two pivotal neuroimaging techniques that measure cerebral hemodynamics—the indirect surrogate of neural activity. Both methods leverage the principle of neurovascular coupling, where neuronal firing triggers hemodynamic changes, but they differ substantially in their underlying physics, operational requirements, and resultant data characteristics [43] [44]. fMRI is widely regarded as the gold standard for in-vivo brain imaging due to its high spatial resolution and whole-brain coverage, yet it is constrained by its immobility, cost, and limited temporal resolution [45] [44]. In contrast, fNIRS offers a portable, cost-effective alternative with superior temporal resolution and higher tolerance for movement, making it suitable for naturalistic environments and vulnerable populations, albeit with more limited spatial resolution and cortical penetration [43] [46] [47]. This technical guide provides an in-depth comparison of these two modalities, detailing their core principles, technical specifications, experimental protocols, and emerging applications, with a specific focus on their roles in spatial and temporal resolution within non-invasive brain research.
Core Technical Principles
Functional Magnetic Resonance Imaging (fMRI)
fMRI measures brain activity indirectly through the Blood-Oxygen-Level-Dependent (BOLD) contrast. The underlying physiological mechanism begins when neural activity increases local oxygen consumption, leading to a subsequent rise in cerebral blood flow (CBF) and cerebral blood volume (CBV) that overcompensates for the initial oxygen extraction. This process reduces the concentration of paramagnetic deoxyhemoglobin (HbR) in the local vasculature. As the MRI scanner employs a strong static magnetic field and radiofrequency pulses, these shifts in hemoglobin concentration cause localized changes in magnetic susceptibility, detectable as a subtle increase in the T2*-weighted signal—the BOLD response [45] [48]. The BOLD signal is a complex, indirect metric that is influenced by cerebral metabolic rate of oxygen (CMRO2), CBF, and CBV.
Preclinical fMRI advancements have been driven by ultrahigh-field scanners (e.g., 7 Tesla to 18 Tesla), which provide a supra-linear increase in functional contrast-to-noise ratio (fCNR). Key hardware innovations include high-performance gradient coils with strengths up to 1000 mT/m and slew rates of 9000 T/m/s for enhanced spatial and temporal resolution, along with specialized radiofrequency coils—such as cryogenically cooled and implantable coils—that significantly boost the signal-to-noise ratio (SNR) [48].
Functional Near-Infrared Spectroscopy (fNIRS)
fNIRS is an optical neuroimaging technique that utilizes near-infrared light (700-900 nm) to measure changes in the concentrations of oxyhemoglobin (HbO) and deoxyhemoglobin (HbR) in cortical tissue. Within this "optical window," biological tissues are relatively transparent, allowing light to penetrate the scalp and skull and be detected after undergoing absorption and scattering [43] [46]. The technique applies the Modified Beer-Lambert Law to convert measured changes in light attenuation into relative changes in chromophore concentrations.
The fundamental physiological premise is neurovascular coupling: during neural activation, a localized increase in metabolic demand triggers a rise in cerebral blood flow, leading to an increase in HbO and a concurrent decrease in HbR. A typical fNIRS setup includes multiple light sources (emitting at least two different wavelengths) and detectors arranged on the scalp, with a source-detector separation of approximately 3 cm for adults. The detected light travels a "banana-shaped" path through the tissues, sampling the outer cortex to a depth of about 1-2.5 cm [43] [47]. fNIRS systems are categorized into continuous-wave, frequency-domain, and time-domain systems, with continuous-wave being the most common due to its simplicity and cost-effectiveness [43].
Technical Comparison and Quantitative Data
Performance Metrics and Capabilities
Table 1: Direct Comparison of fMRI and fNIRS Technical Specifications
| Feature | fMRI | fNIRS |
|---|---|---|
| Spatial Resolution | High (millimeter-level) [44] | Moderate (centimeter-level), limited to cortical surface [44] [47] |
| Temporal Resolution | Low (seconds) [44] | Moderate (tenths of seconds to seconds) [46] |
| Penetration Depth | Whole-brain, including deep structures [44] | Superficial cortex (1-2.5 cm) [44] [47] |
| Measured Signal | BOLD (T2* change, inversely related to HbR) [44] [48] | Direct concentration changes of HbO and HbR [43] [46] |
| Portability | Low (requires fixed scanner) [44] | High (wearable/wireless systems available) [46] [44] |
| Tolerance to Motion | Low (highly sensitive) [44] | Moderate (relatively robust) [43] [47] |
| Subject Compatibility | Restricted (no metal implants, claustrophobia) [44] | High (suitable for infants, children, patients with implants) [43] [44] |
| Operational Cost | Very high [44] | Moderate [44] [47] |
| Acoustic Noise | High (can interfere with tasks) [44] | Minimal [44] |
Spatial and Temporal Correspondence
Multimodal studies investigating the relationship between fMRI and fNIRS signals provide critical insights for cross-modal validation and integration. Research involving motor execution and imagery tasks has demonstrated a significant spatial correspondence between the modalities. In such paradigms, activation clusters identified in fMRI data modeled from fNIRS signals showed significant peak activation overlapping individually defined primary and premotor cortices [49]. No statistically significant differences were observed in multimodal spatial correspondence between HbO, HbR, and total hemoglobin (HbT) concentrations [49]. The temporal correlation between the BOLD signal and fNIRS chromophores varies across studies, with some reporting higher correlations with HbO and others with HbR or HbT, indicating that the relationship is complex and may be task-dependent [49].
Detailed Experimental Protocols
A Representative Multimodal fMRI/fNIRS Protocol
The following protocol, adapted from a study on motor tasks, exemplifies a rigorous approach to acquiring asynchronous fMRI and fNIRS data for comparative analysis [49].
Objective: To analyze the spatial correspondence between fMRI BOLD and fNIRS hemodynamic signals (HbO, HbR, HbT) in motor-network regions during motor imagery and execution.
Participants: 9 healthy adults with no neurological history.
Paradigm Design (Block Design):
- Conditions: Motor Action (MA), Motor Imagery (MI), and Baseline.
- Block Structure: 17 blocks total (9 Baseline, 4 MA, 4 MI), each 30 seconds duration.
- Total Run Time: 8 minutes 30 seconds.
- Task Instructions:
- MA Blocks: Participants executed a bilateral finger-tapping sequence at a specified frequency (e.g., 2 Hz).
- MI Blocks: Participants imagined the same sequence without physical movement.
fMRI Data Acquisition:
- Scanner: 3 Tesla Siemens Magnetom TimTrio with a 12-channel head coil.
- Structural Scan: MPRAGE sequence (176 slices; 1×1×1 mm voxels).
- Functional Scan: T2*-weighted EPI sequence (26 slices; 3×3 mm in-plane resolution; slice thickness: 3.5 mm; TR/TE = 1500/30 ms).
- Preprocessing: Slice-time correction, motion correction, temporal high-pass filtering, spatial smoothing (Gaussian kernel, FWHM=6 mm), co-registration to structural data, and normalization to standard (Talairach) space.
fNIRS Data Acquisition:
- System: NIRSport2 continuous-wave system (NIRx Medical Technologies), sampled at 5.08 Hz.
- Setup: 16 sources (760 & 850 nm) and 15 detectors (54 channels; inter-optode distance: 30 mm) covering bilateral motor areas. 8 short-distance detectors (8 mm separation) were included to regress out extracerebral confounds.
- Preprocessing (Homer3 software): Channel pruning based on signal-to-noise ratio (SNR < 15 dB), conversion of raw intensity to optical density, and application of the Modified Beer-Lambert law to compute HbO and HbR concentration changes.
Analysis:
- fMRI General Linear Model (GLM): Individual analysis with predictors for MA, MI, and Baseline, plus motion parameters and spike covariates. Regions of interest (ROIs) for primary motor (M1) and premotor (PMC) cortices were defined for each subject.
- Spatial Correspondence: Subject-specific fNIRS time series from motor ROIs were used as predictors in GLMs of the fMRI data to identify corresponding brain activation.
A Clinical fNIRS Protocol for Psychiatry
This protocol details the application of fNIRS in a clinical research setting, specifically for classifying patients with Major Depressive Disorder (MDD) [50].
Objective: To use fNIRS to explore prefrontal cortex function in first-episode, drug-naive MDD patients with and without suicidal ideation during a cognitive task and evaluate its diagnostic utility via machine learning.
Participants:
- Patient Groups: 91 first-episode, drug-naive MDD patients, subdivided into 40 with suicidal ideation (SIs) and 51 without (NSIs) based on HAMD-17.
- Control Group: 39 healthy controls (HCs).
Paradigm: Verbal Fluency Task (VFT)
- This task engages executive functions (memory, language, attention) known to be impaired in MDD. Participants generate words according to a given category or initial letter.
- fNIRS Setup: A multi-channel fNIRS system measured hemodynamic responses in the prefrontal cortex, focusing on regions like the dorsolateral prefrontal cortex (DLPFC), frontopolar cortex (FPC), and orbitofrontal cortex (OFC).
Data Analysis:
- Statistical Analysis: Kruskal-Wallis test to analyze HbO changes among the three groups across different prefrontal regions.
- Machine Learning: A one-dimensional Convolutional Neural Network (1D-CNN) was employed to automatically extract features from the fNIRS time-series data and perform three-class classification (SIs, NSIs, HCs). This deep learning approach mitigates the limitations of manual feature engineering in traditional analysis.
Signaling Pathways and Experimental Workflows
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Equipment and Analytical Tools for Hemodynamic Research
| Item | Function/Description | Example Use Case |
|---|---|---|
| Ultra-High Field MRI Scanner | Preclinical systems (7T-18T) with high-performance gradients (e.g., 400-1000 mT/m) for enhanced fCNR and spatial resolution [48]. | Mapping whole-brain functional networks in rodent models with high spatial precision [48]. |
| Cryogenic Radiofrequency Coils | Liquid nitrogen or helium-cooled coils that reduce electronic noise, providing ~3x SNR gain and ~1.8x tSNR improvement at 9.4T [48]. | Boosting detection sensitivity for weak BOLD responses in high-resolution fMRI studies [48]. |
| High-Density fNIRS Systems | Systems with >32 channels and short-distance detectors for improved spatial sampling and signal quality [49] [46]. | Assessing prefrontal cortex function in psychiatric populations during cognitive tasks [46] [50]. |
| Optode Digitalization Kit | 3D digitizers for coregistering fNIRS optode positions with standard brain atlases (e.g., AtlasViewer) [44]. | Ensuring accurate spatial registration of fNIRS channels to underlying cortical anatomy [44]. |
| Integrated EEG-fNIRS Caps | Head caps with pre-defined openings for simultaneous EEG electrode and fNIRS optode placement using the 10-20 system [47]. | Multimodal studies combining EEG's temporal resolution with fNIRS's spatial localization [47]. |
| Homer3 / BrainVoyager | Specialized software packages for fNIRS (Homer3) and fMRI (BrainVoyager) data preprocessing and statistical analysis [49]. | Standardized pipeline for converting raw signals (light intensity/BOLD) into analyzable data [49]. |
| 1D Convolutional Neural Network (1D-CNN) | Deep learning architecture for automatic feature extraction from fNIRS time-series data [50]. | Classifying patient groups (e.g., MDD with vs. without suicidal ideation) based on hemodynamic patterns [50]. |
Emerging Trends and Future Directions
The field of hemodynamic monitoring is rapidly evolving, with several key trends shaping its future. Multimodal integration represents a primary frontier, as evidenced by novel transformer-based encoding models that combine MEG and fMRI data from naturalistic experiments to estimate cortical source activity with both high spatial and temporal resolution [51]. Similarly, simultaneous fNIRS-fMRI measurements are being used to validate fNIRS signals and leverage the complementary strengths of both modalities [49] [44].
In data analysis, deep learning is revolutionizing both fMRI and fNIRS. For fMRI, convolutional neural networks (CNNs), recurrent neural networks (RNNs), and transformers are being deployed for brain function mapping, moving beyond traditional general linear models (GLM) and independent component analysis (ICA) [45]. For fNIRS, 1D-CNNs are demonstrating high accuracy in classifying clinical populations, such as distinguishing depressed patients with suicidal ideation from those without [50].
Technological miniaturization and the push for naturalistic neuroscience are expanding the applications of fNIRS. Its portability and motion tolerance enable brain imaging in real-world settings—classrooms, homes, and during social interactions—that are inaccessible to traditional fMRI [43] [44] [47]. Finally, the emergence of brain foundation models, pre-trained on large-scale neuroimaging datasets, promises to create generalizable representations of brain activity that can be fine-tuned for diverse cognitive tasks and clinical applications across both modalities [45].
The pursuit of technologies that offer high spatial and temporal resolution for mapping brain activity is a central challenge in neuroscience. Traditional invasive methods, such as intracortical electrode arrays, provide high-fidelity signals but carry surgical risks and are limited in spatial coverage. Non-invasive techniques, like electroencephalography (EEG), are safe and versatile but suffer from lower spatial resolution and signal clarity due to the distorting effects of the skull and scalp [17] [1]. This creates a significant resolution gap, limiting our ability to observe brain-wide neural dynamics at a microscopic scale. In response, emerging hybrid and novel approaches are pushing the boundaries of what is possible. This guide details two such transformative technologies—Functional Ultrasound (fUS) and Digital Holographic Imaging (DHI)—which offer new ways to study neural activity with unprecedented resolution and minimal invasiveness, thereby enriching the landscape of spatial and temporal resolution in neural recording research.
Technical Deep Dive: Functional Ultrasound (fUS)
Core Principles and Methodology
Functional ultrasound (fUS) is a hemodynamic-based neuroimaging technology that measures changes in cerebral blood volume (CBV) by detecting backscattered echoes from red blood cells [52] [53]. This process relies on the principle of neurovascular coupling, where neuronal activity triggers localized changes in blood flow. fUS leverages ultrafast Doppler imaging to generate Power Doppler images with high spatiotemporal resolution (~100 µm spatial and ~100 ms temporal), providing a large field of view to visualize widespread brain networks [53] [54].
A typical fUS experimental workflow involves several key steps. First, animals are prepared, often under anesthesia, and a craniotomy may be performed. A linear ultrasound probe (e.g., 15 MHz center frequency) is positioned over the region of interest. The session begins with a 3D Power Doppler scan registered to a vascular reference atlas for anatomical navigation [52] [53]. During functional acquisition, data is continuously recorded, often in blocks. Subsequent processing involves motion correction and advanced filtering techniques to isolate the functional signal from physiological noise such as breathing and heart rate [52] [53].
Advanced Analysis and Machine Learning Integration
To fully exploit fUS's high-resolution data, advanced analytical methods are being developed. Traditional region-of-interest (ROI) analysis can mask finer spatial variations [54]. Spatiotemporal clustering at the single-voxel level offers an unbiased, paradigm-free method to segment active brain regions with high detail, revealing intricate patterns of hemodynamic responses [54].
Furthermore, machine learning models are increasingly applied to fUS data. Studies have successfully used Convolutional Neural Networks (CNNs), Support Vector Machines (SVMs), and Vision Transformers (ViTs) to classify brain states, such as distinguishing the effects of pharmacological agents like the NMDA receptor antagonist MK-801 from controls [53]. When combined with class activation mapping, CNNs can localize drug effects to anatomically specific regions like the prefrontal cortex and hippocampus, creating a powerful, data-driven framework for neuropharmacological research [53].
Pushing Resolution Limits with Deep Learning
The spatial resolution of fUS is fundamentally limited by the diffraction of sound. However, deep learning-based super-resolution techniques are breaking through this barrier. By leveraging data from Ultrasound Localization Microscopy (ULM), Super-Resolution fUS (SR-fUS) can achieve microscopic resolution of 25 µm spatial and 10 ms temporal resolution, enabling the imaging of transient, brain-wide neurovascular activity on a microscopic scale [55]. This allows for the detailed observation of cortical microvasculature during sensory stimulation, as validated by comparative studies with two-photon microscopy [55].
Technical Deep Dive: Digital Holographic Imaging (DHI)
Core Principles and Methodology
Digital Holographic Imaging (DHI) represents a paradigm shift by detecting a novel signature of neural activity: nanometer-scale mechanical deformations of neural tissue that occur during action potentials [4] [56]. Unlike methods relying on hemodynamics or electrical potentials, DHI is a full-field, label-free, optical phase-based technique that measures the out-of-plane velocity and displacement of tissue with sub-nanometer sensitivity [56].
The core of a DHI system is an interferometer that illuminates the cortex with coherent light. The light scattered from the tissue is coherently mixed with a reference beam at a camera sensor, forming an interference pattern or hologram. Digital processing of these holograms involves filtering and applying a Fresnel transform to compute complex images of the sensing volume at kHz rates. The phase information from subsequent images is then cross-correlated and averaged within a region of interest to compute nanometer-scale frame-to-frame tissue displacement [56]. This method is uniquely sensitive to the rapid, tiny physical movements that accompany population-level neuronal firing.
Experimental Validation and Transcranial Potential
DHI has been rigorously validated across multiple in vivo neural activation models, demonstrating its versatility and sensitivity.
- Focal Electrical Stimulation (FES): DHI records tissue deformation adjacent to the stimulation site within milliseconds, with signal amplitude modulated by the stimulation current [56].
- Whisker Barrel Stimulation: DHI can detect intrinsically evoked responses in the primary somatosensory cortex, demonstrating its ability to map functional pathways without direct intervention [56].
- Pharmacological Models: DHI differentiates between brain states, such as anesthetized, normal, and seizure activity, the latter characterized by larger, synchronous neural activation patterns [56].
A key advantage of DHI is its transcranial capability. By using a longer wavelength laser (e.g., 1310 nm) that reduces optical scatter, DHI can achieve at-depth sensing through the intact skull, paving the way for truly non-invasive, high-resolution recording of cortical activity [56].
Comparative Analysis: fUS vs. DHI
The following tables provide a direct comparison of the technical specifications and experimental considerations for fUS and DHI, summarizing their respective positions in the landscape of neural recording technologies.
Table 1: Quantitative Technical Specifications of fUS and DHI
| Feature | Functional Ultrasound (fUS) | Digital Holographic Imaging (DHI) |
|---|---|---|
| Primary Signal | Hemodynamic (Cerebral Blood Volume) [53] | Mechanic (Tissue Deformation) [56] |
| Spatial Resolution | 100 µm (standard); 25 µm (super-resolution) [53] [55] | ~30 µm (lateral) [56] |
| Temporal Resolution | 100 ms (standard); 10 ms (super-resolution) [53] [55] | Sub-millisecond (kHz acquisition) [56] |
| Field of View | Whole-brain (sectional); Wide-field [54] | Several mm² (limited by optics) [56] |
| Penetration Depth | Several cm (through bone) [57] | ~1 mm (transcranial with 1310nm laser) [56] |
| Key Advantage | Large FOV, deep penetration, clinical translation | Ultra-high temporal resolution, direct neural coupling |
Table 2: Experimental and Practical Considerations
| Consideration | Functional Ultrasound (fUS) | Digital Holographic Imaging (DHI) |
|---|---|---|
| Invasiveness | Minimally invasive (often requires craniotomy in animals); transcranial in neonates [52] [57] | Non-invasive (can be performed transcranially) [56] |
| Primary Noise Sources | Motion, cardiac pulsatility, respiration [52] | Physiological clutter (blood flow), optical phase noise [56] |
| Key Analysis Methods | Canonical Correlation Analysis (CCA), dynamic FC, spatiotemporal clustering, CNN [52] [53] [54] | Complex cross-correlation, spatial averaging, stimulus-locked averaging [56] |
| Suitable Applications | Functional connectivity, drug pharmacodynamics, neurodevelopment [52] [53] | Mapping population-level activation, studying neural dynamics at high speed [56] |
Essential Research Toolkit
Table 3: Key Research Reagent Solutions and Materials
| Item | Function | Example in Context |
|---|---|---|
| fUS Scanner & Probe | Hardware for data acquisition. | Iconeus One scanner with IcoPrime linear probe (15 MHz) [52] [53]. |
| DHI Interferometer System | Optical system for detecting nanometer-scale displacements. | Custom DHI system with 780 nm or 1310 nm laser sources [56]. |
| Canonical Correlation Analysis (CCA) | Algorithm for denoising fUS data by removing physiological and motion artifacts [52]. | Used to reveal subtle functional connectivity alterations in a perinatal inflammation mouse model [52]. |
| Spatiotemporal Clustering | Data mining technique for unbiased analysis of single-voxel fUS signals [54]. | Employed to map detailed visually-evoked activity in the mouse superior colliculus without predefined regions [54]. |
| Convolutional Neural Network (CNN) | Machine learning model for classifying and localizing brain states from fUS data [53]. | Applied to identify and map the effects of MK-801 on brain regions with high NMDA receptor density [53]. |
| Super-Resolution fUS (SR-fUS) | Deep-learning method that surpasses diffraction limit for microscopic resolution [55]. | Used to image transient hemodynamic responses in cortical microvasculature during whisker stimulation [55]. |
Experimental Protocols
Detailed Protocol: fUS for Functional Connectivity in a Murine Inflammation Model
This protocol is adapted from a 2025 study investigating the long-term effects of mild perinatal inflammation on brain networks in mice [52].
Animal Model Preparation:
- Use OF1 male mice. Induce systemic perinatal inflammation via intraperitoneal (i.p.) injections of 10 µg.kg⁻¹ IL-1β twice daily from postnatal days P1 to P5. Control groups receive PBS injections.
- At P30, prepare mice for imaging. Induce anesthesia with an i.p. injection of xylazine (15 mg/kg) and ketamine (75 mg/kg).
Surgical Preparation and fUS Acquisition:
- Fix the anesthetized mouse in a stereotaxic frame. Perform a craniotomy.
- Mount a linear ultrasound probe (15 MHz) on a motorized system. Conduct an initial 3D Power Doppler scan for vascular mapping and registration to the Allen Mouse Brain Atlas.
- For functional connectivity recording, acquire resting-state fUS data at a frame rate of 2.5 Hz for a sustained period.
Data Preprocessing with CCA Denoising:
- To isolate neural-related signals from noise (motion, cardiac, respiration), apply Canonical Correlation Analysis (CCA). This method identifies and removes common noise signals present in both functional and non-functional areas while preserving a continuous time course.
Dynamic Functional Connectivity (dFC) Analysis:
- Calculate the succession of connectivity states (or "states of high FC") over time from the denoised data.
- Quantify the occurrence rates, mean dwell times, and transition probabilities between these different brain-wide connectivity states. Compare these metrics between the IL-1β and control groups to reveal subtle alterations induced by inflammation.
Detailed Protocol: DHI for In Vivo Neural Activity Recording
This protocol is based on a 2024 study demonstrating transcranial recording of population-level cortical activity [56].
Animal Preparation:
- Use Sprague-Dawley rats (350-450 g). Anesthetize and paralyze the animal, placing it on a ventilator.
- For transcranial measurements, ensure the skull remains intact. For higher sensitivity recordings with a cranial window, perform a hemicraniectomy.
DHI System Setup:
- Set up the off-axis digital holographic interferometer. For transcranial measurements, use a 1310 nm laser with a short coherence length (~50 µm) to gate signals at depth and reduce optical scatter.
- Position the sensing volume (coherence gate) within the cortical region of interest using a delay line in the reference arm.
Neural Activation and Data Acquisition:
- Choose an activation model:
- Focal Electrical Stimulation (FES): Place tungsten stimulating electrodes on the epidural surface. Deliver repetitive cathodic bipolar stimulations (e.g., 10 Hz) at varying currents (0.25-0.75 mA).
- Whisker Stimulation: Mechanically deflect whiskers to evoke intrinsic activity in the barrel cortex.
- Acquire holograms at a high frame rate (>1.5 kHz) to minimize optical phase noise.
- Choose an activation model:
Data Processing and Displacement Calculation:
- Process acquired holograms by filtering the DC term and applying a Fresnel transform to compute complex images.
- Compute the cross-correlation (\chi(\boldsymbol{x},t)) between subsequent images (Eq. 1).
- Calculate the average phase change ( < \Delta \varphi > (t) ) within an ROI by averaging the real and imaginary components of (\chi) in the complex domain (Eq. 2).
- Compute the tissue velocity (v(t)) by scaling the phase change by the laser wavelength and sampling interval (Eq. 3). Integrate velocity over time to obtain displacement.
- For weak signals, use stimulus-locked averaging (SLA) across multiple trials to enhance the signal-to-noise ratio.
Workflow and Signaling Diagrams
The following diagram illustrates the core signal processing workflow for Digital Holographic Imaging.
DHI Signal Processing Workflow
The following diagram outlines the key steps in a functional ultrasound experiment, from acquisition to advanced analysis.
fUS Experimental and Analysis Pipeline
Functional Ultrasound and Digital Holographic Imaging are at the forefront of a new wave of neurotechnologies that transcend traditional compromises between invasiveness, and spatial and temporal resolution. fUS provides a unique combination of large field-of-view, good penetration, and high sensitivity to hemodynamic signals, making it ideal for studying brain-wide networks and the effects of pharmaceuticals. DHI, by contrast, offers a direct, high-speed optical readout of mechanical tissue deformations associated with neural firing, opening a new window into fast cortical dynamics. Both technologies, especially when augmented by advanced computational methods like machine learning and super-resolution processing, are rapidly closing the resolution gap that has long separated invasive and non-invasive neural recording methods. Their continued development promises to unlock deeper insights into brain function in health and disease, significantly impacting basic neuroscience and drug development.
The selection between invasive and non-invasive neural recording technologies represents a critical strategic decision in both clinical and research neuroscience. This choice fundamentally balances trade-offs between signal quality, risk, spatial resolution, and temporal resolution against the backdrop of specific application requirements. Invasive techniques, which involve surgical implantation of electrodes directly into brain tissue, provide unparalleled access to high-frequency neural signals but carry inherent medical risks [17]. Non-invasive approaches, while safer and more accessible, must contend with signal attenuation and spatial blurring caused by intervening tissues [58]. Understanding these technical characteristics is essential for matching recording methodologies to specific clinical and research objectives in what constitutes a rapidly evolving technological landscape.
This whitepaper examines the technical specifications governing invasive and non-invasive neural interfaces, provides a structured framework for technique selection, and explores emerging technologies that may redefine current limitations. The analysis is situated within the broader context of spatial and temporal resolution considerations that fundamentally constrain what neural phenomena can be successfully captured and utilized for various applications.
Technical Specifications and Quantitative Comparisons
Fundamental Characteristics of Recording Modalities
Spatial resolution refers to the smallest distinguishable spatial detail in neural activity, while temporal resolution refers to the ability to track changes in neural activity over time. These two dimensions are not independent; factors that degrade spatial resolution often simultaneously impair temporal accuracy [58].
For non-invasive electroencephalography (EEG), the spatial resolution is classically considered poor (approximately 5-9 cm) due to volume conduction effects where electrical signals spread through resistive layers like the skull, causing blurring at the scalp level [58]. The temporal resolution of conventional scalp EEG is often overestimated, as volume conduction distorts the recovered time course of underlying sources, effectively degrading its actual temporal resolution despite the instantaneous propagation of electrical activity [58].
Invasive recordings capture signals closer to their neural sources, bypassing the signal-degrading effects of skull and scalp. Intracortical electrodes can record both action potentials (APs) and local field potentials (LFPs) with much higher spatial specificity. These electrodes typically yield LFPs and detectable APs of 0-5 identifiable neurons per intact contact, with information content available up to several kHz [17].
Table 1: Spatial and Temporal Resolution Characteristics of Neural Recording Techniques
| Technique | Spatial Resolution | Temporal Resolution | Primary Signal Sources | Depth Penetration |
|---|---|---|---|---|
| scalp EEG | ~5-9 cm [58] | Degraded by volume conduction [58] | Pyramidal neuron postsynaptic currents [17] [28] | Cortical surface |
| ECoG | Higher than EEG | Millisecond | Pyramidal neurons | Cortical surface |
| Intracortical Arrays | Single neuron resolution | <1 ms [17] | Action potentials, local field potentials | Multiple cortical layers |
| fNIRS | ~1-2 cm | Seconds [22] | Hemodynamic response | Superficial cortex |
| DHI | Potentially high [4] | Unknown | Neural tissue deformation | Unknown |
Table 2: Information Content and Performance Characteristics
| Technique | Frequency Range | Information Transfer Rate | Key Advantages | Principal Limitations |
|---|---|---|---|---|
| Non-invasive EEG | <~90 Hz (lower for dry electrodes) [17] | Lower than invasive [59] | Risk-free, low cost, whole-brain coverage [14] [17] | Spatial smearing, dominated by pyramidal neurons [17] |
| Invasive LFP | Up to several kHz [17] | Higher than non-invasive | Reflects input, processing and output of cortical areas [17] | Surgical risk, limited coverage [17] |
| Invasive AP | Up to several kHz [17] | Highest potential [59] | Single-neuron resolution | Surgical risk, signal stability over time |
Signal Composition and Origin Differences
The fundamental nature of signals differs substantially between invasive and non-invasive approaches. Non-invasive EEG signals originate primarily from postsynaptic extracellular currents of pyramidal neurons, whose morphological structure (long, parallel dendrites) allows their fields to superimpose and reach the scalp [17]. This selective sensitivity means EEG provides a biased view of neural activity, predominantly reflecting the activity of specific cell types.
Invasive recordings capture a more diverse neural landscape, reflecting a superposition of various electrophysiological processes including those underlying EEG plus interneurons, action potentials, and other local phenomena [17]. This comprehensive access to different neural events provides invasive approaches with richer information content for decoding neural representations.
Application-Specific Selection Framework
Clinical Applications and Considerations
Motor Restoration and Rehabilitation
For severely paralyzed patients, invasive Brain-Machine Interfaces (BMIs) offer the potential for multidimensional control of prosthetic limbs or computer cursors. The primary advantage of invasive approaches in this domain is the possibility of achieving higher information transfer rates necessary for dexterous control [17] [59]. Additionally, invasive BMIs enable bidirectional communication, allowing not only control output but also restoration of sensation through cortical microstimulation of sensory areas [17] [28].
Clinical applications of invasive motor BMIs typically target layer 5 of the motor cortex, which contains large corticospinal neurons critical for motor control [17]. Even with relatively short electrode arrays, access to this region is possible due to the thin layer 4 in motor cortex [17]. Performance improves through closed-loop training as neural tuning adapts to control the BMI, facilitated by neuroplasticity [17].
Neuromodulation for Psychiatric Disorders
Both invasive and non-invasive approaches show promise for treating substance use disorders (SUDs) and other psychiatric conditions by targeting dysfunctional neural circuits. Non-invasive techniques like repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) can target components of the mesocorticolimbic system, including the dorsolateral prefrontal cortex (DLPFC) [60].
Invasive neuromodulation via deep brain stimulation (DBS) of the nucleus accumbens has shown promise in reducing cravings in SUDs [60]. The selection between approaches involves weighing the superior target precision of invasive techniques against the lower risk profile of non-invasive methods.
Diagnostic and Monitoring Applications
Simultaneous invasive and non-invasive recordings are advancing the understanding and treatment of epilepsy by providing a "Rosetta stone" for deciphering brain activity [16]. These combined approaches help validate non-invasive source localization against direct intracranial recordings, improving diagnostic accuracy.
Non-invasive approaches offer significant advantages for long-term monitoring of brain function and health. Emerging technologies like digital holographic imaging (DHI) aim to monitor brain function through the skull without invasive methods, potentially enabling noninvasive measurement of intracranial pressure - a significant clinical advancement [4].
Research Applications
Basic Neuroscience Research
For investigating fundamental neural processes at cellular resolution, invasive recordings remain indispensable. They provide access to the full spectrum of neural signals from single-neuron action potentials to population-level LFPs [17]. In human neuroscience, single-neuron recordings in the hippocampal formation of epileptic patients have provided unique insights into memory encoding, revealing concept cells that may provide a neural mechanism underlying memory representations [61].
Simultaneous invasive and non-invasive recordings serve as a powerful approach for understanding the relationship between different signal types and validating non-invasive measures against ground-truth invasive recordings [16]. This approach is particularly valuable for improving source localization algorithms for EEG and MEG.
Social and Cognitive Neuroscience
Hyperscanning techniques using non-invasive methods like EEG and fNIRS have enabled the study of brain-to-brain synchrony during social interactions [22]. This research explores neural synchronization among multiple individuals performing shared tasks, revealing the neural underpinnings of social coordination. The non-invasive nature of these techniques makes them ideally suited for studying naturalistic social interactions.
The most common approach to determining neural connectivity in these studies is coherence measure utilizing phase-locking value (PLV) in EEG studies, followed by wavelet transform coherence (WTC) in fNIRS studies [22]. These methods allow researchers to quantify inter-brain synchronization during social interactions.
Experimental Protocols and Methodologies
Protocol for Invasive Motor BMI Implementation
Surgical Implantation: Multi-electrode arrays are implanted in motor cortical areas. Utah arrays with electrode lengths of 1.5mm or floating microelectrode arrays with longer electrodes (up to 10mm) can be used depending on target depth [17]. For regions like the anterior wall of the central sulcus, longer electrodes may be necessary to follow layers into the sulcus.
Signal Acquisition: Record both action potentials (300Hz-10kHz) and local field potentials (1-300Hz) simultaneously. APs are typically extracted using thresholding techniques, while LFPs are obtained by appropriate filtering [17].
Feature Extraction: For motor control, both spike rates and LFP features (e.g., power in specific frequency bands) can be used as control signals [17].
Decoder Training: Implement closed-loop calibration where users perform attempted movements while feedback is provided. This facilitates adaptive changes in neural tuning necessary for effective BMI control [17].
Bidirectional Interface: Incorporate intracortical microstimulation for sensory feedback when applicable, using charge-balanced pulses to safely stimulate cortical tissue [17].
Protocol for Non-invasive Brain-to-Brain Synchrony Studies
Experimental Setup: Utilize hyperscanning arrangements where multiple participants are recorded simultaneously while engaging in coordinated tasks [22]. Common paradigms include guitar duets, conversation, or cooperative games.
Data Acquisition: Employ synchronized EEG systems with high-density electrode caps (64+ channels). Ensure precise temporal synchronization between recording systems [22].
Preprocessing: Apply surface Laplacian transformation to reduce volume conduction effects and improve spatial resolution [58]. This transform provides a fair estimate of the corticogram by reducing skull-induced volume conduction.
Synchrony Quantification: Calculate phase-locking values (PLV) between brains in specific frequency bands. Use wavelet transform coherence (WTC) for fNIRS-based studies [22].
Statistical Validation: Compare task-related synchrony to appropriate control conditions (e.g., rest periods or pseudo-interactions) to establish significance [22].
Visualization of Technique Selection and Signal Pathways
Diagram 1: Technique selection decision tree
Diagram 2: Neural signal pathways comparison
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Research Reagent Solutions for Neural Recording
| Item | Function | Example Specifications | Application Context |
|---|---|---|---|
| High-Density EEG Cap | Non-invasive scalp potential recording | 64+ electrodes, saline-based or dry electrodes | Brain-to-brain synchrony studies, clinical monitoring [22] |
| Surface Laplacian Algorithm | Spatial deblurring of EEG signals | Current Source Density (CSD) estimation | Improving spatiotemporal resolution of EEG [58] |
| Multi-Electrode Arrays | Invasive neural signal recording | Utah array (1.5mm electrodes) or FMA (10mm electrodes) | Intracortical recording for motor BMIs [17] |
| Digital Holographic Imaging | Non-invasive optical recording | Nanometer-scale displacement sensitivity | Emerging technology for non-invasive recording [4] |
| Phase-Locking Value Algorithm | Neural synchrony quantification | Complex-valued cross-spectral density | Measuring inter-brain synchronization [22] |
| Intracortical Microstimulator | Bidirectional interface | Charge-balanced pulses, current-controlled | Providing sensory feedback in BMIs [17] |
Future Directions and Emerging Technologies
The field of neural recording continues to evolve with several promising developments. Digital holographic imaging (DHI) represents a novel non-invasive approach that detects minute neural tissue deformations (nanometer scale) occurring during neural activity [4]. This technology, initially developed for remote sensing applications, has demonstrated capability to record intracranial pressure noninvasively, suggesting potential for broader clinical monitoring applications.
Simultaneous invasive and non-invasive recordings are providing crucial validation frameworks for improving non-invasive techniques [16]. These approaches serve as a "Rosetta stone" for human neuroscience, enabling researchers to ground-truth scalp recordings against direct intracranial measures.
Closed-loop bidirectional interfaces represent another frontier, particularly for invasive BMIs. These systems not only decode motor intentions but also restore sensation through cortical stimulation, creating more naturalistic and effective neuroprosthetic systems [17]. As these technologies mature, they may significantly narrow the performance gap between biological and artificial movement control.
The progression toward miniaturized and wireless technologies will likely enhance both invasive and non-invasive approaches, making long-term monitoring more practical and reducing the encumbrances associated with current systems. For non-invasive approaches specifically, this may enable more naturalistic study of social interactions and brain-to-brain synchrony in real-world settings [22].
The selection between invasive and non-invasive neural recording techniques requires careful consideration of application-specific requirements balanced against technical capabilities and practical constraints. Invasive approaches provide superior spatiotemporal resolution and information transfer rates essential for high-performance applications like dexterous prosthetic control, but incur surgical risks and higher costs. Non-invasive techniques offer safety, accessibility, and whole-brain coverage advantages that make them suitable for diagnostic applications, basic research, and studying multi-person interactions.
Emerging technologies like digital holographic imaging may potentially expand non-invasive capabilities, while bidirectional interfaces are enhancing the functionality of invasive systems. The optimal technique selection depends fundamentally on the specific neural signals of interest, the required performance parameters, and the practical context of application. As both approaches continue to advance, their complementary strengths suggest that combined or parallel use may provide the most comprehensive understanding of brain function in health and disease.
Performance Limitations and Optimization Strategies for Enhanced Neural Recording
Addressing the Inverse Problem in Source Localization for Non-Invasive Techniques
The inverse problem in neuroimaging refers to the computational challenge of estimating the locations, directions, and magnitudes of neural currents inside the brain based on recordings taken from outside the head, such as from Electroencephalography (EEG) or Magnetoencephalography (MEG) scanners [62] [63]. This problem is mathematically ill-posed, meaning that many different configurations of brain sources can produce an identical pattern of sensor activity, making a unique solution difficult to find without imposing additional constraints [63]. Solving this problem is a cornerstone of non-invasive brain research, as it allows scientists to infer the precise spatial origin and temporal dynamics of cognitive processes with millisecond precision.
This technical guide frames the inverse problem within a broader thesis comparing the spatial and temporal resolution of invasive versus non-invasive neural recordings. While invasive techniques, such as intracranial EEG (iEEG), record electrical activity directly from the cortex and offer high signal-to-noise ratios and spatial specificity, they come with surgical risks and are limited in their coverage [17] [1]. Non-invasive techniques like EEG and MEG, on the other hand, provide a safe and practical method for measuring brain activity over the entire scalp. However, the signals are attenuated and distorted by the skull and other tissues, leading to a more challenging path to accurate source localization [17] [16]. Advances in solving the inverse problem are thus critical for bridging the resolution gap between non-invasive and invasive methods, enabling more precise insights into human brain function for research and clinical applications.
Mathematical Foundation of the Inverse Problem
The relationship between brain sources and external measurements is formulated as a linear forward model. The core equation is:
M = G × D + n
Where:
- M is the matrix of measured data at the sensors (e.g., EEG electrodes).
- G is the leadfield matrix (or gain matrix), which models how a current source at a specific location in the brain contributes to the measurement at each sensor. This matrix is derived from a head model that incorporates anatomical information from MRI scans and the electrical conductivity properties of different head tissues (brain, skull, scalp) [62] [63].
- D is the matrix of unknown dipole moments (the brain sources to be estimated), which include both magnitude and orientation.
- n represents noise in the measurements [63].
The inverse problem consists of inverting this model to find an estimate of D given M and G. The ill-posed nature of this problem arises because the number of sensors (N) is typically in the order of tens to hundreds, while the number of possible sources (p) in the brain can be thousands to millions, resulting in a system with infinitely many solutions [63]. To find a unique and physiologically plausible solution, regularization techniques and a priori constraints must be introduced.
Categories of Inverse Solution Algorithms
Inverse solutions can be broadly classified into two categories: parametric and non-parametric methods. The table below summarizes their core characteristics.
Table 1: Categories of Inverse Solution Algorithms
| Feature | Parametric Methods | Non-Parametric (Imaging) Methods |
|---|---|---|
| Core Principle | Assumes a small, pre-specified number of equivalent current dipoles. | Models the entire brain as a distributed collection of thousands of fixed dipoles. |
| Inversion Type | Non-linear, iterative search in parameter space. | Linear, direct estimation of dipole amplitudes. |
| Spatial Scope | Focal sources. | Distributed sources; can model extended cortical patches. |
| Key Algorithms | Multiple Signal Classification (MUSIC), Beamforming (e.g., LCMV) [62] [63]. | Minimum Norm Estimate (MNE), LORETA, sLORETA, LAURA [63]. |
| Advantages | High spatial resolution for well-localized sources. | No need to specify the number of sources a priori; provides a whole-brain map. |
| Disadvantages | Struggles with extended or highly correlated sources; sensitive to initial conditions. | Solutions tend to be overly smooth and exhibit low spatial resolution (blurring). |
Traditional Non-Parametric Methods
These methods reconstruct a distributed map of brain activity.
- Minimum Norm Estimate (MNE): Selects the solution with the smallest overall amplitude (L2-norm) that fits the data [63]. A key limitation is its tendency to favor superficial, radial sources near the skull while penalizing deeper sources.
- LORETA (Low Resolution Brain Electromagnetic Tomography): Builds upon MNE by adding a constraint that promotes spatial smoothness in the solution, assuming that neighboring neural populations are likely to be co-activated. While its spatial resolution is low, it has been shown to provide reliable solutions without creating false ("ghost") sources [63].
- sLORETA (standardized LORETA): Applies a statistical standardization to the LORETA solution, which theoretically allows for zero localization error under noiseless conditions for single point sources [63].
- LAURA (Local Auto-Regressive Average): Implements biophysical constraints derived from electromagnetic laws, such as the spatial decay of electric fields, leading to superior results in some performance comparisons [63].
Traditional Parametric Methods
These methods model brain activity as coming from a few focal sources.
- Beamformers (e.g., Linearly Constrained Minimum Variance - LCMV): Adaptive spatial filters that scan the brain voxel-by-voxel, designed to pass neural activity from a location of interest while suppressing activity from all other locations [62]. Their performance degrades when sources are highly correlated.
- Multiple Signal Classification (MUSIC): A scanning technique that employs signal subspace analysis to identify the dipole locations that best explain the structure of the recorded data [62] [63]. It is particularly effective for localizing multiple focal sources.
Emerging Deep Learning Approaches
Recently, deep learning (DL) has emerged as a powerful framework for solving the inverse problem. These methods learn a non-linear mapping from sensor measurements to source activities from large datasets of simulated or real brain activity.
- Architecture: DL models for M/EEG often use hybrid architectures, combining Convolutional Neural Networks (CNNs) to extract spatial-temporal features from the sensor data with fully-connected layers to perform the final source estimation [62].
- Advantages: DL approaches can incorporate both spatial and temporal information in an end-to-end fashion, potentially overcoming the limitations of linear methods. They can directly output full-brain source estimates and have shown superiority in handling deep sources and achieving higher spatial resolution in simulations [62].
- Example - Deep-MEG: One proposed model, "Deep-MEG," employs a cascade of convolutional and fully-connected layers. It operates with only the leadfield matrix and a short time window of MEG data, eliminating the need for covariance matrix estimation, which makes it robust against potential estimation errors [62].
The following diagram illustrates a generalized workflow for solving the inverse problem, encompassing both traditional and deep learning approaches.
Experimental Protocols & Validation
Validating the accuracy of inverse solutions is a fundamental challenge, as the true configuration of brain sources is rarely known. Researchers rely on a combination of simulated data, physical phantoms, and ground-truth data from invasive recordings.
Protocol for Simulation-Based Validation
This is the most common method for quantitatively comparing algorithms.
- Define "Ground Truth" Sources: Simulate one or multiple brain sources (dipoles) with known locations, orientations, and time courses. These can be focal or extended patches [62].
- Compute Forward Solution: Use a realistic head and leadfield model to calculate the simulated sensor data that would be generated by the ground truth sources [62] [63].
- Add Noise: Add realistic noise (e.g., Gaussian white noise or real brain background noise) to the simulated sensor data to mimic real-world conditions.
- Apply Inverse Algorithms: Feed the noisy simulated sensor data into various inverse algorithms (e.g., MNE, LORETA, beamformer, Deep-MEG) to estimate the sources.
- Quantitative Metrics: Compare the inverse solution to the known ground truth using metrics like:
- Localization Error: The Euclidean distance (in mm) between the true dipole location and the centroid of the estimated source activity [63].
- Spatial Resolution: The ability to distinguish between two closely spaced sources.
- Ghost Sources: The absence of spurious source activations in silent brain areas.
Protocol for Validation with Simultaneous Invasive Recordings
This method provides a crucial "Rosetta stone" for translating non-invasive signals to their true neural generators [16].
- Participant Selection: Typically performed with epilepsy patients who are undergoing pre-surgical monitoring with implanted intracranial EEG (iEEG) electrodes.
- Data Acquisition: Simultaneously record non-invasive (scalp EEG or MEG) and invasive (iEEG) brain signals.
- Identify iEEG Events: Identify specific neural events (e.g., interictal epileptic spikes or high-frequency oscillations) that are clearly visible in the iEEG recordings. The location of the iEEG electrodes providing the strongest signal serves as the ground truth source [16] [64].
- Cross-Modality Comparison: Perform source localization on the simultaneous scalp EEG/MEG data and check if the estimated source converges on the brain region identified by the iEEG electrodes.
Table 2: Key Metrics for Quantitative Performance Analysis of Inverse Solvers
| Metric | Description | Ideal Value | Typical Performance (from Literature) |
|---|---|---|---|
| Localization Error | Distance between true and estimated source center. | 0 mm | < 5-10 mm for acceptable performance [63]. |
| Spatial Resolution | Minimum distance at which two sources can be distinguished. | < 5 mm | Varies widely; deeper sources have poorer resolution. |
| Temporal Accuracy | Accuracy in reconstructing the source time-course. | Perfect correlation | High for MNE/LORETA; beamformers can distort correlated sources. |
| Robustness to Noise | Consistency of localization under different noise levels. | High | sLORETA and LAURA show good robustness [63]. |
The Scientist's Toolkit: Essential Research Reagents & Materials
Successfully conducting source localization research requires a suite of software, hardware, and data resources.
Table 3: Essential Tools for Source Localization Research
| Tool Category | Specific Examples | Function & Importance |
|---|---|---|
| Neuroimaging Hardware | MEG system (e.g., Elekta Neuromag, CTF), High-density EEG (128+ channels), MRI scanner (for anatomy) | Provides the raw, high-fidelity spatiotemporal data essential for the forward and inverse problems. MEG offers better spatial resolution for superficial cortical sources [62] [5]. |
| Head Modeling Software | FreeSurfer, SPM, FieldTrip, MNE-Python, BrainStorm | Processes anatomical MRI to create a subject-specific head model, which is critical for computing an accurate leadfield matrix [63]. |
| Inverse Algorithm Software | MNE-Python, BrainStorm, FieldTrip, SPM, LORETA-KIT | Provides open-source implementations of all major traditional inverse algorithms (MNE, dSPM, LORETA, beamformers, etc.), enabling direct comparison and application. |
| Computational Resources | High-performance CPU/GPU clusters, Cloud computing (AWS, Google Cloud) | Deep learning approaches and large-scale simulations are computationally intensive and require significant processing power [62]. |
| Validation Datasets | Simulated data, OpenNEURO [62], Simultaneous iEEG-M/EEG datasets [16] | Provide the essential ground truth for benchmarking and validating the performance of new and existing inverse solvers. |
Addressing the inverse problem remains a vibrant and critical area of research in non-invasive neuroimaging. While traditional linear methods like MNE, LORETA, and beamforming provide robust and well-understood solutions, they are inherently limited by the ill-posed nature of the problem and the smoothing constraints they impose. The emergence of deep learning offers a promising paradigm shift, leveraging data-driven models to learn complex, non-linear mappings that can potentially achieve higher spatial and temporal resolution, including for deep brain sources.
The future of this field lies in the continued development and validation of these novel algorithms, particularly through the use of simultaneous invasive and non-invasive recordings as a gold standard [16]. Furthermore, the integration of multimodal data (e.g., combining EEG/MEG with fMRI) and the adoption of personalized, anatomically precise head models will be crucial for improving accuracy. As these computational techniques evolve, they will significantly narrow the performance gap between non-invasive and invasive neural recordings, unlocking deeper and more precise insights into the dynamic functioning of the human brain for both basic research and clinical neurology.
应对信号衰减与噪声:硬件与算法解决方案
引言:神经记录中的信号衰减与噪声挑战
在神经科学研究与神经技术开发领域,信号衰减与噪声是影响数据质量与可靠性的核心挑战。无论是侵入式还是非侵入式神经记录方法,都面临着信号在传输过程中减弱以及被各种噪声源污染的问题。侵入式记录方法(如皮层内电极和深部脑刺激电极)虽能获取高时空分辨率的神经信号,但面临组织反应、免疫排斥和长期信号衰减的挑战 [65]。非侵入式方法(如EEG、fNIRS、fMRI和MEG)则因信号需穿越脑组织、颅骨和头皮而遭受严重衰减,导致空间分辨率降低和噪声干扰增加 [66]。
理解并克服这些挑战对于推进基础神经科学研究和完善临床神经技术应用都至关重要。本文系统性地分析了神经记录中信号衰减和噪声的来源,全面介绍了当前最先进的硬件解决方案和算法创新,并提供了详细的实验方案和实用工具,旨在为研究人员和工程技术人员提供一份全面的技术指南。
神经记录方式的特征比较
表1:侵入式与非侵入式神经记录技术的信号特征比较
| 记录方式 | 空间分辨率 | 时间分辨率 | 信号衰减程度 | 主要噪声源 |
|---|---|---|---|---|
| 侵入式记录 | 高(可达百微米级别) [65] | 高(毫秒级) [65] | 低(直接记录) | 热噪声,生物污染,电极组织界面噪声 [67] |
| 非侵入式EEG | 低(>5毫米) [65] | 高(毫秒级) | 高(原始脑信号的5%) [66] | 肌电,眼动,环境噪声,工频干扰 [67] [66] |
| fNIRS | 中等(1-2厘米) | 低(秒级) | 高 | 运动伪影,生理噪声 |
| fMRI | 高(毫米级) | 低(秒级) | 中等 | 运动伪影,磁场不均匀性 |
| MEG | 中等(毫米级) | 高(毫秒级) | 中等 | 环境磁场噪声,运动伪影 |
表2:神经记录中常见的噪声类型及其特征
| 噪声类型 | 来源 | 频率特征 | 影响程度 | 消除难度 |
|---|---|---|---|---|
| 生理噪声 | 心跳,肌电,眼动 | 低频(<40Hz)与高频(>50Hz) | 高 | 中等 |
| 环境噪声 | 电源线,电子设备 | 窄带(50/60Hz及其谐波) | 中等 | 低 |
| 仪器噪声 | 电极阻抗,放大器 | 全频段 | 低-中等 | 低-中等 |
| 运动伪影 | 头动,身体运动 | 低频(<5Hz) | 高 | 高 |
| 生物污染 | 电极组织界面反应 | 低频 | 高(长期植入) | 高 |
硬件创新与解决方案
侵入式记录的硬件进展
侵入式脑机接口通过将电极直接植入大脑皮层或深部组织,能够记录单个或小群神经元的电活动,获得高时空分辨率(可达百微米级别)、高信噪比的原始神经信号 [65]。然而,电极-组织界面处的信号衰减和生物污染是主要挑战。
柔性电极与微创技术:近年来,柔性材料和微创手术技术的进步显著降低了组织损伤和术后并发症 [65]。柔性微针电极的接触阻抗低于10kΩ,能显著抑制运动伪影,提升信号质量 [65]。这些电极尺寸仅为早期产品的1/5,柔性提升百倍,植入体直径仅26毫米,极大减少组织损伤 [65]。
无线供能与数据传输:无线供能技术减少体内外硬件依赖,提升患者舒适度和长期稳定性 [65]。高速无线数据传输支持实时高精度信号解码和远程医疗监控,解决了传统有线系统的移动限制和感染风险。
非侵入式记录的硬件进展
非侵入式脑机接口通过在头皮表面放置电极帽或传感器来采集大脑信号,无需手术,但信号需穿越头皮和颅骨,导致信号强度和空间分辨率受限 [65]。
材料与传感器创新:柔性微针电极和电子文身墨水的应用,显著提升了佩戴舒适性和信号稳定性 [65]。这些材料可直接贴附于皮肤,改善传统干电极的接触阻抗问题。
多模态融合系统:EEG与fNIRS等多模态联合采集系统通过结合电生理和血流动力学信息,提供更全面的脑活动视角,分类准确率可提升18.7% [65]。这种多模态方法能克服单一模态的局限性,提高信号解码的可靠性。
算法与数据处理创新
信号增强与去噪算法
基于深度学习的去噪方法:深度学习模型如卷积神经网络(CNN)、循环神经网络(RNN)和深度信念网络(DBN)被广泛用于特征提取和分类,尤其是在睡眠阶段识别、运动想象任务、情绪状态检测和神经系统疾病诊断中 [66]。这些网络通过深度结构捕获代表性的高级特征和潜在的依赖关系,直接处理原始大脑信号,从而避免耗时的预处理和特征提取 [66]。
表3:常用去噪算法性能比较
| 算法类型 | 适用信号 | 计算复杂度 | 去噪效果 | 实时性 |
|---|---|---|---|---|
| 传统滤波 | EEG,LFP | 低 | 中等 | 高 |
| 自适应滤波 | EEG,EMG | 中等 | 中高 | 中等 |
| 独立成分分析 | EEG,MEG | 高 | 高 | 低 |
| 小波变换 | EEG,fMRI | 中高 | 高 | 中等 |
| 深度学习 | 所有信号类型 | 高 | 最高 | 依赖硬件 |
混合去噪框架:节能的去噪方法将图像增强和K-means聚类作为预处理技术相结合,提高应用神经网络之前的输入质量 [68]。在应用卷积自动编码器之前,集成了使用锐化核的图像增强和通过K-means聚类进行图像分割 [68]。预处理步骤使模型能够识别解剖边界并分离受噪声影响的区域,从而提高输入质量并增强训练收敛性,将训练时间减少约20%,并降低GPU利用率 [68]。
跨模态信号融合与增强
联合LFP-EEG记录协议:结合侵入式(局部场电位LFP)和非侵入式(脑电图EEG)人脑记录的实验协议能同时评估基底神经节的认知情绪功能 [69]。这种联合记录方法可以时间-频率耦合的基础上建立脑振荡和行为之间的不同关系的分析,为患者的个性化认知提供潜在生物标志物 [69]。
基于机器学习的噪声预测:在磁共振成像领域,通过机器学习模型训练梯度波形与噪声的响应关系,可对不同序列的梯度波形进行噪声预测 [70]。该方法获取磁共振扫描过程中的梯度波形及对应的声压波形,对数据进行预处理后,使用卷积神经网络模型、循环神经网络模型或深度神经网络模型进行训练,得到梯度波形与噪声的响应关系 [70]。
实验方案与流程
联合侵入式与非侵入式神经记录协议
以下协议旨在通过同时对局部场电位和非侵入性脑皮层活动(EEG)进行神经生理学记录来评估基底神经节的认知情绪功能 [69]:
实验范式设计:
- 选择涉及情感内涵的语音刺激或涉及认知控制的Flanker任务作为实验范式 [69]。
- 确定刺激呈现顺序(块或混合设计),设定刺激持续时间、刺激间隔和试验次数 [69]。
- 对于Flanker任务,包括兼容条件(60%)、不兼容条件(20%)和停止试验(20%),随机呈现刺激 [69]。
记录设置:
- 建立EEG设备,设置采样频率(如5 kHz),低截止频率(DC)和高截止频率(1000 Hz) [69]。
- 根据国际10/20系统配置EEG通道,至少包括Fz、Cz、FPz和乳突参考电极 [69]。
- 配置LFP通道(左半球:LFPL0、LFPL1、LFPL2、LFPL3;右半球:LFPR0、LFPR1、LFPR2、LFPR3) [69]。
信号处理与分析:
- 对EEG和LFP信号进行时间-频率分析。
- 比较不同实验条件下的大脑振荡活动。
- 分析皮层-皮层下信号耦合与行为表现的关系。
节能医学图像去噪流程
该协议为节能医学图像去噪提供了一个可重复的管道,适用于神经影像数据 [68]:
数据集准备:
- 使用公开可用的、去识别化的CT和MRI成像数据集 [68]。
- 将每张图像的大小调整为256 x 256像素,并以PNG或DICOM格式存储 [68]。
- 将数据集随机分配为70%用于训练,15%用于验证,15%用于测试 [68]。
图像预处理:
去噪模型训练:
- 构建基于CNN的自动编码器架构。
- 使用预处理后的数据训练模型。
- 通过峰值信噪比(PSNR)和结构相似性指数(SSIM)评估性能 [68]。
研究工具与可视化
研究试剂解决方案
表4:神经记录与信号处理关键研究工具
| 工具类别 | 具体示例 | 主要功能 | 适用场景 |
|---|---|---|---|
| 记录设备 | EEG电极帽,深部脑刺激电极 | 神经信号采集 | 侵入式/非侵入式记录 |
| 信号放大器 | 生物电位放大器,颅内信号放大器 | 信号放大 | 所有神经记录场景 |
| 预处理软件 | EEGLAB,FieldTrip,MNE-Python | 信号预处理与可视化 | 非侵入式信号分析 |
| 深度学习框架 | TensorFlow,PyTorch | 信号去噪与解码 | 复杂信号处理任务 |
| 数据分析工具 | MATLAB,Python | 信号分析与统计 | 所有研究场景 |
技术流程可视化
讨论与未来方向
应对神经记录中信号衰减与噪声的挑战需要多学科协同创新。硬件方面的进步,特别是柔性电极和无线传输技术,已经显著改善了信号采集质量并减少了运动伪影 [65]。算法方面的创新,尤其是深度学习方法和多模态融合策略,提高了从噪声环境中提取有用神经信息的能力 [66] [68]。
未来研究应当聚焦于以下几个方向:一是开发更加生物相容的长期植入材料,减少组织反应和信号衰减;二是创建更高效的节能算法,使复杂信号处理能在便携设备上实时运行;三是建立标准化协议和数据集,促进不同研究之间的比较和算法验证。
随着神经技术的不断进步和应用的扩展,解决信号衰减和噪声问题将继续是推动该领域发展的关键。通过硬件和算法的协同优化,我们有望在不久的将来实现更加可靠、高精度的神经记录系统,为基础研究和临床应用开辟新的可能性。
Strategies for Improving Signal-to-Noise Ratio in Electrically Noisy Environments
In neuroscience, the fidelity of neural recordings is paramount for decoding brain activity, and the signal-to-noise ratio (SNR) is a critical determinant of data quality. This metric, which compares the level of a desired signal to the level of background noise, directly impacts the accuracy of interpreting neural dynamics [71]. The challenge of achieving a high SNR is intrinsically linked to the ongoing research on the spatial and temporal resolution of neural recording techniques. Invasive methods, such as electrocorticography (ECoG) and micro-electrocorticography (µECoG), offer high spatial resolution and SNR by placing electrodes directly on the cortical surface, capturing fast neural dynamics at a millimeter or sub-millimeter scale [72]. Non-invasive methods, like magnetoencephalography (MEG) and functional magnetic resonance imaging (fMRI), provide a safer alternative for human studies but are fundamentally constrained by a trade-off; fMRI offers millimeter spatial resolution but tracks slow hemodynamic changes, while MEG provides millisecond temporal precision but suffers from poor spatial detail [35]. Improving SNR is thus not merely a technical exercise but a necessary endeavor to bridge this resolution gap, enhancing our ability to study brain function with greater clarity from both invasive and non-invasive perspectives. This guide outlines core strategies, experimental validations, and practical toolkits for researchers aiming to improve SNR in electrically noisy environments.
A multi-faceted approach is required to enhance SNR, encompassing hardware design, signal processing, and advanced modeling. The following table summarizes the primary strategies available to researchers.
Table 1: Core Strategies for Improving Signal-to-Noise Ratio
| Strategy Category | Specific Technique | Underlying Principle | Key Consideration for Neural Recordings |
|---|---|---|---|
| Hardware & Acquisition | High-Density Micro-Scale Electrodes (e.g., µECoG) | Increases spatial resolution and electrode density, capturing finer neural signals and providing a higher inherent SNR [72]. | Invasive procedure; requires surgical implantation. |
| Shielding & Grounding | Uses conductive materials to block external electromagnetic interference and provides a path for stray currents to minimize noise [71]. | Essential for all recording types; critical in environments with heavy machinery or radio frequency sources. | |
| Differential Signaling & Twisted-Pair Cables | Transmits two complementary signals; noise induced along the cable is canceled at the receiver, rejecting common-mode interference [73] [71]. | Effective for reducing noise in analog signal transmission over longer distances. | |
| Optimizing Excitation Levels | Increases the amplitude of the source signal before noise is introduced, improving the raw SNR [74]. | Must be balanced against side effects like self-heating in strain gauges or tissue damage in neural stimulators. | |
| Signal Processing | Ensemble Averaging | Repeats the signal acquisition and averages the results, reinforcing the true signal while random noise averages toward zero [73]. | Only applicable to reproducible signals; reduces effective data rate. |
| Filtering (Hardware & Software) | Uses low-pass, high-pass, band-pass, or notch filters to remove noise outside the frequency band of interest [75] [71]. | Digital filtering post-acquisition allows for sophisticated methods like adaptive filtering and wavelet transforms. | |
| Regenerative Circuits (Q-multiplier) | An active filter that uses positive feedback to reinforce a signal hidden within the noise, effectively increasing gain [73]. | An older technique from radio, can be applied to specific signal types. | |
| Computational & Modeling | Advanced Deep Learning Architectures | Uses neural networks with parallel attention mechanisms (e.g., CT-ParaNet) to hierarchically extract features and suppress noise in the feature space [76]. | Requires large datasets for training; computationally intensive but highly effective for pattern recognition in noise. |
| Multi-Modal Data Fusion | Integrates data from different modalities (e.g., MEG and fMRI) in an encoding model to estimate a high-resolution source signal that is faithful to both temporal and spatial features [35]. | Aims to overcome the inherent limitations of individual non-invasive techniques. |
Experimental Protocols for SNR Validation
To ensure that SNR improvement strategies are effective, rigorous experimental validation is required. The following protocols detail methodologies cited in recent literature.
Protocol: Validating High-Density µECoG for Speech Decoding
This protocol is adapted from studies demonstrating that high-density µECoG arrays significantly improve speech decoding accuracy by providing a higher SNR and spatial resolution [72].
- Objective: To quantify the improvement in SNR and decoding accuracy achieved by µECoG compared to standard intracranial electrodes.
- Materials:
- Liquid crystal polymer thin-film (LCP-TF) µECoG arrays (e.g., 128 or 256 channels with 1.33-1.72 mm inter-electrode distance).
- Standard macro-ECoG or stereoelectroencephalography (SEEG) electrodes for comparison.
- Intraoperative neural recording system with impedance measurement capability.
- Audio recording equipment for synchronized speech capture.
- Methodology:
- Patient Task: Participants (e.g., undergoing surgery for tumor resection or DBS implantation) perform a speech repetition task, listening to and repeating auditory non-words (CVC or VCV tokens).
- Data Acquisition: Neural activity is recorded from the speech motor cortex (SMC) using the µECoG array. Simultaneously, the participant's spoken output is recorded to align neural data with speech production.
- Signal Processing: Compute the high-gamma band (HG, 70-150 Hz) power, a key marker for local neural activity, from the recorded signals.
- SNR Calculation: Calculate the Evoked-Signal-to-Noise Ratio (ESNR) for the HG power. This is typically done by comparing the power during the speech utterance epoch (e.g., -500 ms to +500 ms around utterance onset) to a pre-stimulus baseline period.
- Decoding Analysis: Train a machine learning model (e.g., a non-linear decoder) to predict the spoken phonemes from the HG neural activations. Use cross-validation to assess accuracy.
- Validation Metric: Compare the ESNR and phoneme decoding accuracy of the µECoG array directly against the performance achievable with simulated or concurrently recorded standard IEEG signals. The study reported a 48% higher SNR and 35% improved decoding accuracy with µECoG [72].
Protocol: Testing a Channel-Time Parallel Attention Network (CT-ParaNet)
This protocol tests a modern deep-learning approach for maintaining high diagnostic accuracy in extremely noisy conditions, as applied to bearing fault diagnosis [76]. The principles are directly transferable to neural signal analysis.
- Objective: To evaluate the robustness of a CT-ParaNet model in classifying signals under different types and levels of noise.
- Materials:
- Dataset of labeled neural signals (e.g., different brain states or stimulus responses) or vibration signals for validation.
- Computing platform with GPU acceleration for deep learning.
- Software for generating Gaussian white noise, impulse noise, and colored noise.
- Methodology:
- Data Preparation: Split the dataset into training, validation, and test sets.
- Model Architecture: Implement the CT-ParaNet, which features:
- A Channel-Time Parallel Attention Mechanism that computes feature correlations in channel and temporal dimensions simultaneously, avoiding information loss from serial processing.
- Multi-scale parallel attention residual blocks to capture features at different scales.
- An adaptive mixing pooling strategy to balance between preserving salient features and maintaining global information.
- Noise Introduction: Artificially corrupt the test signals with various types of noise (Gaussian white, impulse, colored) at different SNR levels, including extreme conditions (e.g., -5 dB).
- Training & Evaluation: Train the model on clean or moderately noisy data. Evaluate its classification accuracy on the noisy test set and compare it against traditional models.
- Validation Metric: Classification accuracy under varying noise conditions and SNR levels. The cited study showed CT-ParaNet maintained accuracy above 87% even at -5 dB SNR [76].
Visualization of SNR Improvement Workflows
The following diagrams illustrate the logical relationships and workflows for the key strategies discussed.
Hierarchical SNR Improvement Pathway
This diagram outlines a structured, hierarchical approach to tackling noise, from physical hardware to computational intelligence.
Multi-Modal Data Fusion for Enhanced Resolution
This workflow shows how combining non-invasive modalities can estimate brain activity with higher spatiotemporal resolution, overcoming the inherent SNR and resolution trade-off.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of SNR strategies requires specific tools and materials. The following table details key components for a modern research pipeline.
Table 2: Key Research Reagents and Materials for SNR-Conscious Neural Recording
| Item Name | Function/Application | Specific Example/Specification |
|---|---|---|
| High-Density µECoG Array | Invasive neural interface for capturing fine-grained cortical activity with high SNR and spatial resolution. | Liquid crystal polymer thin-film (LCP-TF) arrays with 128-256 channels, inter-electrode distance of 1.33-1.72 mm, and exposed electrode diameter of 200 µm [72]. |
| Low-Noise Amplifier & Data Acquisition System | Amplifies weak neural signals while introducing minimal additional noise, crucial for preserving SNR. | Systems with a high Spurious Free Dynamic Range (SFDR, e.g., >100 dB) and 24-bit analog-to-digital converters for high resolution on small input ranges [74]. |
| Shielded Enclosure & Cabling | Physically blocks external electromagnetic interference from contaminating the neural signal during acquisition. | Conductive metal enclosures (Faraday cages) for equipment and shielded, twisted-pair cables for signal transmission [71]. |
| CT-ParaNet Model Architecture | A deep learning model for robust feature extraction and classification from signals heavily corrupted by noise. | Features a parallel attention mechanism, multi-scale residual blocks, and adaptive pooling. Implemented in PyTorch/TensorFlow [76]. |
| Multi-Modal Encoding Model Software | Software framework for fusing MEG and fMRI data to estimate high-fidelity source activity. | Transformer-based encoding model trained to predict both MEG and fMRI from stimuli, yielding a latent source estimate [35]. |
| py_neuromodulation Platform | An open-source software for standardized, reproducible analysis of invasive brain signals, facilitating feature extraction and decoding. | A modular Python platform for extracting oscillatory dynamics, waveform shape, and connectivity features for machine learning [26]. |
The fundamental trade-off between spatial resolution and invasiveness defines modern neural recording research. Invasive techniques, such as intracranial electroencephalography (iEEG) and microelectrode arrays (MEAs), provide direct neural access with high spatial resolution, while non-invasive methods like electroencephalography (EEG) and magnetoencephalography (MEG) offer broader applicability at the cost of resolution [16] [77]. This technical guide examines how emerging technologies—specifically high-density arrays and sophisticated multimodal data fusion strategies—are bridging this divide, enabling unprecedented insights into neural function across scales.
High-density microelectrode arrays (HD-MEAs) represent a paradigm shift, with recent planar devices featuring 236,880 electrodes within a 5.51 × 5.91 mm² sensing area, achieving electrode densities exceeding 3000 per mm² and enabling simultaneous readout of 33,840 channels at 70 kHz [30]. These platforms facilitate recordings spanning subcellular compartments to entire networks across temporal scales from microseconds to months. Complementary advances in non-invasive decoding demonstrate that sophisticated deep learning pipelines can now decode individual words from M/EEG signals across hundreds of participants, achieving up to 37% top-10 accuracy [5]. The convergence of these recording technologies with multimodal fusion methodologies creates a powerful framework for overcoming fundamental resolution barriers in neuroscience research and therapeutic development.
High-Density Array Technologies: From Design to Application
HD-MEA Chip Design and Specifications
The evolution of HD-MEA technology has been driven primarily by innovations in complementary metal-oxide-semiconductor (CMOS) fabrication, enabling miniaturization of electrodes and direct on-chip integration of electronic components such as filters, amplifiers, and analog-to-digital converters (ADCs) [30]. This integration resolves the "connectivity problem" of traditional passive MEAs while significantly enhancing channel count, array area, spatial density, and signal-to-noise ratio (SNR) through reduced parasitic capacitance and resistive losses.
Table 1: Key Design Specifications and Trade-offs in HD-MEA Development
| Design Parameter | Specifications & Options | Performance Implications |
|---|---|---|
| Technology Node | Smaller nodes (90nm, 65nm, 40nm) vs. older nodes (130nm, 180nm, 350nm) | Advanced digital processing vs. analog optimization and cost |
| Electrode Density | >3000 electrodes per mm² [30] | Higher spatial resolution for subcellular monitoring |
| Array Size | Up to ~32.5 mm² (5.51 × 5.91 mm) sensing area [30] | Balance between coverage area and spatial resolution |
| Channel Count | Simultaneous readout of 33,840 channels at 70 kHz [30] | Increased data volume and processing requirements |
| Readout Strategy | Full-frame vs. partial readout | Comprehensive sampling vs. manageable data volumes |
Design optimization requires careful balancing of these parameters against application-specific requirements. Research focusing on subcellular resolution may prioritize extreme density over large array size, while network-level studies might sacrifice some density for broader coverage [30]. Similarly, the choice between full-frame and partial readout strategies involves trade-offs between data comprehensiveness and computational load, with partial readout reducing data volume but potentially missing critical information from unmonitored array regions.
Advanced Non-Invasive Arrays and Hyperscanning
Beyond invasive arrays, non-invasive technologies have also achieved density breakthroughs. High-density EEG (HD-EEG) systems now feature 256+ electrodes, significantly improving spatial sampling for source localization. Simultaneously, hyperscanning approaches—recording from multiple brains simultaneously—have emerged as powerful tools for studying social interactions and brain-to-brain synchrony using EEG, MEG, or functional near-infrared spectroscopy (fNIRS) [22]. These non-invasive array applications employ neural connectivity measures like phase-locking value (PLV) and wavelet transform coherence (WTC) to quantify inter-brain synchronization during tasks ranging from guitar playing and conversation to classroom learning [22].
Multimodal Data Fusion Strategies for Enhanced Neural Decoding
Fusion Techniques and Architectures
Multimodal data fusion integrates information from disparate neural recording modalities to create unified representations that overcome the limitations of individual techniques. The three primary fusion strategies—early, intermediate, and late fusion—offer distinct advantages and limitations for neural applications [78] [79] [80].
Table 2: Multimodal Data Fusion Techniques for Neural Data Integration
| Fusion Technique | Implementation Approach | Advantages | Limitations |
|---|---|---|---|
| Early Fusion (Data-Level) | Combine raw/low-level data before feature extraction [78] [80] | Preserves comprehensive information; enables learning of joint representations | Requires precise temporal/spatial alignment; sensitive to noise/missing data |
| Intermediate Fusion (Feature-Level) | Combine extracted features into joint representation [79] [80] | Balances modality-specific processing with joint learning; captures cross-modal interactions | Requires all modalities present; complex model architecture |
| Late Fusion (Decision-Level) | Combine outputs from independently processed modalities [78] [79] | Handles asynchronous/missing data; leverages specialized unimodal models | May miss subtle cross-modal interactions; limited joint representation learning |
Advanced fusion architectures increasingly employ transformer-based models with cross-attention mechanisms, enabling dynamic weighting of feature relevance across modalities [79] [80]. These approaches create shared embedding spaces where neural features from different recording techniques can be directly compared and integrated. For neural decoding applications, intermediate fusion has demonstrated particular promise, balancing modality-specific processing with cross-modal integration to enhance decoding accuracy for tasks such as speech reconstruction and intent recognition [5] [81].
Experimental Evidence and Performance Metrics
Recent large-scale validation studies demonstrate the power of multimodal fusion for neural decoding. One investigation across 723 participants reading or listening to 5 million words across three languages showed that deep learning pipelines with transformer architectures significantly outperformed linear models, with accuracy further scaling with training data volume and test-time averaging [5]. The study revealed important protocol dependencies: MEG outperformed EEG for decoding, while reading tasks proved easier to decode than listening tasks, highlighting how modality-specific characteristics influence fusion strategy effectiveness.
Simultaneous invasive and non-invasive recordings provide a "Rosetta stone" for validating fusion approaches, offering ground truth correlations between high-resolution intracranial signals and scalable non-invasive measurements [16]. In epilepsy monitoring, for example, simultaneous iEEG and HD-EEG recordings have advanced understanding of seizure networks while improving source localization accuracy, creating foundational knowledge for developing more effective non-invasive biomarkers [16].
Experimental Protocols and Methodological Guidelines
Protocol 1: Multimodal Language Decoding from Non-Invasive Recordings
Objective: Decode individual words from M/EEG recordings during language processing tasks. Equipment: MEG system (e.g., 306-channel Triux) or high-density EEG system (128+ electrodes), stimulus presentation software, eye-tracking for artifact control. Stimuli: Carefully controlled word sets presented auditorily or visually with precise timing. Procedure:
- Data Acquisition: Record M/EEG while participants read or listen to sentences, with precise word-onset markers.
- Preprocessing: Apply standard preprocessing (filtering, artifact removal, baseline correction) and extract trials time-locked to word onsets.
- Feature Extraction: Compute relevant neural features (time-series, frequency power, connectivity measures) for each modality.
- Model Training: Implement a deep learning architecture with subject-specific layers and transformer components for cross-modal context integration.
- Validation: Evaluate using cross-validation and compute top-k accuracy metrics on held-out test sets [5].
Key Parameters: Trial counts ≥1000 per participant, sampling rates ≥1000Hz, precise stimulus-response alignment.
Protocol 2: Simultaneous Invasive and Non-Invasive Recording Validation
Objective: Establish correlation between invasive and non-invasive neural signals to validate fusion approaches. Equipment: Simultaneous iEEG/ECoG and HD-EEG/MEG recording systems, synchronized acquisition hardware. Participants: Epilepsy patients with clinically indicated intracranial monitoring. Procedure:
- Synchronized Setup: Precisely time-synchronize invasive and non-invasive recording systems.
- Stimulus Presentation: Administer controlled sensory, motor, or cognitive tasks.
- Parallel Recording: Acquire simultaneous data from both recording modalities.
- Signal Processing: Apply matched filtering and preprocessing pipelines.
- Cross-Modal Mapping: Establish quantitative relationships between iEEG features and HD-EEG/MEG signatures using regression models and cross-correlation analyses [16].
Key Analysis: Compare source localization accuracy between HD-EEG alone and HD-EEG informed by iEEG ground truth.
Protocol 3: Cellular-Level Network Analysis with HD-MEA
Objective: Characterize functional network dynamics in vitro at single-cell resolution. Equipment: CMOS-based HD-MEA system with thousands of simultaneously readable electrodes, in vitro preparations (cell cultures, brain slices). Procedure:
- Sample Preparation: Plate electrogenic cells (neurons, cardiomyocytes) on HD-MEA or prepare acute brain slices.
- Recording: Conduct large-scale extracellular voltage recordings across the array.
- Spike Detection: Apply threshold-based or template-matching algorithms to detect action potentials.
- Network Analysis: Compute functional connectivity metrics (cross-correlation, transfer entropy) from spike trains.
- Perturbation Studies: Use integrated stimulation capabilities to probe network resilience [30].
Key Advantage: HD-MEAs enable tracking AP propagation along individual axons and dendrites while simultaneously monitoring network-scale dynamics.
Visualization Frameworks and Workflows
Multimodal Fusion Strategy Decision Framework
This decision framework guides researchers in selecting appropriate fusion strategies based on their specific data characteristics and research objectives, balancing implementation complexity with performance requirements.
Simultaneous Invasive/Non-Invasive Recording Validation Workflow
This workflow outlines the comprehensive process for acquiring and validating simultaneous invasive and non-invasive recordings, highlighting the critical stages from ethical preparation through to analytical validation.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Toolkit for High-Density Neural Recording and Fusion Studies
| Tool/Reagent | Specification | Research Function |
|---|---|---|
| CMOS HD-MEA Chips | 236,880 electrodes, 33,840 simultaneous channels [30] | Cellular-scale electrophysiology in vitro |
| High-Density EEG Systems | 256+ electrode caps, high-input impedance amplifiers | Scalp-level neural dynamics with enhanced spatial sampling |
| MEG Systems | 306-channel (204 planar gradiometers, 102 magnetometers) | Non-invasive recording of magnetic brain activity |
| iEEG/ECoG Grids | High-density subdural grids (e.g., 256-contact) | Clinical intracranial recording with high SNR |
| Synchronization Hardware | Multi-system trigger interfaces, shared clock signals | Temporal alignment of multimodal data streams |
| Deep Learning Frameworks | Transformer architectures, contrastive learning modules [5] [79] | Multimodal representation learning and decoding |
| Data Fusion Toolboxes | Early, intermediate, and late fusion implementations | Flexible implementation of fusion strategies |
The synergistic combination of high-density arrays and multimodal data fusion represents a transformative approach to overcoming fundamental resolution barriers in neural recording. HD-MEAs provide unprecedented spatial resolution at microscopic scales, while sophisticated fusion methodologies enable the integration of complementary information across recording modalities. Together, these technologies create a powerful framework for advancing both basic neuroscience and therapeutic development.
Future progress will likely come from several emerging frontiers. Continued CMOS miniaturization will push electrode densities even higher, while novel non-invasive sensors like optically pumped magnetometers (OPMs) offer more flexible MEG recording [81]. Computational advances in transformer architectures and foundation models pretrained on large-scale neural datasets will enhance cross-modal transfer learning [79] [80]. Most importantly, increased collection of simultaneous multimodal datasets will provide the essential "Rosetta stones" needed to validate and refine fusion approaches [16]. Through continued innovation across these technical domains, the field moves closer to a comprehensive understanding of neural function across spatial and temporal scales, ultimately enabling more effective diagnostics and therapies for neurological disorders.
Mitigating Long-Term Stability Challenges in Chronic Invasive Implants
The choice between invasive and non-invasive neural recording techniques represents a fundamental trade-off between signal quality and long-term stability. Invasive techniques, which involve surgically implanting electrodes directly into brain tissue, provide superior spatial and temporal resolution and a higher signal-to-noise ratio (SNR) compared to non-invasive methods [1]. This high-fidelity signal is crucial for complex applications such as neuroprosthetics. However, these benefits come with significant long-term stability challenges, including foreign body response, electrode degradation, and signal quality deterioration over time [1]. Understanding and mitigating these challenges is paramount for advancing the reliability and lifespan of chronic invasive neural implants, enabling their successful translation from research laboratories to clinical applications.
Core Mechanisms of Implant Failure
Chronic invasive implants face multiple interconnected biological and technical challenges that compromise their long-term functionality and stability.
Biological Response and Interface Stability
The foreign body response triggers a cascade of biological events that ultimately lead to implant encapsulation. Upon implantation, protein adsorption occurs immediately, followed by inflammatory cell migration (including neutrophils and macrophages) to the injury site. This inflammatory process leads to the formation of a glial scar around the implant, comprising activated microglia and astrocytes. Ultimately, the body forms a fibrous encapsulation layer that electrically isolates the electrode from target neurons. This increasing distance between recording sites and neurons significantly attenuates signal amplitude and increases electrical impedance over time.
Material Degradation and Mechanical Mismatch
The stability of the neural interface is critically dependent on the material properties of the implant and their compatibility with neural tissue. Key degradation mechanisms include:
- Corrosion and Delamination: Metallic electrodes and conductive traces are susceptible to electrochemical corrosion in the saline-rich neural environment, while multilayer implants experience delamination of insulating layers [1].
- Mechanical Mismatch: The significant stiffness difference between conventional implant materials (e.g., silicon, metals) and soft brain tissue (elastic modulus ~1 kPa) creates micromotions that continuously irritate surrounding tissue, exacerbating the foreign body response.
- Insulation Failure: Polymeric insulation materials can degrade through hydrolysis or enzymatic activity, leading to current leakage and reduced recording performance.
Table 1: Primary Failure Mechanisms in Chronic Invasive Implants
| Failure Mechanism | Impact on Function | Temporal Progression |
|---|---|---|
| Foreign Body Response (Glial Scarring) | Increased electrode impedance, Signal attenuation | Acute (days-weeks), Chronic (months-years) |
| Electrode Degradation / Corrosion | Reduced charge transfer capacity, Increased noise | Progressive (months-years) |
| Mechanical Mismatch & Micromotion | Chronic inflammation, Tissue damage | Continuous |
| Insulation Failure | Signal cross-talk, Current leakage | Progressive (months-years) |
Material and Engineering Solutions
Advanced materials and innovative engineering approaches have emerged to address the core challenges of chronic implant stability.
Surface Modifications and Biofunctionalization
Surface properties significantly influence the biological response to implanted materials. Strategic surface modifications can mitigate unfavorable immune reactions and promote seamless integration:
- Surface Topography: Engineering surface features at multiple scales (macro, micro, and nano) can direct cellular responses. Rough surfaces enhance tissue integration but may promote bacterial colonization, while smooth surfaces demonstrate more favorable outcomes in managing inflammatory conditions [82].
- Bioactive Coatings: Immobilizing biomolecules such as laminin, collagen, or neurotrophic factors onto implant surfaces can promote neuronal attachment and inhibit glial cell proliferation. These coatings create a more favorable microenvironment for neural integration.
- Antifouling Coatings: Grafting hydrophilic polymers like polyethylene glycol (PEG) or applying hydrogel coatings resists protein adsorption and reduces inflammatory cell adhesion.
- Drug-Eluting Systems: Incorporating anti-inflammatory agents (e.g., dexamethasone) into polymer coatings allows localized, controlled release to modulate the inflammatory response.
Novel Material Platforms
Emerging material systems offer promising alternatives to conventional rigid implants:
- Flexible and Soft Substrates: Using polyimide, parylene, or elastomeric materials reduces the mechanical mismatch with neural tissue. Recent advances include ultraflexible mesh electrodes and conductive polymer-based arrays that minimize tissue disruption.
- Conductive Polymers: Materials like PEDOT:PSS offer improved electrochemical properties, lower impedance, and better biocompatibility compared to traditional metals. Their soft, ionic conduction mechanism provides a more compliant neural interface.
- Dissolvable Platforms: Temporary implants using biodegradable materials (e.g., silicon, magnesium) that dissolve after a designated service period eliminate long-term foreign body risks and the need for explantation surgery.
Table 2: Material Solutions for Enhanced Chronic Stability
| Material Approach | Key Advantages | Limitations & Challenges |
|---|---|---|
| Nanotextured Surfaces | Enhanced neural integration, Reduced glial scarring | Standardization of fabrication, Long-term stability of coatings |
| Conductive Polymers (PEDOT) | Lower impedance, Better mechanical compliance, Improved charge injection | Long-term stability under electrical stimulation, Adhesion to substrates |
| Flexible Polymer Substrates | Reduced mechanical mismatch, Minimized tissue damage | Handling during implantation, Potential for delamination |
| Hydrogel Coatings | Tissue-like mechanical properties, Drug delivery capability | Swelling-induced stress, Potential for increased impedance |
Experimental Validation and Assessment Methodologies
Rigorous experimental protocols are essential for evaluating the long-term performance and stability of neural implants across spatial and temporal dimensions.
In Vitro Characterization Protocols
Electrochemical Impedance Spectroscopy (EIS)
- Purpose: Monitor electrode integrity and interface stability
- Protocol: Measure impedance spectrum from 1 Hz to 100 kHz at regular intervals
- Parameters: Electrode impedance at 1 kHz (indicative of recording capability), phase angle, and Bode plot analysis
- Accelerated Aging: Subject implants to elevated temperature (e.g., 67°C) in phosphate-buffered saline to simulate long-term degradation
Cyclic Voltammetry for Charge Storage Capacity
- Purpose: Quantify charge injection capacity and electrochemical stability
- Protocol: Perform voltage sweeps (typically -0.6 V to 0.8 V vs. Ag/AgCl) at scan rates of 50 mV/s
- Analysis: Calculate charge storage capacity from integrated cathodic current, monitor for redox peaks indicating material degradation
In Vivo Assessment Methods
Chronic Neural Recording in Rodent Models
- Surgical Procedure: Sterotaxic implantation of neural probes under anesthesia using aseptic technique
- Longitudinal Tracking: Record neural signals regularly over implant duration (weeks to months)
- Signal Metrics: Quantify single-unit yield, signal-to-noise ratio, spike amplitude, and local field potential power spectra over time
- Control Experiments: Compare different material systems or surface modifications within the same subject using bilateral implants
Histological Analysis for Tissue Response
- Perfusion and Tissue Processing: Transcardial perfusion with paraformaldehyde at experimental endpoint
- Immunohistochemistry: Label neurons (NeuN), astrocytes (GFAP), microglia (Iba1), and macrophages (CD68)
- Quantitative Analysis: Calculate neuronal density at increasing distances from implant, glial scar thickness, and cellular density in peri-implant region
- Correlative Analysis: Relate histological findings with electrophysiological performance metrics
The experimental workflow for validating implant stability integrates these methodologies systematically, as visualized below:
The Scientist's Toolkit: Research Reagent Solutions
Successful research on chronic implant stability requires specialized materials and assessment tools. The following table details essential research reagents and their applications in developing and validating stable neural interfaces.
Table 3: Essential Research Reagents for Neural Implant Studies
| Reagent / Material | Function & Application | Technical Considerations |
|---|---|---|
| Neuropixels Probes | High-density neural recording; Stability assessment across brain regions [6] [83] | NXT version offers more compact design; Enables large-scale electrophysiology |
| Conductive Polymers (PEDOT:PSS) | Electrode coating to improve interface properties; Lower impedance & better integration [84] | Electro-polymerization parameters critical; Stability varies with deposition method |
| Anti-inflammatory Agents (Dexamethasone) | Modulate foreign body response; Localized delivery from coatings [84] | Controlled release kinetics important; Dose optimization required |
| Immunohistochemistry Antibodies (GFAP, Iba1, NeuN) | Quantify tissue response; Label astrocytes, microglia, neurons [83] | Multiplex labeling enables comprehensive analysis; Quantification methods vary |
| Flexible Polymer Substrates (Polyimide, Parylene) | Reduce mechanical mismatch; Minimize tissue damage [83] | Young's modulus should match neural tissue; Handling requires specialized tools |
| Functional Ultrasound (fUS) | Non-invasive monitoring of implant effect on brain activity [85] | Emerging modality; Provides hemodynamic correlates of inflammation |
| Bioactive Peptides (RGD, Laminin) | Promote neuronal attachment; Enhance integration[ccitation:1] | Immobilization chemistry critical; Stability in biological environment varies |
Mitigating long-term stability challenges in chronic invasive implants requires a multidisciplinary approach addressing biological, material, and engineering factors. The fundamental trade-offs between spatial/temporal resolution and chronic stability continue to drive innovation in neural interface technology. As research advances, the integration of novel materials, sophisticated surface engineering, and comprehensive validation protocols will enable the development of next-generation implants with extended functional longevity and improved clinical outcomes.
Comparative Analysis and Validation Metrics Across Neural Recording Modalities
The field of neural recording is fundamentally divided by a critical trade-off: the choice between invasive techniques, which provide high-fidelity data by accessing the brain directly, and non-invasive techniques, which offer safety and accessibility by measuring neural activity from outside the skull. This technical guide provides an in-depth comparison of these methodologies, focusing on their core performance metrics—spatial resolution, temporal resolution, and signal-to-noise ratio (SNR)—within the broader research context of understanding brain function. These metrics are paramount for researchers and drug development professionals who rely on precise neural data to study brain dynamics, validate therapeutic interventions, and develop neurotechnologies. The inherent limitations of any single method have driven innovation in multimodal integration and sophisticated signal processing, aiming to approximate a more complete picture of brain activity without compromising patient safety [16] [35].
Quantitative Comparison of Core Performance Metrics
The performance characteristics of invasive and non-invasive neural recording techniques directly dictate their suitability for specific research and clinical applications. The table below summarizes the key quantitative differences across spatial resolution, temporal resolution, and signal-to-noise ratio (SNR).
Table 1: Direct Performance Comparison of Neural Recording Techniques
| Metric | Invasive Techniques (e.g., iEEG, Utah Array) | Non-Invasive Techniques (e.g., EEG, MEG) |
|---|---|---|
| Spatial Resolution | Millimeter to sub-millimeter [1] | Centimetres (EEG); Millimetres to centimetres (MEG) [1] [86] |
| Temporal Resolution | Millisecond-range; records activity up to several kHz [28] [17] | Millisecond-range (EEG & MEG) [86] |
| Signal-to-Noise Ratio (SNR) | High; reduced signal attenuation [1] | Lower; susceptible to noise and artifacts [1] |
| Information Transfer Rate | High; enables complex control (e.g., robotic arms) [77] | Lower; limits speed and complexity of BCI commands [77] |
| Typical BCI Control Accuracy | 85-95% for complex motor tasks [87] | 65-75% for similar tasks [87] |
| Key Signal Types | Action Potentials (APs), Local Field Potentials (LFPs) [28] | Scalp potentials from post-synaptic currents (EEG), magnetic fields (MEG) [28] [86] |
| Cortical Coverage | Limited to implanted areas; cannot cover the whole neocortex [28] [17] | Broad coverage of the entire cortex adjacent to the neurocranium [28] [17] |
The fundamental advantage of invasive methods lies in their proximity to neural signal sources. Action potentials and local field potentials (LFPs) can be recorded directly, providing a rich, high-fidelity signal. The electric fields from neurons decay exponentially with distance, meaning the number of neurons that need to be simultaneously active to produce a detectable signal is magnitudes smaller for invasive Local Field Potentials (LFPs) than for non-invasive EEG. Furthermore, tissue acts as a natural low-pass filter, attenuating high-frequency components before they reach the scalp. This limits non-invasive signals largely to lower-frequency activity (<~90 Hz), whereas invasive methods can capture information up to several kHz [28] [17]. This high SNR and bandwidth underpin the superior performance of invasive Brain-Computer Interfaces (BCIs) in applications requiring precise, high-dimensional control [87].
Non-invasive techniques, while safer and more accessible, face intrinsic physical constraints. The skull and scalp cause significant spatial blurring of electrical signals (EEG), and although MEG is less susceptible to this distortion, its spatial resolution remains coarser than invasive methods [86]. The signals recorded are a weighted average of activity over a large area, dominated by the synchronized post-synaptic currents of pyramidal neurons which are optimally aligned to generate external fields. This makes it difficult to isolate the activity of small, specific neuronal clusters [28] [17].
Experimental Protocols for Direct Comparison
To objectively compare these techniques, researchers employ rigorous experimental paradigms. The following protocols highlight methodologies that directly pit invasive against non-invasive recordings or validate one against the other.
Protocol 1: Simultaneous Intracranial and Scalp EEG Recordings
This powerful paradigm serves as a "Rosetta stone" for deciphering brain activity, allowing for direct, moment-to-moment comparison of signals [16].
- Objective: To establish a ground-truth relationship between invasive intracranial EEG (iEEG) and non-invasive scalp EEG signals, improving source localization and our understanding of how deep brain activity manifests at the scalp.
- Methodology:
- Participant Cohort: The study typically involves patients with drug-resistant epilepsy who are already undergoing invasive monitoring with stereotactic EEG (sEEG) or subdural grids as part of their clinical pre-surgical evaluation [16].
- Equipment: A high-density (hd-EEG) scalp cap with 128-256 electrodes is used simultaneously with the clinically implanted intracranial electrodes [16].
- Recording: Neural activity is recorded concurrently from both the scalp and intracranial electrodes during rest and/or various cognitive tasks.
- Data Analysis: The analysis focuses on correlating specific signal features (e.g., spectral power, event-related potentials, high-frequency oscillations) between the two recording systems. This helps quantify the attenuation and spatial smearing of signals from the depth to the scalp.
- Key Findings: Such studies have been instrumental in advancing the understanding of epilepsy networks and providing a validation benchmark for source localization algorithms, which attempt to infer the location of brain activity from scalp recordings [16].
Protocol 2: Comparative Assessment of Stimulation Techniques
This protocol compares the physiological effects of non-invasive and invasive brain stimulation techniques using a common readout (hd-EEG), rather than comparing recording techniques directly.
- Objective: To compare the electrophysiological effects of single-pulse Transcranial Magnetic Stimulation (TMS) and Intracranial Electrical Stimulation (IES) via their evoked potentials on the scalp [88].
- Methodology:
- Participant Cohorts: TMS-evoked potentials (TEPs) are acquired from healthy subjects, while IES-evoked potentials (IEPs) are recorded from drug-resistant epileptic patients with implanted electrodes during wakefulness and NREM sleep [88].
- Stimulation and Recording: Single-pulse TMS is applied to the cortex of healthy subjects, and IES is applied directly to the cortex of patient subjects. In both cases, the resulting brain responses are measured using high-density scalp EEG.
- Data Analysis: Researchers compare the amplitude, spectral content, and spatiotemporal complexity of the TEPs and IEPs. They also model the estimated electrical fields of the respective stimuli.
- Key Findings: The study found marked differences. During wakefulness, IEPs were larger, slower, and associated with cortical suppression, whereas TEPs exhibited faster, recurrent waves of activation. These differences attenuated during NREM sleep, suggesting brain state-dependent responses to different stimulation modalities [88].
Protocol 3: Validating Non-Invasive Source Estimation with Invasive Recordings
This protocol tests whether advanced computational models combining non-invasive data can accurately estimate signals that resemble those obtained directly from the cortex.
- Objective: To validate a novel model that integrates MEG and fMRI to estimate high-resolution source activity by predicting held-out electrocorticography (ECoG) data [35].
- Methodology:
- Data Collection: MEG data is collected from subjects listening to narrative stories. An open-access fMRI dataset obtained using the same stimuli is used [35].
- Model Training: A transformer-based encoding model is trained to predict both MEG and fMRI signals from the stimulus features. The model contains a latent layer representing the estimated cortical source activity.
- Validation: The model's performance is tested by using its source estimates to predict ECoG data from a completely separate cohort of epileptic patients. The accuracy of these predictions is compared against a baseline model trained directly on the ECoG data.
- Key Findings: The model's source estimates, derived purely from non-invasive data, were able to predict the invasive ECoG signals more accurately than a model trained directly on the ECoG data itself. This demonstrates the potential of integrated models to bridge the resolution gap in non-invasive neuroimaging [35].
Signaling Pathways and Experimental Workflows
The journey of a neural signal from its origin in the brain to its measurement and interpretation involves a complex pathway that is fundamentally different for invasive versus non-invasive techniques. The following diagram illustrates this workflow and the points of divergence between the two approaches.
Diagram 1: Neural Signal Recording and Processing Workflow. This diagram contrasts the pathways for invasive (red) and non-invasive (blue) neural signal acquisition, highlighting key points of signal degradation and preservation.
The workflow begins with the fundamental source of measurable neural activity: post-synaptic currents in populations of neurons, particularly pyramidal cells with parallel dendrites that create summating electric fields [28] [86]. The pathway then diverges dramatically.
In the invasive pathway, the electrode is placed directly on the cortex (ECoG) or within the brain tissue (intracortical arrays), minimizing the distance to the signal source. This allows for the direct recording of high-frequency components like action potentials (APs) and detailed local field potentials (LFPs) with minimal attenuation, resulting in a High SNR Recording [28] [1].
In the non-invasive pathway, the signal must traverse the cerebrospinal fluid, skull, and scalp before reaching the scalp electrodes (EEG). This journey has two major consequences, represented by the Signal Attenuation & Low-Pass Filtering node. First, the tissue layers act as a severe low-pass filter, attenuating high-frequency neural activity beyond ~90 Hz. Second, the signals from multiple distinct neural sources are spatially blurred and distorted. Furthermore, non-invasive recordings are more susceptible to non-neural artifacts (e.g., from muscle movement or eyes) which further reduce the effective SNR [28] [17] [86]. Finally, both pathways converge on the steps of signal processing, feature extraction, and decoding, where advanced algorithms attempt to mitigate the limitations of the non-invasive pathway and leverage the advantages of the invasive one.
The Scientist's Toolkit: Research Reagent Solutions
Selecting the appropriate tools is critical for designing experiments in neural recording. The table below details key hardware, software, and analytical "reagents" essential for work in this field.
Table 2: Essential Research Tools for Neural Recording Experiments
| Tool Name/Type | Function/Purpose | Relevant Technique |
|---|---|---|
| Utah Array | A multi-electrode array implanted into the cortex to record action potentials and LFPs from multiple single neurons. | Invasive (Intracortical) |
| WIMAGINE Implant | A self-contained, implantable ECoG grid that can be wirelessly operated, designed for long-term BCI applications. | Invasive (ECoG) |
| Stentrode | An endovascular electrode array inserted via blood vessels to record from the cortex, minimizing tissue damage. | Minimally Invasive |
| High-Density EEG HydroCel Net | A cap with 128-256 saline-soaked electrodes for comprehensive scalp coverage and improved source localization. | Non-Invasive (EEG) |
| Portable EEG System (e.g., BrainVision LiveAmp) | A mobile system with active electrodes for recording EEG in naturalistic, community settings outside the lab. | Non-Invasive (EEG) |
| MEG System | A whole-head scanner that measures magnetic fields induced by neural currents, offering better spatial resolution than EEG. | Non-Invasive (MEG) |
| MNE-Python | An open-source Python software package for processing and analyzing EEG/MEG data, including source localization. | Data Analysis (Non-Invasive) |
| Transformer-based Encoding Models | A deep learning architecture used to model the relationship between complex stimuli (e.g., narrative stories) and brain responses. | Data Analysis (Multimodal) |
| Intracranial Microstimulation (ICMS) | The delivery of small electrical currents through implanted electrodes to provide direct sensory feedback in a closed-loop BCI. | Invasive (Stimulation) |
This toolkit highlights the diversity of technologies available, from established clinical tools like the Utah Array [28] [77] to emerging platforms like the Stentrode [77] and innovative software like MNE-Python [35] that underpin modern computational analysis. The choice of tool is dictated by the specific research question, balancing the need for signal fidelity against considerations of safety, accessibility, and ecological validity.
Information Transfer Rates as a Key Metric for Brain-Computer Interface Applications
Information Transfer Rate (ITR), often measured in bits per second (bps) or bits per trial, serves as a fundamental metric for quantifying the performance and communication capacity of Brain-Computer Interface (BCI) systems [89]. It provides a standardized measure that combines speed and accuracy, enabling the comparison of different BCI technologies, paradigms, and research studies. For researchers investigating the spatial and temporal resolution of invasive versus non-invasive neural recordings, ITR is a crucial dependent variable that directly reflects the trade-offs between signal quality, invasiveness, and practical utility [11] [28]. This whitepaper examines the role of ITR as a key metric, its relationship with neural signal sources, and the experimental methodologies used for its quantification in contemporary BCI research.
The choice of neural signal acquisition method fundamentally constrains the achievable ITR in a BCI system. This limitation arises from the intrinsic spatial and temporal resolution of each recording technique, which directly influences the signal-to-noise ratio (SNR) and the amount of information that can be decoded from brain activity.
Table 1: Comparison of Neural Signal Acquisition Methods and Their Impact on ITR Potential
| Acquisition Method | Invasiveness | Spatial Resolution | Temporal Resolution | Typical ITR Range (bps) | Key Advantages & Limitations |
|---|---|---|---|---|---|
| Microelectrode Arrays (MEA) | Invasive | Very High (Single Neurons) | Very High (∼1 ms) | Up to 200+ bps [90] | Records action potentials; high SNR but risks from brain surgery [11] |
| Electrocorticography (ECoG) | Semi-Invasive | High (Neural Populations) | High (Milliseconds) | Moderate to High | Good balance of resolution and stability; requires craniotomy [11] [91] |
| Stereo-EEG (sEEG) | Invasive | High (Deep Structures) | High (Milliseconds) | Moderate to High | Accesses deep brain structures; surgical risk [11] |
| Electroencephalography (EEG) | Non-Invasive | Low (∼2-3 cm) | High (Milliseconds) | 0.5 - 3 bps (Practical) [92] | Safe & portable; low spatial resolution and SNR [28] [91] |
| Functional Near-Infrared Spectroscopy (fNIRS) | Non-Invasive | Low | Very Low (Seconds) | Very Low (<1 bps) | Portable & safe; measures slow hemodynamic response [92] [91] |
| Magnetoencephalography (MEG) | Non-Invasive | High | High (Milliseconds) | Low to Moderate | Excellent resolution; expensive and bulky equipment [91] |
Invasive BCIs, such as those using microelectrode arrays, provide direct access to neural signals with high spatial and temporal resolution. This allows them to achieve high ITRs, as evidenced by recent reports of over 200 bps with negligible delay in fully implantable systems [90]. The primary advantage lies in the ability to record action potentials and local field potentials (LFPs) close to their source, minimizing the signal degradation and spatial blurring that occurs with non-invasive methods [11] [28]. Conversely, non-invasive BCIs, primarily using EEG, contend with fundamental physical limitations. The skull and other tissues act as a strong low-pass filter, attenuating high-frequency signals and limiting the detectable information to a fraction of that available with invasive methods [28]. This results in ITRs that are orders of magnitude lower, typically sufficing for basic communication but falling short of enabling naturalistic, fluid interaction [92].
The Encoding-Decoding Framework and ITR Calculation
At its core, a BCI operates on the principles of neural encoding and decoding, forming a closed-loop system. ITR quantifies the efficiency of this loop.
Figure 1: The BCI closed-loop information pathway, where ITR measures the system's efficiency.
Mathematical Foundation of ITR
The standard ITR metric, often called the Bit Rate, is derived from Shannon's information theory and is calculated as follows:
- For a BCI with N equally probable targets:
- Accuracy (P): The probability of a correct selection.
- Bit Rate (B):
B = log2(N) + P * log2(P) + (1-P) * log2((1-P)/(N-1))bits per trial.
- ITR in bits per minute (bpm):
ITR = B * (60 / T), where T is the time per trial in seconds. - ITR in bits per second (bps):
ITR = B / T.
This formula accounts for both the speed (time per trial) and accuracy of selection, providing a commensurate metric that allows for comparison across systems with different numbers of classes and trial durations [89]. Mutual information, a more general form of this metric, measures the dependence between the presented stimulus and the decoded output, representing the true information throughput of the system [90] [89].
Benchmarking and Contemporary ITR Performance
The pursuit of higher ITRs has driven the development of rigorous benchmarking standards. A key recent development is the SONIC framework, which measures the mutual information between presented stimuli and BCI-predicted outputs in a controlled setting [90]. This approach emphasizes reporting performance with associated latency, preventing systems from achieving superficially high ITRs by introducing long, application-prohibitive delays.
Table 2: Reported ITR Performance of Select BCI Systems
| BCI System / Type | Reported ITR | Key Context and Conditions | Citation |
|---|---|---|---|
| Paradromics Connexus (Invasive) | >200 bps (56 ms latency)>100 bps (11 ms latency) | Preclinical benchmark in sheep; fully implantable wireless system. Exceeds transcribed human speech (~40 bps). | [90] |
| Modern Invasive Speech Decoding | N/A (Naturalistic speech synthesis) | Achieved continuous streaming speech synthesis with minimal delay, enabling naturalistic communication. High performance but not reported as a discrete ITR value. | [92] |
| Non-Invasive EEG-based BCIs | 0.5 - 3 bps (Practical scenarios) | Corresponds to slow, discrete selection methods (e.g., P300 speller). Far below natural speech rates of 120-180 words per minute. | [92] |
| Synchron (Endovascular) | ~1-2 bps (Estimated) | Reported performance is orders of magnitude lower than high-end invasive systems like Paradromics. | [90] |
These benchmarks highlight the vast performance gap between invasive and non-invasive technologies. Invasive systems are now demonstrating ITRs sufficient for complex tasks like conversational speech, while non-invasive systems remain limited to basic communication [90] [92]. This disparity is a direct consequence of the underlying spatial and temporal resolution of the recorded neural signals.
Experimental Protocols for Quantifying ITR
The SONIC Benchmarking Protocol
A contemporary protocol for assessing BCI performance involves the SONIC benchmark, designed to provide a rigorous, application-agnostic measure of information transfer [90].
- Objective: To measure the true information transfer rate between a presented stimulus and the BCI's decoded output, accounting for latency.
- Materials:
- Animal Model: Sheep (preclinical).
- BCI System: Fully implanted, wireless Connexus BCI.
- Stimulus Presentation System: Audio equipment for delivering sound sequences.
- Procedure:
- Stimulus Encoding: Controlled sequences of sounds are played to the subject. Each unique sound (or a sequence of sounds) is mapped to a character in a dictionary.
- Neural Recording: The implanted BCI records neural activity from the auditory cortex during stimulus presentation.
- Decoding: The recorded neural signals are processed by a decoding algorithm to predict which sounds were presented.
- Mutual Information Calculation: The mutual information between the sequence of presented sounds and the sequence of predicted sounds is calculated. This value, in bits per second, constitutes the SONIC benchmark score.
- Key Consideration: The benchmark explicitly reports the total system latency, ensuring that high ITR is not achieved at the cost of impractical delay.
Protocol for Invasive Motor Control BCI
Classic experiments demonstrating high-ITR control of external devices often follow this general methodology.
- Objective: To decode movement parameters from motor cortex activity to control an external device (e.g., robotic arm, cursor) in real-time.
- Materials:
- Subject: Non-human primate or human clinical trial participant (e.g., with tetraplegia).
- Implant: Microelectrode array (e.g., Utah Array) implanted in the primary motor cortex (M1).
- Data Acquisition System: High-samplng-rate neural signal processors.
- External Device: Robotic arm or computer cursor.
- Procedure:
- Signal Acquisition: Record action potentials and local field potentials from multiple neurons in M1.
- Feature Extraction: In real-time, extract features such as firing rates of individual neurons or spectral power in specific LFP frequency bands.
- Decoding: Use a decoding algorithm (e.g., Kalman filter, population vector algorithm, optimal linear estimator) to map the neural features to movement parameters like direction, velocity, and grip force [11].
- Closed-Loop Control: The decoded movement commands are sent to the external device. The subject receives visual (and sometimes tactile) feedback, creating a closed-loop system that enables neuroplasticity and learning [11] [28].
- Performance Quantification: ITR is calculated based on the speed and accuracy of task completion (e.g., target acquisition). The high spatial and temporal resolution of the invasive signals enables the high-dimensional control necessary for high ITRs.
Figure 2: A standard experimental workflow for an invasive motor BCI.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Solutions for BCI Development and Testing
| Reagent / Material | Function / Application | Specific Examples & Notes |
|---|---|---|
| Microelectrode Arrays (MEA) | Invasive recording of neural action potentials and LFPs. | Utah Array (Blackrock), Neuropixels probes. Biocompatibility and long-term stability are key challenges [11]. |
| Electrocorticography (ECoG) Grids | Subdural recording of cortical surface potentials. | High-density ECoG grids for improved spatial resolution in speech decoding studies [92]. |
| EEG Systems | Non-invasive recording of scalp potentials. | High-density wet or dry electrode systems. Critical for artifact removal algorithms due to low SNR [91]. |
| Deep Brain Stimulation (DBS) Electrodes | For bidirectional BCIs; both record and modulate deep brain activity. | Used in treating disorders like Parkinson's disease and explored for closed-loop therapies [11]. |
| Neural Signal Processors | Real-time amplification, filtering, and digitization of neural signals. | Systems from Blackrock Microsystems, Intan Technologies. Require high sampling rates (>30 kSps) for spikes. |
| Decoding Algorithms | Translate neural signals into control commands. | Traditional: Kalman filter, linear discriminant analysis. Modern: Convolutional Neural Networks (CNNs), Recurrent Neural Networks (RNNs) [92] [93]. |
| Computational Models | Simulate neural encoding/decoding and test algorithms. | Bayesian models of neural activity; head models for EEG source localization [11] [28]. |
| Calibration Paradigms | Train and calibrate the BCI decoder for a specific user. | Motor imagery tasks, visual evoked potentials (P300), auditory stimuli. |
Information Transfer Rate stands as a critical, standardized metric for evaluating and advancing BCI technologies. It objectively captures the trade-offs between the high spatial-temporal resolution of invasive recordings, which enable ITRs over 200 bps, and the safety and accessibility of non-invasive methods, which are currently limited to low ITRs. As the field progresses with new materials, algorithms, and benchmarking standards like SONIC, the pursuit of higher ITRs will continue to drive innovation. This will ultimately expand the capabilities of BCIs for both restorative neurotechnology and fundamental research into brain function, firmly grounded on the principles of information theory.
The field of neural interfaces is defined by a fundamental choice between invasive and non-invasive recording methodologies, each presenting a distinct profile of advantages and limitations. This choice hinges on a critical trade-off between signal fidelity and practical deployment, factors that directly influence the safety, accessibility, and ultimate application of the technology. Invasive Brain-Computer Interfaces (BCIs), which require surgical implantation, provide high-resolution signals from specific neural populations, enabling complex decoding tasks such as speech restoration and robotic arm control [77] [94]. Conversely, non-invasive systems, which measure neural signals from outside the skull, offer greater safety and accessibility but contend with signals that are attenuated by the skull and other tissues, resulting in lower spatial resolution and bandwidth [14] [77]. This whitepaper provides a structured analysis of these trade-offs, synthesizing recent research advances to guide researchers and drug development professionals in selecting appropriate technologies for specific applications. We summarize quantitative performance data, detail experimental protocols from key studies, and outline the essential toolkit for research in this rapidly evolving field, with a particular focus on the implications of spatial and temporal resolution for research and therapeutic development.
Quantitative Comparison of Invasive and Non-Invasive Interfaces
The performance gap between invasive and non-invasive neural interfaces can be quantified across several key metrics, including information transfer rate, decoding accuracy, and long-term stability. The tables below consolidate recent data from the literature to illustrate these differences clearly.
Table 1: Performance Comparison of Select Neural Interfaces
| Interface Type | Application | Reported Performance | Key Study / Technology |
|---|---|---|---|
| Invasive (Intracortical) | Speech Decoding | Up to 97.5% accuracy for attempted speech in ALS patients [94] | Patentsight Analysis [94] |
| Invasive (Intracortical) | Motor Control (e.g., robotic arm) | >85% accuracy with sufficient training [94] | Patentsight Analysis [94] |
| Minimally Invasive (Stentrode) | Computer Cursor Control | Successful control in patients; endovascular placement [77] | Stentrode [77] |
| Non-Invasive (sEMG Wristband) | Handwriting Decoding | 20.9 words per minute (WPM); 16% improvement with personalization [95] | Surface EMG Research Device [95] |
| Non-Invasive (sEMG Wristband) | Discrete Gesture Detection | 0.88 detections per second [95] | Surface EMG Research Device [95] |
| Non-Invasive (EEG/MEG) | Word Decoding (from speech/listening) | Up to 37% top-10 accuracy with a 250-word retrieval set [5] | Transformer-based Deep Learning Pipeline [5] |
Table 2: Safety and Deployment Considerations
| Consideration | Invasive BCI | Non-Invasive BCI |
|---|---|---|
| Primary Safety Risks | Surgical risks, tissue damage, infection (4-12% rate), inflammatory foreign body response, glial scarring [96] | Minimal physical risk; primary concerns are mental privacy and data security [94] |
| Signal Longevity | Degradation over months to years (typically 1-5 years) due to biofouling and tissue response [96] | Stable long-term signal quality; performance depends on hardware donning and external noise [95] [97] |
| Regulatory Status | Stringent medical device regulation (FDA) [96] | "Regulatory vacuum" for consumer devices; considered medical devices in clinical applications [94] |
| Key Deployment Challenge | High cost ($35,000 - $150,000); requires neurosurgery; limited to specific patient populations [96] | Consumer privacy practices often fail to meet global norms; data can be accessed and shared by companies [94] |
| Target Population | Patients with severe disabilities (e.g., ALS, spinal cord injury) in clinical trials [77] [96] | Broad, including consumer markets, rehabilitation, and research [14] [77] |
Experimental Protocols for Neural Decoding
To achieve the performance metrics outlined above, researchers employ sophisticated experimental protocols and decoding algorithms. The following section details the methodologies from several landmark studies in non-invasive decoding.
Protocol: Non-Invasive Handwriting Decoding via Surface EMG
This protocol [95] enables the decoding of handwriting and gestures from neuromuscular signals at the wrist, offering a high-bandwidth communication channel without brain implantation.
- Objective: To develop a generic non-invasive neuromotor interface for computer input (e.g., continuous navigation, gesture detection, handwriting) using surface electromyography (sEMG) that generalizes across users.
- Materials and Setup:
- sEMG Research Device (sEMG-RD): A dry-electrode, multichannel wristband with high sample rate (2 kHz) and low noise (2.46 μVrms), fabricated in four sizes to fit a range of wrist circumferences [95].
- Data Collection Infrastructure: Custom software for prompting participants and recording synchronized sEMG and label timestamps [95].
- Participants: A large and anthropometrically diverse cohort (e.g., 162 to 6,627 participants, depending on the task) [95].
- Procedure:
- Participants don the sEMG-RD on their dominant wrist and are seated in front of a computer running the prompting software [95].
- For the handwriting task, participants are prompted to hold their fingers together as if holding a pen and to "write" the prompted text in the air [95].
- For the discrete-gesture task, a prompter instructs participants to perform one of nine distinct gestures (e.g., finger pinches, thumb swipes) in a randomized order [95].
- A real-time processing engine records raw sEMG data and the timestamps of the prompted labels. A time-alignment algorithm is applied post-hoc to infer the actual onset of the gesture events, accounting for participant reaction time [95].
- Data Processing and Decoding:
- Neural networks are trained on the collected dataset to map sEMG patterns to intended actions.
- Model performance is evaluated in closed-loop (online) tasks, reporting metrics such as words per minute (WPM) for handwriting and gestures per second for discrete tasks [95].
- The generic model can be personalized with user-specific data to improve performance, as demonstrated by a 16% improvement in handwriting decoding [95].
Protocol: Decoding Words from Non-Invasive MEG and EEG
This protocol [5] leverages deep learning to decode individual words from non-invasive brain recordings across a large number of participants and languages.
- Objective: To decode individual words from magnetoencephalography (MEG) and electroencephalography (EEG) signals during reading or listening tasks.
- Materials and Setup:
- Procedure:
- Data Processing and Decoding:
- Model Architecture: A deep learning pipeline featuring a transformer encoder is trained with a contrastive objective. The model includes a "subject layer" to account for inter-participant variability [5].
- Input Features: The model uses a concatenation of three stimulus feature streams: contextual word embeddings from GPT-2, phoneme features, and mel-spectrograms of the audio [5].
- Training and Evaluation: The model is trained to decode brain activity into a representation that can be matched to the correct word from a set of candidates. Performance is reported as top-10 accuracy (the percentage of trials where the correct word is among the model's top 10 guesses) [5].
- Scaling Analysis: The study systematically analyzes how decoding performance scales with the amount of training data and with the number of trial averages used during testing [5].
Signaling Pathways and Experimental Workflows
The following diagrams, generated using Graphviz DOT language, illustrate the core signaling pathways targeted by neuromodulation and the experimental workflows for non-invasive neural decoding.
Diagram 1: Neural circuits and neuromodulation targets for substance use disorders. This pathway illustrates the brain regions and connections implicated in addiction, based on reviews of neuromodulation therapies [60]. Invasive (DBS) and non-invasive (rTMS, tDCS) techniques target specific nodes within this network, such as the Nucleus Accumbens (NAc) and Prefrontal Cortex (PFC), to modulate circuit activity and reduce symptoms like craving.
Diagram 2: sEMG-based neuromotor interface workflow. This workflow outlines the experimental procedure for developing and using a non-invasive interface based on surface electromyography (sEMG), as described in [95]. The process involves collecting data from a diverse population to train a generic decoder that can be personalized for individual users, enabling high-performance gesture and handwriting decoding.
The Scientist's Toolkit: Research Reagent Solutions
This section details key hardware, software, and analytical tools essential for contemporary research into invasive and non-invasive neural interfaces.
Table 3: Essential Research Tools for Neural Interface Development
| Tool Name / Category | Type | Primary Function | Key Features / Examples |
|---|---|---|---|
| sEMG Research Device (sEMG-RD) | Hardware | Records neuromuscular signals from the wrist for gesture and intent decoding. | Dry electrodes, wireless, multi-size bands, high sample rate (2 kHz) [95]. |
| High-Density EEG Net | Hardware | Traditional lab-based EEG recording with high spatial sampling. | 129-channel HydroCel Geodesic Sensor Net (used in lab vs. community comparisons) [97]. |
| Portable EEG System | Hardware | Community-based neural recording outside the laboratory. | 32-active electrode system (e.g., BrainVision LiveAmp); enables home-based studies [97]. |
| MEG Scanner | Hardware | Records magnetic fields induced by neural currents. | Provides higher signal-to-noise ratio than EEG for non-invasive decoding tasks [5]. |
| Deep Learning Pipelines | Software/Algorithm | Decodes complex intentions from neural data. | Transformer-based models for word decoding [5]; neural networks for cross-user sEMG decoding [95]. |
| Source Localization Software | Software/Algorithm | Solves the "inverse problem" to estimate brain source activity from M/EEG. | MNE-Python, used to construct source spaces and lead-field matrices [35]. |
| fUS Imaging | Hardware | Functional ultrasound imaging of brain activity. | Measures hemodynamic responses; requires cranial window but offers better resolution than fMRI [77]. |
| Stentrode | Hardware | Minimally invasive endovascular recording electrode. | Placed in blood vessels; targets motor cortex for BCI control [77]. |
| WIMAGINE System | Hardware | Implantable ECoG grid for chronic brain recording. | Wireless, designed for long-term BCI applications like exoskeleton control [77]. |
The trade-off between safety/accessibility and signal fidelity remains the central paradigm in neural interface research. Invasive BCIs provide unparalleled resolution for restoring communication and motor function in severe paralysis but are constrained by surgical risks, finite longevity, and high costs, limiting them to narrow clinical populations [96]. Non-invasive technologies, while safer and more scalable, have historically been bottlenecked by lower bandwidth. However, recent breakthroughs demonstrate a dramatic narrowing of this performance gap. The combination of large datasets, advanced deep learning models, and novel hardware—such as the sEMG wristband achieving 20.9 WPM for handwriting [95] and MEG decoders approaching word-level language reconstruction [5]—signals a pivotal shift. For researchers and drug development professionals, this evolution expands the toolkit for probing brain function and developing therapeutics. The future lies not in a single superior technology, but in the context-dependent selection of the appropriate interface, and increasingly, in the integration of multiple modalities to harness the complementary strengths of each approach.
The field of neuroprosthetics is dedicated to developing technologies that can restore lost neurological function, with a predominant focus on motor control and communication. A central challenge in this endeavor lies in the method of recording neural activity, which fundamentally splits into two paradigms: invasive techniques, which require surgical implantation of electrodes on or in the brain tissue, and non-invasive techniques, which record signals from outside the skull. The choice between these approaches involves a critical trade-off between the superior signal quality of invasive methods and the superior safety and accessibility of non-invasive ones. This trade-off is quantified through key performance metrics such as spatial resolution (the ability to distinguish signals from adjacent neural populations), temporal resolution (the speed at which neural dynamics can be tracked), and the resulting decoding performance (the accuracy and speed of interpreting neural intent). This review delves into recent case studies to provide a technical comparison of these approaches, framing the analysis within the broader thesis of spatial and temporal resolution in neural recordings for motor control and communication.
The following tables consolidate key performance metrics from recent seminal studies, providing a direct comparison between invasive and non-invasive neuroprosthetic systems for motor and communication tasks.
Table 1: Performance Metrics for Communication and Motor Decoding
| Study & Modality | Task | Key Performance Metric | Information Transfer Rate (ITR) | Subjects (n) |
|---|---|---|---|---|
| Invasive (Intracortical) [98] | Imagined Handwriting | 94.1% accuracy, 90 chars/min | ~180 bits/min (est. from text) | N/S |
| Invasive (ECoG) [26] | Movement Decoding (Rest vs. Move) | 0.98 movement detection rate | N/S | 56 |
| Non-Invasive (EEG) [98] | Imagined Handwriting | 89.83% character accuracy | ~60 bits/min (est. from text) | 15 |
| Non-Invasive (EEG/MEG) [5] | Individual Word Decoding | 37% top-10 accuracy (250-word set) | 0.5-3 words/min | 723 |
Table 2: Spatial and Temporal Resolution by Recording Modality
| Modality | Spatial Resolution | Temporal Resolution | Signal-to-Noise Ratio (SNR) | Clinical Risk |
|---|---|---|---|---|
| ECoG / Intracortical [26] [98] | Millimeter (ECoG) to single neuron (Intracortical) | Millisecond (≤100 ms) | High | High (surgical implantation) |
| EEG [98] [92] [5] | ~2-3 cm | Millisecond | Low (typically < 1:10) | Low |
| MEG [5] [35] | Higher than EEG | Millisecond | Higher than EEG | Low |
| fMRI [35] | Millimeter | Seconds (sluggish hemodynamic response) | High for slow signals | Low |
Case Study 1: Invasive Approaches for Motor and Speech Decoding
Experimental Protocols and Workflows
A landmark study in invasive neural decoding developed an open-source platform, py_neuromodulation, to standardize brain signal decoding from patients with implanted electrodes for movement disorders, depression, and epilepsy [26]. The methodology involved:
- Data Acquisition: Recording intracranial electroencephalography (iEEG) and electrocorticography (ECoG) from 73 neurosurgical patients, amounting to 123 hours of brain data. For motor decoding, ECoG strips were implanted through deep brain stimulation (DBS) burr holes in patients with Parkinson's disease [26].
- Signal Processing: A modularized feature estimation chain was implemented. This included extracting oscillatory dynamics in frequency bands from 4-400 Hz, waveform shape, and interregional coherence. Raw data was processed in 1,000 ms segments, continuously updated at 10 Hz, yielding a temporal resolution of 100 ms [26].
- Machine Learning: A ridge-regularized logistic regression classifier was trained for a two-class problem (rest vs. movement) and evaluated using balanced accuracy with 3-fold cross-validation. To overcome the limitation of patient-specific training, the researchers pioneered a connectomic approach. This method uses functional or structural connectivity fingerprints from normative brain maps to select the optimal recording channel for movement decoding across patients without individual training [26].
For speech decoding, a state-of-the-art invasive system utilized high-density surface recordings from the speech sensorimotor cortex of a participant with anarthria (severe paralysis). The protocol employed deep learning recurrent neural network transducer models to achieve online, large-vocabulary speech synthesis personalized to the participant's pre-injury voice, performing neural decoding in 80 ms increments [92].
Key Findings and Limitations
The invasive approach demonstrated exceptional performance. Movement decoding achieved a 0.98 movement detection rate with high spatiotemporal precision [26]. The speech neuroprosthesis enabled continuously streaming speech synthesis with naturalistic quality, a feat that directly leverages the high spatial and temporal resolution of intracranial recordings [92]. However, these systems face significant hurdles: they require neurosurgical implantation with associated risks, their long-term stability and longevity can be limited, and the presence of therapeutic stimulation (like DBS) can complicate signal decoding and necessitate specialized model adaptation [26] [92].
Case Study 2: Non-Invasive Approaches for Handwriting and Word Decoding
Experimental Protocols and Workflows
Non-invasive research has made significant strides by leveraging advanced machine learning to overcome inherent signal quality challenges.
EEG-based Handwriting Decoding [98]:
- Data Acquisition: EEG data was collected from 15 participants using a 32-channel headcap.
- Signal Preprocessing: Data was preprocessed with bandpass filtering and Artifact Subspace Reconstruction (ASR) to mitigate noise.
- Feature Engineering: A set of 85 time-domain, frequency-domain, and graphical features was extracted. Pearson correlation coefficient-based feature selection was used to identify 10 key features, reducing computational latency with minimal accuracy loss.
- Model & Deployment: A hybrid Temporal Convolutional Network and multilayer perceptron (TCN-MLP) architecture,
EEdGeNet, was developed. The entire pipeline was deployed for real-time inference on an NVIDIA Jetson TX2 edge device, emphasizing portability.
Large-Scale Word Decoding from M/EEG [5]:
- Data Acquisition: This study created an unprecedented dataset of 723 participants recorded with EEG or MEG while reading or listening to sentences, amounting to 5 million words across three languages.
- Model Architecture: A deep learning pipeline with a contrastive objective was used. It incorporated a subject-specific layer and a transformer to operate at the sentence level, trained to decode individual words.
- Validation: The model was validated for single-trial decoding performance to simulate real-time conditions and its generalizability was tested across devices, languages, and tasks.
Key Findings and Limitations
Non-invasive systems have achieved remarkable feats, such as ~90% accuracy for decoding imagined handwriting from EEG in real-time on a portable device [98]. The large-scale word decoding study revealed important scaling laws: performance increases log-linearly with the amount of training data and with the number of trials averaged during testing [5]. It also found that MEG and reading tasks are easier to decode than EEG and listening tasks [5].
The primary limitation remains the low signal-to-noise ratio (SNR) and limited spatial resolution of non-invasive signals [92] [5]. This fundamentally constrains the amount of information that can be decoded, resulting in lower information transfer rates compared to invasive methods. Furthermore, there is substantial inter-individual variability, which complicates the creation of universal models and often necessitates user-specific calibration [92].
The Resolution Trade-off and Emerging Hybrid Solutions
The core trade-off is clear: invasive techniques provide high spatial and temporal resolution at the cost of clinical risk, while non-invasive techniques offer safety and accessibility but with fundamentally lower resolution.
Emerging research is focused on bridging this gap. One promising approach is the multi-modal integration of non-invasive techniques. For instance, a novel framework combines MEG and fMRI within a transformer-based encoding model. This model uses anatomical information and biophysical forward models to estimate latent cortical source activity from MEG and fMRI data simultaneously, aiming to achieve a unified view with both high spatial and temporal resolution [35]. Initial validation shows that these estimated sources can predict invasive electrocorticography (ECoG) data more effectively than models trained directly on the ECoG, suggesting a path toward millisecond-and-millimeter brain mapping non-invasively [35].
Another integrative strategy involves combining non-invasive brain stimulation (NIBS) with AI. A proposed bidirectional framework would use techniques like transcranial direct current stimulation (tDCS) to enhance neural signals related to speech production or auditory processing, which are then decoded by AI algorithms, creating a more effective and natural communication loop [92].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Technologies for Neural Decoding Research
| Item Name | Function/Application | Example Use Case |
|---|---|---|
py_neuromodulation [26] |
An open-source, modularized Python platform for standardized invasive brain signal decoding. | Processing iEEG/ECoG data; extracting oscillatory features, waveform shape, and coherence for machine learning. |
| ECoG Strips / Intracortical Arrays [26] [98] | Implantable electrodes for recording neural signals with high spatial and temporal resolution. | Surgical implantation for decoding movement in Parkinson's disease or speech in anarthric patients. |
| High-Density EEG/MEG Systems [98] [5] [35] | Non-invasive neuroimaging devices for recording electrical (EEG) or magnetic (MEG) brain activity. | Acquiring neural data for imagined handwriting or word decoding paradigms from a large cohort of participants. |
| Artifact Subspace Reconstruction (ASR) [98] | A statistical method for real-time removal of large-amplitude artifacts from EEG signals. | Preprocessing pipeline for cleaning noisy EEG data before feature extraction and model training. |
| Edge Computing Device (NVIDIA Jetson) [98] | A portable, low-power hardware platform for on-device machine learning inference. | Deploying trained decoding models (e.g., EEdGeNet) for real-time, low-latency BCI operation outside the lab. |
| Transformers & Deep Learning Models [5] [35] | Advanced neural network architectures for sequence modeling and cross-modal integration. | Decoding individual words from M/EEG by capturing context; integrating MEG and fMRI data in a unified model. |
| Normative Connectomic Templates [26] | Standardized maps of brain connectivity derived from population-level data (e.g., fMRI). | Enabling across-patient decoding by selecting optimal recording channels based on network overlap, without individual training. |
The development of neural interfaces and brain-computer interfaces (BCIs) represents a frontier in neuroscience and neurotechnology. A critical challenge in this domain is establishing robust validation frameworks that ensure these technologies provide reliable and meaningful data for both basic research and clinical applications. The validation process is particularly complex due to the fundamental trade-offs between spatial and temporal resolution in invasive versus non-invasive neural recording modalities. Non-invasive techniques like electroencephalography (EEG) and magnetoencephalography (MEG) offer broad accessibility and safety but capture neural signals with limited spatial resolution and through the distorting filters of the skull and scalp [5] [99]. In contrast, invasive methods such as electrocorticography (ECoG) and stereotactic EEG (sEEG) provide signals with high spatial and temporal fidelity by recording directly from the cortical surface or within brain structures, but they require neurosurgical implantation and carry associated risks [26] [7].
The core objective of validation frameworks in this context is to bridge the gap between the neural signals acquired through these modalities and their real-world interpretation, whether for decoding mental states, controlling external devices, or informing therapeutic interventions. This requires a structured approach to demonstrate that the measures derived from neural recordings accurately reflect underlying neurophysiological processes and can reliably predict behavioral or clinical outcomes. The V3 Framework (Verification, Analytical Validation, and Clinical Validation), adapted from the Digital Medicine Society's approach for clinical digital measures, provides a comprehensive structure for this evidence-building process [100]. This framework distinguishes between verifying that technologies accurately capture raw data, validating that algorithms correctly transform this data into biological metrics, and confirming that these metrics meaningfully reflect relevant biological or behavioral states within a specific context of use.
Theoretical Foundations of the V3 Validation Framework
The V3 Framework offers a systematic approach for establishing the reliability and relevance of neural measures across the development pipeline. Each component addresses a distinct aspect of the validation process, creating a comprehensive evidence base for interpreting neural recording data.
Verification: Ensuring Data Fidelity
Verification constitutes the foundational layer, focusing on the technical performance of the data acquisition system itself. This process ensures that digital technologies accurately capture and store raw neural signals without introducing artifacts or distortions that could compromise downstream analyses. For neural recording systems, verification involves characterizing key performance parameters such as signal-to-noise ratio, sampling fidelity, channel consistency, and temporal stability. In practice, this includes testing the amplifier systems for appropriate gain and bandwidth, validating electrode impedance across channels, verifying sampling rate stability, and confirming the absence of systematic electrical artifacts or cross-talk between channels. For invasive systems, additional verification steps may include assessing the biocompatibility of implant materials and the stability of long-term recordings [95] [26]. The verification process is largely modality-agnostic but must account for the specific technical constraints of each recording approach, whether it's dealing with the microvolt-scale signals of scalp EEG or the rich spectral content of intracranial recordings.
Analytical Validation: From Signal to Feature
Analytical validation assesses the performance of algorithms that transform raw neural signals into meaningful features or biomarkers. This stage establishes that computational pipelines reliably extract relevant information from neural data with appropriate precision and accuracy. Key aspects include evaluating feature extraction methods for time-frequency analyses, spatial filtering techniques for source localization, decoding algorithms for predicting stimuli or behavior, and classification approaches for identifying neural states. For example, in speech decoding research, analytical validation would assess how well algorithms can reconstruct linguistic units (phonemes, words) from neural activity patterns, using metrics like accuracy, precision, and recall [5] [7]. This process must account for the substantial differences in signal properties between invasive and non-invasive recordings; invasive signals typically contain broader frequency content (including high-gamma activity) and higher spatial specificity, requiring different analytical approaches than the spatially-blurred signals obtained non-invasively.
Clinical Validation: Establishing Biological Relevance
Clinical validation (or, in preclinical contexts, "biological validation") constitutes the highest level of the framework, confirming that neural measures accurately reflect meaningful behavioral, cognitive, or pathological states relevant to their context of use. This stage moves beyond technical signal quality to establish functional correspondence between neural features and real-world phenomena. For neural interfaces, this might involve demonstrating that motor intention decoders actually facilitate device control, that speech decoding approaches enable communication, or that pathological neural biomarkers reliably track disease severity or treatment response [26]. The validation criteria are necessarily specific to the application domain but should establish clear linkage between neural measures and clinically or behaviorally relevant outcomes. For invasive recordings, clinical validation often leverages the superior signal quality to establish more direct relationships between neural activity and specific behaviors or symptoms, while non-invasive approaches may focus on more aggregate neural patterns and their correlation with behavioral states.
Comparative Analysis of Neural Recording Modalities
The validation approach must be tailored to the specific capabilities and limitations of different neural recording modalities. The table below summarizes key characteristics of major invasive and non-invasive techniques that influence validation strategies.
Table 1: Characteristics of Neural Recording Modalities Influencing Validation Approaches
| Modality | Spatial Resolution | Temporal Resolution | Key Advantages | Primary Validation Challenges |
|---|---|---|---|---|
| sEMG | Moderate (muscle groups) | High (ms) | High signal-to-noise ratio for motor commands | Cross-user generalization, sensor placement variability [95] |
| EEG | Low (cm) | High (ms) | Non-invasive, portable, low cost | Low spatial resolution, skull distortion, artifacts [5] [101] |
| MEG | Moderate (mm-cm) | High (ms) | Excellent temporal resolution, no reference needed | Expensive, limited availability, motion sensitivity [5] |
| fMRI | High (mm) | Low (s) | High spatial resolution, whole-brain coverage | Indirect neural measure (hemodynamic response), poor temporal resolution [35] |
| ECoG/sEEG | High (mm) | High (ms) | High signal quality, broad frequency range | Invasive implantation, limited spatial coverage [26] [7] |
| Intracortical | Very High (μm) | Very High (ms) | Single-neuron resolution, precise timing | Most invasive, tissue response, long-term stability [26] |
Performance Benchmarks Across Modalities
Different neural recording approaches demonstrate varying capabilities for decoding behavioral and cognitive states, which directly influences validation strategies and performance expectations.
Table 2: Performance Benchmarks for Neural Decoding Across Modalities
| Modality | Decoding Task | Reported Performance | Validation Approach |
|---|---|---|---|
| sEMG [95] | Handwriting decoding | 20.9 words per minute | Cross-user generalization without calibration |
| sEMG [95] | Discrete gesture detection | 0.88 detections per second | Closed-loop evaluation with diverse participants |
| EEG/MEG [5] | Word decoding | 37% top-10 accuracy (250-word set) | Cross-dataset validation with 723 participants |
| ECoG [26] | Movement detection (Parkinson's disease) | 0.8 balanced accuracy, 0.98 movement detection rate | Patient-specific training with cross-validation |
| MEG-fMRI Fusion [35] | Speech comprehension mapping | Higher spatial and temporal fidelity than single modalities | Comparison with ground-truth ECoG data |
| EEG [101] | Listened speech decoding | 2x improvement over previous methods | Parallel phoneme sequence prediction accuracy |
Figure 1: V3 Validation Framework for Neural Recordings. This diagram illustrates the sequential relationship between verification, analytical validation, and clinical validation, and how both invasive and non-invasive recording approaches influence each stage.
Methodological Approaches for Benchmarking Neural Recordings
Establishing Ground Truth for Validation
A fundamental challenge in validating neural decoding approaches is establishing appropriate ground truth for comparison. Different strategies have emerged depending on the recording modality and research question:
Behavioral Correspondence: For motor decoding, actual limb movements or muscle activity provide ground truth. In ECoG studies of movement in Parkinson's disease patients, movement timings were precisely tracked using motion capture systems to align with neural signals [26].
Stimulus Reconstruction: In perceptual experiments, the presented stimuli (images, sounds, text) serve as ground truth. For speech decoding, the exact timing and content of listened or produced speech provides reference points for evaluating neural decoding algorithms [5] [7].
Cross-Modal Validation: Using superior recording modalities to validate inferior ones. For example, ECoG recordings have been used to validate source-localized MEG signals, and fMRI has been used to constrain MEG source modeling [35].
Clinical Outcomes: For therapeutic applications, clinical assessment scores provide validation targets. In deep brain stimulation for Parkinson's disease, Unified Parkinson's Disease Rating Scale (UPDRS) scores have been correlated with neural decoding performance [26].
Experimental Protocols for Neural Decoding Validation
Robust validation requires carefully designed experimental protocols that generate the necessary data for evaluating neural decoding approaches:
Speech Decoding Protocol (as implemented in [5] and [101]):
- Stimulus Presentation: Participants listen to or read words/sentences while neural activity is recorded. The LittlePrince dataset used identical stimuli across listening and reading conditions.
- Signal Acquisition: Simultaneous recording using EEG, MEG, or ECoG with precise timestamping of stimulus onsets.
- Preprocessing: Standardized filtering, artifact removal, and epoch extraction time-locked to word onsets (-0.5 to 2.5 seconds).
- Feature Extraction: For EEG/MEG, transformation to word embeddings using neural networks; for ECoG, calculation of mutual information between neural signals and speech features [7].
- Decoding Model Training: Using contrastive learning objectives with transformer architectures to predict words from neural features.
- Cross-Validation: Leave-one-subject-out or leave-one-cohort-out validation to assess generalizability.
Motor Decoding Protocol (as implemented in [26]):
- Movement Task: Patients perform structured upper limb movements (rest vs. movement blocks) with motion capture validation.
- Neural Recording: ECoG strips placed over sensorimotor cortex during deep brain stimulation surgery.
- Feature Extraction: Fast Fourier Transform features computed in 1-second windows updated at 10Hz resolution across eight frequency bands.
- Classification: Ridge-regularized logistic regression trained to distinguish movement vs. rest states.
- Performance Metrics: Balanced accuracy for sample-wise classification and movement detection rate for entity-level recognition.
Figure 2: Comparative Validation Workflows for Invasive vs. Non-Invasive Neural Recordings. This diagram contrasts the distinct validation approaches required for different recording modalities, highlighting how data quality influences methodological choices.
Table 3: Essential Research Tools for Neural Recording Validation
| Tool Category | Specific Examples | Function in Validation | Considerations |
|---|---|---|---|
| Recording Hardware | sEMG wristband [95], High-density EEG systems, MEG systems [5], ECoG strips [26] | Signal acquisition with specific spatial/temporal resolution | Signal-to-noise ratio, channel count, portability |
| Signal Processing | Py_neuromodulation [26], MNE-Python [35], BENDR [99] | Feature extraction, artifact removal, normalization | Computational efficiency, reproducibility |
| Decoding Algorithms | Transformer architectures [5] [102], Ridge regression [26], Mutual information analysis [7] | Mapping neural signals to behavior or stimuli | Generalizability, computational demands, interpretability |
| Validation Metrics | Balanced accuracy, Top-k accuracy, Movement detection rate, F1-score | Quantifying decoding performance | Appropriateness for imbalanced data, clinical relevance |
| Data Augmentation | Sequential sampling, Random contiguous sampling [102] | Addressing class imbalance, improving generalizability | Preservation of physiological plausibility |
Advanced Validation Strategies for Cross-Modal Generalization
Addressing the Generalization Challenge
A persistent challenge in neural interface validation is achieving robust performance across participants, sessions, and recording conditions. Multiple approaches have emerged to address this fundamental limitation:
Subject-Independent Models: The development of generic decoders that work across individuals without recalibration. The sEMG research demonstrated the feasibility of this approach, with a wristband interface achieving 0.66 target acquisitions per second in continuous navigation and handwriting at 20.9 words per minute across users without person-specific training [95].
Connectomic Decoding: Using functional or structural connectivity fingerprints to improve cross-participant generalization. This approach maps recording locations to normative brain space, identifying optimal connectomic templates for decoding that can be applied across individuals with different electrode placements [26].
Multi-Modal Fusion: Combining complementary recording modalities to overcome individual limitations. The MEG-fMRI fusion approach demonstrated how integrating temporal precision from MEG with spatial specificity from fMRI produces source estimates with higher spatiotemporal fidelity than either modality alone [35].
Data Augmentation: Using computational methods to increase effective dataset size and diversity. Transformer-based frameworks have employed sequential sampling, random contiguous sampling, and random non-contiguous sampling to improve model robustness and address class imbalance in seizure detection [102].
Scaling Laws and Data Requirements
Recent large-scale studies have revealed important relationships between data quantity and decoding performance:
Training Data Scaling: Word decoding performance from M/EEG signals follows a roughly log-linear improvement trend with increasing training data volume, showing no clear signs of diminishing returns even with hundreds of participants [5].
Test-Time Averaging: Decoding performance steadily increases with the number of predictions averaged during testing, following a clear log-linear trend. Some datasets show two-fold improvement with averaging just 8 predictions of the same word, suggesting that noise rather than fundamental algorithmic limitations constrains single-trial performance [5].
Participant-Specific Optimization: While generic models show promise, personalization continues to offer benefits. sEMG decoding models for handwriting showed 16% improvement when personalized to individual users [95].
Validation frameworks for neural recordings must continue evolving to address emerging challenges and opportunities in the field. The fundamental trade-offs between spatial and temporal resolution in invasive versus non-invasive approaches necessitate tailored validation strategies that acknowledge the distinct strengths and limitations of each modality. Future validation frameworks should prioritize several key areas:
First, standardized benchmarking datasets and metrics across the field would enable more direct comparison between different neural decoding approaches and more systematic assessment of progress. Second, explicit cross-modal validation protocols would help establish correspondences between different recording modalities, potentially enabling non-invasive technologies to leverage insights from invasive approaches. Third, real-world validation environments that move beyond controlled laboratory settings will be essential for translating neural interfaces to practical applications.
The rapid advancement of large-scale AI models and foundation models specifically designed for neural signal processing represents a particularly promising direction [99]. These approaches demonstrate unprecedented capabilities in capturing spatiotemporal dependencies in neural data and generalizing across tasks and individuals. As these methods mature, validation frameworks must adapt to address new questions about model interpretability, biological plausibility, and clinical reliability.
Ultimately, robust validation frameworks that rigorously benchmark neural recording technologies against clinical outcomes and behavioral metrics will be essential for translating promising research findings into practical applications that improve human health and capability. By systematically addressing verification, analytical validation, and clinical validation across the spectrum of neural recording modalities, the field can build the evidentiary foundation needed for meaningful advancement.
Conclusion
The choice between invasive and non-invasive neural recording techniques presents a fundamental trade-off between signal fidelity and practical accessibility. Invasive methods provide unparalleled spatial and temporal resolution for decoding detailed neural processes but face challenges in long-term stability, surgical risk, and scalability. Non-invasive techniques offer safe, accessible brain monitoring but are constrained by intrinsic physical limits on resolution. Future directions point toward multimodal integration, advanced signal processing algorithms, and emerging technologies like functional ultrasound that may bridge the current resolution gap. For biomedical research and drug development, selection criteria must align technique capabilities with specific application requirements, whether for high-precision neuroprosthetic control, large-scale brain dynamics mapping, or therapeutic intervention monitoring. Continued innovation promises to expand the boundaries of what is measurable in the human brain, driving advances in both fundamental neuroscience and clinical applications.
References
- 1. 3.4 Comparison of invasive and non-invasive techniques [https://fiveable.me/brain-computer-interfaces/unit-3/comparison-invasive-non-invasive-techniques/study-guide/zessvADvpYjeDIN7]
- 2. Ultra-high-density Neuropixels probes improve detection ... [https://www.sciencedirect.com/science/article/pii/S0896627325006658]
- 3. From Neurons to Cognition: Technologies for Precise ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10521756/]
- 4. A New Path to Noninvasive Brain-Computer Interface [https://www.jhuapl.edu/news/news-releases/241114-noninvasive-brain-computer-interface]
- 5. Towards decoding individual words from non-invasive ... [https://www.nature.com/articles/s41467-025-65499-0]
- 6. A brain-wide map of neural activity during complex behaviour [https://www.nature.com/articles/s41586-025-09235-0]
- 7. Revealing Spatiotemporal Neural Activation Patterns in ... [https://www.sciencedirect.com/science/article/pii/S2772528625000470]
- 8. Real-time, neural signal processing for high-density brain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12275325/]
- 9. Non-invasive action potential recordings using printed ... [https://www.nature.com/articles/s41467-025-63484-1]
- 10. From Neurons to Circuits: Linear Estimation of Local Field ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2924964/]
- 11. Modulating Brain Activity with Invasive Brain–Computer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9856340/]
- 12. Local Field Potential - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/local-field-potential]
- 13. Electrophysiological Source Imaging: a Noninvasive Window ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7941524/]
- 14. The history, current state and future possibilities of the non- ... [https://www.sciencedirect.com/science/article/pii/S2590093525000049]
- 15. novel noninvasive EEG proxy markers of the neural ... [https://www.nature.com/articles/s41398-022-02218-z]
- 16. A novel Rosetta stone for deciphering brain activity [https://www.sciencedirect.com/science/article/pii/S0165027024001055]
- 17. Invasive vs. Non-Invasive Neuronal Signals for Brain-Machine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4921501/]
- 18. Making a case for endovascular approaches for neural and... recording [https://pmc.ncbi.nlm.nih.gov/articles/PMC9928900/]
- 19. The neuronal sources of EEG: modeling of simultaneous scalp and... [https://inserm.hal.science/file/index/docid/285582/filename/index.xhtml]
- 20. μECoG Recordings Through a Thinned Skull - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC6779785/]
- 21. Unlocking the Potential of EEG Data [https://www.linkedin.com/pulse/unlocking-potential-eeg-data-chanchal-m-]
- 22. Recent Trends in Non-invasive Neural Recording Based ... [https://www.frontiersin.org/journals/computational-neuroscience/articles/10.3389/fncom.2022.875282/full]
- 23. Cortical Pyramidal and Parvalbumin Cells Exhibit Distinct ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9744183/]
- 24. Enhanced spatiotemporal resolution imaging of neuronal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7778454/]
- 25. Pyramidal cell types drive functionally distinct cortical ... [https://www.nature.com/articles/s41593-022-01245-9]
- 26. Invasive neurophysiology and whole brain connectomics ... [https://www.nature.com/articles/s41551-025-01467-9]
- 27. In vivo neural activity of electrosensory pyramidal cells [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1013711]
- 28. Invasive vs. Non-Invasive Neuronal Signals for Brain- ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2016.00295/full]
- 29. ECoG, Utah Arrays, and Neuropixels in BCI [https://michaelguth.com/?p=2620]
- 30. Advances in large-scale electrophysiology with high-density ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12394932/]
- 31. Minimally invasive implantation of scalable high-density ... [https://www.nature.com/articles/s41551-025-01501-w]
- 32. Multi-Channel Neural Interface for Neural Recording and ... [https://pubmed.ncbi.nlm.nih.gov/40995804/]
- 33. A Brain Foundation Model for Unified EEG and MEG Signals [https://arxiv.org/html/2505.18185v1]
- 34. Using Magnetoencephalography to Advance the Science of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12451052/]
- 35. Estimating Brain Activity with High Spatial and Temporal ... [https://arxiv.org/html/2510.09415v1]
- 36. Exploring the limits of MEG spatial resolution with ... [https://www.sciencedirect.com/science/article/pii/S105381192300099X]
- 37. Comparing MEG and EEG measurement set-ups for a brain ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0319328]
- 38. The Mismatch Negativity Compared: EEG, SQUID-MEG ... [https://pubmed.ncbi.nlm.nih.gov/41014290/]
- 39. Disrupted temporal structure of the M/EEG meta-states ... [https://www.sciencedirect.com/science/article/pii/S1053811925005580]
- 40. Robust interpolation of EEG/MEG sensor time-series via ... [https://pubmed.ncbi.nlm.nih.gov/39719120/]
- 41. Opportunities for use of neuroimaging in de-risking drug ... [https://www.nature.com/articles/s41386-024-01970-8]
- 42. Why Brain Wearables Are Making Traditional EEG Labs ... [https://globalrph.com/2025/10/why-brain-wearables-are-making-traditional-eeg-labs-obsolete-in-2025/]
- 43. Functional near-infrared spectroscopy for the assessment ... [https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2025.1524806/full]
- 44. of Comparison with other neuroimaging modalities – fNIRS vs.... fNIRS [https://www.artinis.com/blogpost-all/comparison-fnirs-vs-fmri]
- 45. Advances in functional magnetic resonance imaging- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12093097/]
- 46. Applications of Functional Near-Infrared Spectroscopy (fNIRS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12347915/]
- 47. vs EEG: fNIRS Non-Invasive Brain Comparing ... - iMotions Imaging [https://imotions.com/blog/learning/research-fundamentals/fnirs-vs-eeg/]
- 48. Advanced preclinical functional magnetic resonance ... [https://www.nature.com/articles/s44303-025-00085-z]
- 49. Multimodal assessment of the spatial correspondence between fNIRS ... [https://www.nature.com/articles/s41598-023-29123-9?error=cookies_not_supported&code=6e5efe04-30ef-46bc-a396-c114efc67cf5]
- 50. Classify the fNIRS signals of first-episode drug-naive MDD patients... [https://bmcpsychiatry.biomedcentral.com/articles/10.1186/s12888-025-07394-y]
- 51. Estimating Brain Activity with High Spatial and Temporal ... [https://arxiv.org/abs/2510.09415]
- 52. Functional ultrasound (fUS) detects mild cerebral alterations ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12406050/]
- 53. Functional ultrasound imaging combined with machine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12423641/]
- 54. Spatiotemporal Clustering of Functional Ultrasound ... [https://www.eneuro.org/content/12/2/ENEURO.0438-24.2025]
- 55. Deep-Learning Based Super-Resolution Functional ... [https://arxiv.org/abs/2511.14071]
- 56. High-resolution transcranial optical imaging of in vivo ... [https://www.nature.com/articles/s41598-024-70876-8]
- 57. PhysMed team to present advances in functional ultrasound ... [https://www.physicsformedicine.espci.fr/ieee-ius-2025/]
- 58. Spatial and temporal resolutions of EEG - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC4548479/]
- 59. What limits the performance of current invasive brain ... [https://www.frontiersin.org/journals/systems-neuroscience/articles/10.3389/fnsys.2014.00068/full]
- 60. Invasive and Non-Invasive Neuromodulation for the Treatment ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12294003/]
- 61. Plugging in to Human Memory: Advantages, Challenges ... [https://www.sciencedirect.com/science/article/pii/S0092867419311663]
- 62. Magnetoencephalographic source localization and ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1578473/full]
- 63. Review on solving the inverse problem in EEG source analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC2605581/]
- 64. Multimodal quantitative analysis guides precise ... [https://pubmed.ncbi.nlm.nih.gov/40815323/]
- 65. 技术原理与优缺点对比_非侵入式脑机接口技术路径 [https://blog.csdn.net/yuntongliangda/article/details/149350929]
- 66. 基于深度学习的非侵入性大脑信号研究(上) 翻译 [https://blog.csdn.net/cc1609130201/article/details/131996735]
- 67. CN113827253A - 计算设备和从脑电信号中去除噪声的方法 [https://patents.google.com/patent/CN113827253A/zh]
- 68. 基于高能效机器学习的可持续医学成像去噪技术 [https://www.jove.com/cn/t/68968/energy-efficient-machine-learning-based-denoising-techniques-for]
- 69. jove.com/cn/t/53466/combined-invasive-subcortical-non-invasive... [https://www.jove.com/cn/t/53466/combined-invasive-subcortical-non-invasive-surface-neurophysiological]
- 70. CN110222843A - 一种噪声预测系统及方法 [https://patents.google.com/patent/CN110222843A/zh]
- 71. Effective Techniques for Reducing Electrical Noise [https://www.ai-futureschool.com/en/electrotechnics/effective-techniques-for-reducing-electrical-noise.php]
- 72. High-resolution neural recordings improve the accuracy of ... [https://www.nature.com/articles/s41467-023-42555-1]
- 73. Techniques to Improve Your Signal-to-Noise Ratio [https://www.nutsvolts.com/magazine/article/march2016_SignalToNoiseRatio]
- 74. Improving SNR and Determining Optimal Excitation Levels [https://www.ni.com/en/support/documentation/supplemental/21/making-accurate-strain-measurements-improving-snr-and-determining-optimal-excitation-levels.html?srsltid=AfmBOopJKO9cxE0CMt6QtTUQ0qUR0Nf5bXJjT017BD1g945Ir6b3H9iG]
- 75. How to Achieve Consistent Analog Signal Performance in ... [https://runtimerec.com/how-to-achieve-consistent-analog-signal-performance-in-noisy-environments/]
- 76. Rolling bearing fault diagnosis in noisy environments using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12504641/]
- 77. Non-Invasive Brain-Computer Interfaces: State of the Art and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11861396/]
- 78. Effective Techniques for Multimodal Data Fusion - PMC - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC10007548/]
- 79. Multimodal Data Fusion: Key Techniques, Challenges & ... [https://www.sapien.io/blog/mastering-multimodal-data-fusion]
- 80. Multimodal Data Fusion - an overview [https://www.sciencedirect.com/topics/computer-science/multimodal-data-fusion]
- 81. Decoding non-invasive brain activity with novel deep- ... [https://arxiv.org/abs/2510.24733]
- 82. Impact of implant surface modifications on long-term ... [https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2025.1661369/full]
- 83. Open Data In Neurophysiology: Advancements, Solutions ... [https://www.eneuro.org/content/12/11/ENEURO.0486-24.2025]
- 84. The Future of Dental Implants: A Narrative Review ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12442331/]
- 85. Outlook for Noninvasive Brain-Computer Interfaces for AI [https://etcjournal.com/2025/09/21/outlook-for-noninvasive-brain-computer-interfaces-for-ai/]
- 86. 2.3: Non-Invasive Techniques- Direct Functional Imaging ... [https://socialsci.libretexts.org/Courses/Sacramento_City_College/Psyc_310%3A_Biological_Psychology_(Keys)/02%3A_Research_Methods_and_Ethical_Considerations_of_Biological_Psychology_and_Neuroscience/2.03%3A_Non-Invasive_Techniques-_Direct_Functional_Imaging_Techniques]
- 87. Comparative accuracy of invasive versus non- ... [https://eureka.patsnap.com/report-comparative-accuracy-of-invasive-versus-non-invasive-brain-computer-interfaces-approaches]
- 88. a direct comparison of their effects via scalp EEG recordings [https://www.sciencedirect.com/science/article/pii/S1935861X25002888]
- 89. Performance assessment in brain-computer interface-based ... [https://biomedical-engineering-online.biomedcentral.com/articles/10.1186/1475-925X-12-43]
- 90. Think Fast | Setting new standards and new records for ... [https://www.paradromics.com/blog/bci-benchmarking]
- 91. Recent applications of EEG-based brain-computer-interface in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11931852/]
- 92. Non-Invasive Brain Stimulation and Artificial Intelligence in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12110241/]
- 93. Harnessing the synergy of statistics and deep learning for BCI ... [https://braininformatics.springeropen.com/articles/10.1186/s40708-025-00250-5]
- 94. Mental privacy: navigating risks, rights and regulation [https://pmc.ncbi.nlm.nih.gov/articles/PMC12287510/]
- 95. A generic non-invasive neuromotor interface for human-computer interaction | Nature [https://www.nature.com/articles/s41586-025-09255-w]
- 96. Invasive Brain-Computer Interfaces safety considerations ... [https://eureka.patsnap.com/report-invasive-brain-computer-interfaces-safety-considerations-in-long-term-deployment]
- 97. A direct comparison of laboratory and community EEG ... [https://www.nature.com/articles/s41598-025-03569-5]
- 98. A low-latency neural inference framework for real-time ... [https://www.nature.com/articles/s41598-025-24972-y]
- 99. Foundation and Large-Scale AI Models in Neuroscience [https://arxiv.org/html/2510.16658v1]
- 100. Validation framework for in vivo digital measures - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11751004/]
- 101. Towards A Non-Invasive Method to Unlock Brain's ... [https://viterbischool.usc.edu/news/2025/04/towards-a-non-invasive-method-to-unlock-brains-understanding-of-speech/]
- 102. A Transformer-Based Framework With Data Augmentation ... [https://pubmed.ncbi.nlm.nih.gov/40905387/]