Benchmarking BCI Performance: A 2025 Analysis of Invasive vs. Non-Invasive Technologies for Biomedical Research
This article provides a comprehensive analysis of performance benchmarks for invasive and non-invasive Brain-Computer Interfaces (BCIs), tailored for researchers and drug development professionals.
Benchmarking BCI Performance: A 2025 Analysis of Invasive vs. Non-Invasive Technologies for Biomedical Research
Abstract
This article provides a comprehensive analysis of performance benchmarks for invasive and non-invasive Brain-Computer Interfaces (BCIs), tailored for researchers and drug development professionals. It explores the fundamental principles, trade-offs, and signal characteristics of each approach. The scope covers the latest methodological advances in neural decoding, key application areas from robotic control to communication, and critical challenges in signal stability and real-world usability. A central focus is the emerging framework for objective performance validation, centered on metrics like Information Transfer Rate (ITR) and latency, with a comparative analysis of leading commercial and research systems. This review synthesizes the current state-of-the-art to inform strategic R&D and clinical trial design in neurotechnology.
Defining the Landscape: Core Principles and Performance Trade-offs of Invasive and Non-Invasive BCIs
Brain-Computer Interfaces (BCIs) represent a revolutionary technology that enables direct communication between the brain and external devices, bypassing conventional neuromuscular pathways [1]. These systems hold transformative potential for restoring function to individuals with disabilities and enhancing human-computer interaction. BCIs can be broadly categorized into invasive and non-invasive approaches based on the placement of signal acquisition components [2]. Invasive BCIs require surgical implantation of electrodes either directly into brain tissue (intracortical) or on the surface of the brain (electrocorticography, ECoG). Non-invasive BCIs measure brain activity from outside the skull using technologies such as electroencephalography (EEG), functional near-infrared spectroscopy (fNIRS), and magnetoencephalography (MEG) [2] [3]. The fundamental distinction between these approaches involves a critical trade-off: invasive methods provide higher signal quality and spatial resolution at the cost of surgical risk and long-term stability concerns, while non-invasive methods offer greater safety and accessibility but with reduced signal resolution and increased susceptibility to noise [1]. This comparative analysis examines the technical specifications, performance benchmarks, experimental methodologies, and research applications of these BCI paradigms within the context of ongoing performance optimization research.
Technical Specifications and Performance Benchmarks
Fundamental Characteristics of BCI Modalities
Invasive BCI Approaches involve direct contact with brain tissue, achieving superior signal quality. Intracortical microelectrode arrays (such as Utah Arrays) penetrate the cortex to record action potentials and local field potentials from individual neurons or small neuronal populations [4] [2]. These systems provide the highest spatial resolution (micron-scale) and temporal resolution (millisecond-scale), enabling precise decoding of movement intentions and complex commands. Alternatively, Electrocorticography (ECoG) utilizes electrode grids placed on the surface of the dura mater (epidural) or beneath it (subdural) [4]. ECoG records local field potentials from larger neuronal populations with higher spatial resolution and signal-to-noise ratio than non-invasive methods, but with less granularity than intracortical approaches. A key advantage of ECoG is its demonstrated long-term stability, with studies reporting maintained signal quality and decoder performance over five years in fully implanted systems [4].
Non-Invasive BCI Approaches measure brain activity through the skull and scalp. Electroencephalography (EEG) records electrical activity from the scalp using electrode caps, providing excellent temporal resolution (milliseconds) but limited spatial resolution (centimeters) due to signal smearing by skull and scalp tissues [1] [5]. Functional Near-Infrared Spectroscopy (fNIRS) measures hemodynamic responses by detecting light absorption in brain tissue, offering better spatial resolution than EEG but poorer temporal resolution due to the slow nature of blood flow changes [3]. Magnetoencephalography (MEG) detects the magnetic fields generated by neuronal activity, providing improved spatial resolution compared to EEG, but requires bulky, expensive shielded rooms, limiting its practical application for BCIs [6] [3].
Table 1: Technical Specifications of Major BCI Paradigms
| Parameter | Intracortical | ECoG | EEG | fNIRS | MEG |
|---|---|---|---|---|---|
| Spatial Resolution | Micron-scale | Millimeter-scale | Centimeter-scale | Centimeter-scale | Millimeter-scale |
| Temporal Resolution | <1 ms | 1-10 ms | ~10-100 ms | 1-10 seconds | <1 ms |
| Signal Type | Action potentials, Local Field Potentials | Local Field Potentials | Scalp potentials | Hemodynamic (oxygenation) | Magnetic fields |
| Risk Level | High (surgical implantation) | Moderate-High (surgical implantation) | Minimal | Minimal | Minimal |
| Long-Term Stability | Signal degradation over time [4] | Stable for years in chronic implants [4] | Stable with proper setup | Stable with proper setup | Stable with proper setup |
| Typical Applications | Dexterous prosthetic control, complex communication | Motor control, communication, seizure monitoring [4] | Basic communication, environmental control, neurofeedback | Cognitive state monitoring, basic BCIs | Brain mapping, research |
| Key Advantage | Highest information transfer rate | Balance of signal quality and stability | Widely accessible, safe | Less susceptible to motion artifacts | Excellent spatiotemporal resolution |
| Primary Limitation | Tissue response, signal degradation | Limited cortical coverage, surgical risk | Low spatial resolution, noise susceptibility | Low temporal resolution | Bulky equipment, high cost |
Quantitative Performance Comparison
Performance benchmarking for BCIs utilizes standardized metrics including classification accuracy, information transfer rate (ITR), signal-to-noise ratio, and long-term stability. Invasive BCIs typically achieve superior performance across most metrics, particularly for complex control tasks.
Table 2: Performance Benchmarks for BCI Paradigms
| Metric | Intracortical | ECoG | EEG | fNIRS |
|---|---|---|---|---|
| Classification Accuracy | >95% for movement decoding [2] | 95.9% AUROC for motor imagery over 54 months [4] | 76.7% for motor imagery with advanced processing [5] | 70-80% for binary tasks |
| Information Transfer Rate (bits/min) | 200-300 [2] | 100-200 | 20-60 | 5-15 |
| Signal-to-Noise Ratio | Very High | High | Low-Medium | Medium |
| Daily Use Pattern | N/A | 38±24 minutes in home use [4] | Session-based (1-2 hours) | Session-based (1-2 hours) |
| Channels Typically Used | 64-256 | 4-64 [4] | 8-64 (up to 128+) | 16-64 |
| Setup Time | Surgical procedure | Surgical procedure | 10-30 minutes | 5-15 minutes |
Recent research demonstrates the performance boundaries of each approach. A 5-year follow-up study of a fully implanted ECoG system showed remarkable stability with an average decoder area under the receiver operating characteristic curve (AUROC) of 0.959 for motor intention detection during home use [4]. Meanwhile, non-invasive approaches continue to narrow the performance gap through advanced signal processing, with one EEG study achieving 76.7% classification accuracy for motor imagery using only eight optimized EEG channels with the CPX (CFC-PSO-XGBoost) pipeline [5].
Experimental Protocols and Methodologies
Signal Acquisition Pathways
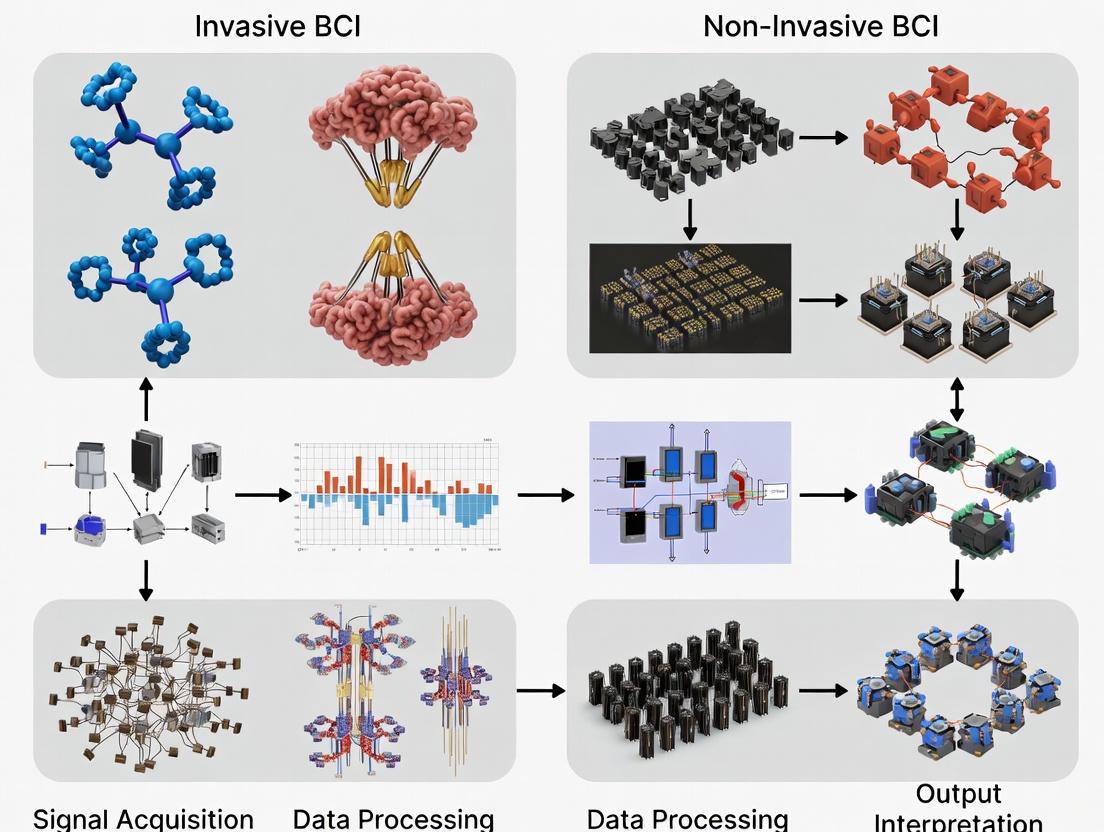
The experimental workflow for BCI research follows a structured pathway from signal acquisition to device control. The fundamental differences between invasive and non-invasive approaches begin at the acquisition stage, which subsequently influences all downstream processing requirements.
Standardized Evaluation Frameworks
Comprehensive evaluation of BCI systems extends beyond basic performance metrics to include usability, user satisfaction, and real-world applicability [7]. The transition from offline analysis to online closed-loop testing represents a critical milestone in BCI validation, with online evaluation considered the "gold standard" for assessing practical utility [7].
Standardized experimental protocols vary by BCI paradigm:
Motor Imagery Protocols: For both invasive and non-invasive approaches, motor imagery tasks involve users imagining limb movements without physical execution. In EEG-based systems, protocols typically involve cue-based trials where users imagine specific movements (e.g., left hand vs. right hand) with random inter-trial intervals [5]. Signal processing incorporates techniques like Common Spatial Patterns (CSP) or Cross-Frequency Coupling (CFC) for feature extraction, followed by classification with algorithms such as Linear Discriminant Analysis or XGBoost [5].
P300 Speller Protocols: This non-invasive approach presents matrix layouts where rows and columns flash sequentially while users count flashes of target characters [8]. Protocols standardize flash duration, inter-stimulus intervals, and matrix sizes, with evaluation metrics including character selection accuracy and information transfer rate [9].
Long-Term Stability Assessment: For invasive BCIs, chronic implantation studies monitor signal quality metrics (signal-to-noise ratio, electrode impedance) and decoder performance (AUROC, classification accuracy) over months to years, with recent ECoG studies demonstrating stable performance over 54 months of home use [4].
The Researcher's Toolkit
Advancing BCI research requires specialized tools and methodologies tailored to each paradigm. The following table outlines essential research reagents and solutions for working with different BCI approaches.
Table 3: Essential Research Tools for BCI Paradigms
| Tool Category | Specific Examples | Research Function | Compatible Paradigms |
|---|---|---|---|
| Electrode Technologies | Utah Microelectrode Arrays, Neuropixels probes, Medtronic Activa PC+S with Resume II leads [4] | High-resolution neural signal acquisition | Intracortical, ECoG |
| Signal Processing Algorithms | Common Spatial Patterns (CSP), Cross-Frequency Coupling (CFC), Phase-Amplitude Coupling (PAC) [5] | Feature extraction for classification | EEG, ECoG, Intracortical |
| Classification Frameworks | XGBoost, FBCNet, EEGNet, CSP-based classifiers [5] | Intent decoding from neural features | All paradigms |
| Optimization Methods | Particle Swarm Optimization (PSO) for channel selection [5] | Optimizing electrode montages | EEG, fNIRS |
| Validation Toolboxes | BCI Competition datasets, OpenVibe, BCILAB | Standardized performance assessment | All paradigms |
| Performance Metrics | Area Under ROC Curve (AUROC), Information Transfer Rate, Classification Accuracy, F1 Score [4] [9] [5] | Quantitative performance benchmarking | All paradigms |
| Usability Assessment | Quebec User Evaluation of Satisfaction with Assistive Technology (QUEST), System Usability Scale (SUS) | Evaluating practical implementation [7] | All paradigms |
The BCI field continues to evolve with both invasive and non-invasive approaches demonstrating progressive improvements. Invasive BCIs are trending toward less risky implantation procedures and more stable long-term performance, with recent research focusing on fully implanted wireless systems that enable extended home use [4] [2]. Non-invasive approaches are benefiting from advanced signal processing techniques, multimodal integration (EEG+fNIRS), and artificial intelligence applications that continue to narrow the performance gap with invasive methods [6] [5] [2].
Market analysis projects steady growth in the BCI field, with the overall market forecast to exceed US$1.6 billion by 2045, representing a compound annual growth rate of 8.4% since 2025 [3]. Both invasive and non-invasive technologies are expected to find specialized applications across medical, research, assistive technology, and eventually consumer markets.
The choice between invasive and non-invasive BCI paradigms ultimately depends on the specific application requirements and risk-benefit considerations. For critical applications requiring high precision, such as advanced prosthetic control for individuals with severe disabilities, invasive approaches may justify their surgical risks [1] [4]. For broader applications where safety and accessibility are paramount, non-invasive approaches offer increasingly viable alternatives [1] [2]. As both trajectories advance, the future of BCI research will likely focus on hybrid approaches that leverage the strengths of multiple paradigms, personalized adaptations to individual user capabilities, and comprehensive evaluation frameworks that assess real-world usability alongside technical performance metrics [7].
The development of Brain-Computer Interfaces (BCIs) is fundamentally governed by a persistent engineering and clinical challenge: the inverse relationship between signal fidelity and procedural safety. As BCI technologies transition from laboratory research to clinical applications and potential consumer use, understanding and navigating this trade-off becomes paramount for researchers, clinicians, and developers. Higher-quality neural signals, which enable more complex and reliable control of external devices, typically require increasingly invasive procedures that carry greater surgical risk and ethical considerations [10] [11]. This dichotomy forms the central axis along which all BCI technologies are positioned, balancing the need for information-rich data against the imperative of patient safety.
The terminology describing this spectrum has evolved, reflecting a more nuanced understanding of the risks involved. While the broad categories of "invasive" and "non-invasive" remain common, recent frameworks propose more precise classifications. One influential model introduces a two-dimensional view, evaluating BCIs based on the invasiveness of the surgical procedure (non-invasive, minimal-invasive, invasive) and the operating location of the sensors (non-implantation, intervention, implantation) [10]. This refined taxonomy allows for a more accurate assessment of the risk-profile associated with each technology, moving beyond a simple binary distinction to inform better clinical decision-making [12].
A Comparative Framework for BCI Signal Acquisition Technologies
BCI signal acquisition methods can be categorized based on the degree of intrusion into the body, which directly correlates with both the potential signal quality and the associated clinical risk. The following table summarizes the key characteristics, advantages, and limitations of the primary BCI categories.
Table 1: Comparison of Major BCI Signal Acquisition Technologies
| Technology Category | Representative Modalities | Spatial Resolution | Key Advantages | Primary Limitations |
|---|---|---|---|---|
| Non-Invasive | Electroencephalography (EEG), functional Near-Infrared Spectroscopy (fNIRS) | Low (cm-scale) | Safe; No surgery required; High temporal resolution; Lower cost [11] | Low signal-to-noise ratio; Signal attenuation by skull & tissues; Susceptible to artifacts [10] [11] |
| Minimal-Invasive | Endovascular (Stentrode), Epidural/ESubdural Electrodes | Medium (mm-scale) | Higher signal quality than non-invasive; Reduced tissue trauma vs. fully invasive; Potentially lower chronic immune response [13] [10] | Limited brain coverage; Surgical procedure still required; Long-term biocompatibility questions [14] |
| Fully Invasive | Intracortical Microelectrode Arrays (e.g., Utah Array, Neuralink) | High (µm-scale) | Very high spatial & temporal resolution; Records single-neuron activity [13] | Highest surgical risk (infection, bleeding); Tissue damage & scarring; Long-term stability & biocompatibility challenges [3] [13] |
This relationship between sensor location and theoretical signal quality can be visualized as a function of invasiveness. The following diagram illustrates the fundamental trade-off and the general classification of major BCI technologies within this framework.
Figure 1: The fundamental BCI design trade-off. Signal quality improves with greater invasiveness, analogous to moving closer to a sound source. Non-implantation methods like EEG have a low theoretical signal ceiling, while implantation methods can achieve high fidelity but require penetrating brain tissue [10].
The Surgical and Detection Dimensions
A more granular understanding of BCI technologies can be achieved by considering the two-dimensional framework proposed in recent literature, which synthesizes clinical (surgical) and engineering (detection) perspectives [10]. The surgery dimension focuses on the anatomical trauma caused by the procedure, while the detection dimension focuses on the final operational location of the sensor. This model is crucial for cross-disciplinary dialogue, ensuring that clinicians and engineers have a shared understanding of both the risks and capabilities of a given BCI.
Table 2: Two-Dimensional Classification of BCI Technologies
| Detection Dimension | Non-Invasive Surgery | Minimal-Invasive Surgery | Invasive Surgery |
|---|---|---|---|
| Non-Implantation | EEG, fNIRSElectrodes on scalp. No anatomical trauma [10]. | - | - |
| Intervention | - | Endovascular StentrodeDeployed via blood vessels. Trauma spares brain tissue [10] [14]. | - |
| Implantation | - | ECoG GridsPlaced on brain surface. May require craniotomy but doesn't pierce tissue [13]. | Intracortical ArraysPenetrate brain tissue, causing micron-scale trauma [10]. |
Experimental Protocols & Performance Benchmarks
Quantifying the Performance Gap
The theoretical trade-off between signal quality and invasiveness is borne out in empirical performance data. The following table summarizes key performance metrics for different BCI modalities, illustrating the clear performance gradient.
Table 3: Experimental Performance Benchmarks Across BCI Types
| BCI Type & Paradigm | Information Transfer Rate (Bits/min) | Accuracy (%) | Key Applications in Research |
|---|---|---|---|
| Non-Invasive (EEG - Motor Imagery) | 5 - 25 | 70 - 85 | Control of robotic arms, wheelchair navigation [15] |
| Non-Invasive (EEG - P300 Speller) | ~20 | ~80 | Character spelling for communication [15] |
| Non-Invasive (AI-Enhanced EEG) | N/A | Performance increased 3.9x with AI copilot [16] | Cursor control, robotic arm control [16] |
| Invasive (Intracortical - Speech Decoding) | N/A | Up to 99% word inference [13] | Restoration of speech for paralyzed individuals [13] |
| Invasive (Intracortical - Motor Control) | N/A | High-fidelity 2D cursor control [14] | Control of computer interfaces, robotic limbs |
Detailed Experimental Protocol: AI-Enhanced Non-Invasive BCI
A landmark study from UCLA demonstrates how advanced algorithms can mitigate the limitations of non-invasive BCIs, offering a pathway to improved performance without increased surgical risk [16].
1. Objective: To significantly improve the performance of a non-invasive BCI for cursor and robotic arm control by using an artificial intelligence (AI) copilot to interpret noisy EEG signals.
2. Signal Acquisition:
- Equipment: A 64-channel EEG cap was used for signal acquisition [16].
- Subjects: The study involved three healthy participants and one participant with paraplegia due to a T5-level spinal cord injury [16].
3. Processing & Decoding:
- Algorithm: A hybrid Convolutional Neural Network-Kalman Filter (CNN-KF) was developed.
- CNN Role: The Convolutional Neural Network learned features from the EEG data to predict the user's intended movement direction. It is particularly effective for processing spatial and temporal patterns in noisy time-series data [16].
- KF Role: The Kalman filter provided a recursive method for estimating the unknown state of the cursor or robotic arm (e.g., position, velocity) based on the noisy input from the CNN, effectively smoothing the trajectory and improving control [16].
4. The AI Copilot & Shared Autonomy:
- This component introduced "shared autonomy," where the AI actively collaborates with the user. The AI copilot uses observations of the environment (e.g., the location of a target) to change the distribution of possible actions, effectively assisting the user in achieving the task goal. For instance, it might make it easier for the cursor to move toward a visible target [16].
5. Outcome:
- The system led to a 3.9-fold performance improvement for the paralyzed participant in both cursor control and robotic arm tasks. Critically, the participant could not complete the tasks without the AI copilot, highlighting the transformative potential of this software-based approach to overcoming hardware limitations [16].
The workflow of this closed-loop BCI system, which is representative of modern BCI experiments, is detailed below.
Figure 2: The closed-loop BCI workflow. Neural signals are acquired, processed, and decoded by AI algorithms. The resulting command drives an external device, and the visual feedback provided to the user allows them to adapt their mental strategy, creating a continuous loop [16] [10] [15].
The Scientist's Toolkit: Essential Research Reagents & Materials
The advancement of BCI research relies on a suite of specialized materials, hardware, and software solutions. The following table details key components of the modern BCI researcher's toolkit.
Table 4: Essential Reagents and Materials for BCI Research
| Category / Item | Specific Examples | Function & Application in Research |
|---|---|---|
| Electrode Types | Wet Gel Electrodes, Dry Electrodes, Microneedle Arrays | Function: Transduce ionic currents in the body into electrical signals for recording. Application: Dry electrodes are a key innovation for improving the usability and setup time of non-invasive EEG systems, potentially facilitating consumer adoption [3]. |
| Implantable Arrays | Utah Array (Blackrock Neurotech), Neuralace (Blackrock), N1 Implant (Neuralink) | Function: High-density recording or stimulation of neuronal activity. Application: The gold-standard for high-fidelity invasive BCI research; newer technologies like Neuralace aim to increase channel count and reduce tissue damage through flexible substrates [3] [13]. |
| Signal Acquisition Systems | Neuroport (Blackrock), Synchron Stentrode, OpenBCI Kits | Function: Amplify, filter, and digitize analog neural signals from electrodes. Application: Research-grade systems are used in clinical trials (e.g., Stentrode) while open-source platforms like OpenBCI lower the barrier to entry for non-invasive BCI prototyping [13] [16]. |
| Decoding Algorithms | Convolutional Neural Networks (CNN), Kalman Filters, Support Vector Machines (SVM) | Function: Translate raw or pre-processed neural signals into intended user commands. Application: Deep learning models (CNNs) are increasingly used for their pattern recognition capabilities in noisy signals, while Kalman filters are effective for smoothing continuous trajectories like cursor movement [16] [10]. |
| Biocompatible Materials | PEDOT:PSS, Graphene, parylene-C | Function: Coat electrodes or form the substrate of flexible implants. Application: Critical for improving the long-term stability and signal-to-noise ratio of invasive BCIs by reducing the immune response and improving biocompatibility [3]. |
The trade-off between signal fidelity and procedural risk remains a defining feature of the BCI landscape. Non-invasive technologies like EEG and fNIRS offer safety and accessibility but are constrained by a low theoretical ceiling for signal quality, limiting their application to relatively simple control tasks. Conversely, invasive technologies provide the high bandwidth necessary for complex applications like speech decoding and dexterous motor control but are accompanied by significant surgical risks and long-term biocompatibility challenges [13] [11].
The future of BCI research is focused on breaking this trade-off through engineering and clinical innovation. Key directions include:
- Algorithmic Advancements: As demonstrated by the AI copilot study, sophisticated machine learning and shared autonomy models can extract more information from existing signal sources, effectively improving performance without changing the hardware's inherent risk profile [16].
- Material Science: The development of more biocompatible, flexible, and high-density electrode arrays aims to reduce the foreign body response and improve the long-term stability of invasive devices, thereby increasing their safety and effective lifespan [3].
- Novel Form Factors: Approaches like endovascular BCIs (e.g., Synchron's Stentrode) and ultra-thin cortical surface arrays (e.g., Precision Neuroscience's Layer 7) seek to occupy a "middle ground," offering higher signal quality than non-invasive methods with a lower risk profile than traditional penetrating electrodes [13] [14].
For researchers and clinicians, the choice of a BCI platform must be a deliberate calculation, balancing the information requirements of the intended application against the acceptable level of clinical risk for the target population. As the field progresses, the continued refinement of both hardware and software promises to push the boundaries of what is possible, gradually flattening the curve of the inherent trade-off that has long guided BCI design.
Brain-computer interfaces (BCIs) represent a revolutionary technology that enables direct communication between the brain and external devices, offering transformative potential for individuals with paralysis, spinal cord injuries, and other motor impairments [3] [13]. As both invasive and non-invasive BCI technologies advance toward clinical and consumer applications, rigorous benchmarking of their signal quality becomes paramount for researchers, clinicians, and developers. The core performance characteristics of any BCI system—spatial resolution, temporal resolution, and signal-to-noise ratio (SNR)—exist in a complex trade-space that fundamentally determines their capabilities and suitable applications [3] [17].
Spatial resolution refers to a system's ability to distinguish between separate neural sources located in close proximity within the brain, typically measured in millimeters [17]. Temporal resolution describes the precision with which a system can track changes in neural activity over time, commonly measured in milliseconds [17]. SNR quantifies the ratio between the power of meaningful neural signals and background noise, determining how clearly neural information can be extracted from recorded data [18] [19]. Understanding the relationships and inherent trade-offs between these three fundamental parameters is essential for selecting appropriate technologies for specific research questions and clinical applications.
This comparison guide provides an objective analysis of these critical performance metrics across major BCI modalities, presenting synthesized experimental data and methodological protocols to inform researchers and drug development professionals working at the intersection of neuroscience and neurotechnology. By framing these technical specifications within the context of invasive versus non-invasive BCI performance, we aim to equip scientists with the necessary information to make evidence-based decisions in their neurotechnology research and development efforts.
Core Performance Metrics in Neural Signal Acquisition
The performance of any brain-computer interface is governed by three interdependent metrics that form the foundation of signal quality assessment. These parameters not only determine the current capabilities of neural interfaces but also define their limitations and potential applications across research, clinical, and consumer domains.
Spatial resolution represents the granularity with which a neural interface can localize brain activity. In practical terms, it determines whether a system can distinguish activity in adjacent cortical columns or must instead aggregate signals across broader brain regions [17]. This metric is physically constrained by fundamental principles including sensor density, proximity to neural sources, and the biophysics of signal transmission through various biological tissues [17] [19].
Temporal resolution indicates the precision with which a system can track neural dynamics over time. High temporal resolution enables researchers to capture the precise timing and sequence of neural events, which is crucial for understanding brain dynamics, functional connectivity, and information processing pathways [17]. Unlike spatial resolution, temporal resolution is primarily limited by sampling rates and signal processing capabilities rather than fundamental physical barriers.
Signal-to-noise ratio (SNR) quantifies the relative strength of meaningful neural information compared to background interference and noise. Mathematically, SNR is defined as the ratio of signal power to noise power, often expressed in decibels (dB) [18]. In BCI systems, SNR determines the fidelity of recorded neural data and directly impacts the accuracy of decoding algorithms. As established by fundamental information theory, the maximum achievable information transmission capacity of any channel is governed by its bandwidth and SNR according to the Shannon-Hartley theorem [18] [19].
The relationship between these three metrics is not independent; instead, they exist in a delicate balance where optimizing one parameter often necessitates compromises in others. Theoretical and empirical studies have demonstrated that the square of the SNR is generally proportional to the "volume" of the spatial resolution unit, indicating a fundamental trade-off between these parameters in linear imaging systems [19]. This relationship has profound implications for BCI design and implementation across different modalities.
Comparative Analysis of BCI Modalities
The landscape of brain-computer interfaces can be broadly categorized into invasive and non-invasive approaches, each with distinct performance characteristics, advantages, and limitations. The following analysis systematically compares these modalities across the three core metrics of spatial resolution, temporal resolution, and SNR.
Table 1: Performance Comparison of Major BCI Technologies
| Technology | Spatial Resolution | Temporal Resolution | SNR | Primary Applications |
|---|---|---|---|---|
| EEG (Non-invasive) | Low (cm range) [17] | High (milliseconds) [17] [20] | Low [16] | Brain monitoring, basic research, consumer applications [3] |
| fNIRS (Non-invasive) | Moderate (~1-2 cm) [3] | Low (seconds) | Low-Moderate | Brain monitoring, assistive technology [3] |
| MEG (Non-invasive) | Moderate-High [3] | High (milliseconds) | Moderate | Cognitive neuroscience, clinical diagnostics [3] |
| ECoG (Invasive) | High (mm range) | High (milliseconds) | High | Medical applications, fundamental research [3] |
| Microelectrode Arrays (Invasive) | Very High (sub-mm) [3] [13] | Very High (sub-millisecond) | Very High [13] | Motor control, speech decoding, complex device control [13] |
Table 2: Technical Specifications and Market Positioning of BCI Technologies
| Technology | Invasiveness | Key Players/Examples | Market Forecast | Primary Limitations |
|---|---|---|---|---|
| EEG | Non-invasive | ANT Neuro, EMOTIV, NeuroSky [16] | Established market; 9.35% CAGR (2025-2032) [16] | Poor spatial resolution, low SNR [17] [16] |
| fNIRS | Non-invasive | Emerging research systems [3] | Emerging opportunity in assistive technology [3] | Limited temporal resolution, depth penetration |
| MEG | Non-invasive | Research institutions [3] | Niche research applications [3] | Requires shielded environments, expensive [3] |
| ECoG | Minimally invasive | Precision Neuroscience [13] | Growing medical applications [3] [13] | Requires cranial access, limited penetration depth |
| Microelectrode Arrays | Fully invasive | Neuralink, Blackrock Neurotech, Paradromics [3] [13] | 1.49% CAGR (2025-2032); $160.44B global market (2024) [13] [16] | Tissue response, long-term stability, surgical risk [3] [13] |
Non-Invasive BCI Technologies
Non-invasive BCIs record neural activity from outside the skull, offering greater accessibility and reduced risk at the cost of signal quality. Electroencephalography (EEG) represents the most established non-invasive approach, recording electrical activity through electrodes placed on the scalp [3] [17]. While EEG offers excellent temporal resolution on the millisecond scale, its spatial resolution is severely limited by the blurring effect of volume conduction—where neural signals must pass through cerebrospinal fluid, skull, and scalp tissues, each with different conductive properties [17]. This biological filtering effect spreads and distorts electrical potentials, resulting in limited spatial resolution typically in the centimeter range [17]. Additionally, the considerable distance between cortical sources and scalp electrodes dramatically reduces SNR, necessitating sophisticated signal processing to extract meaningful neural information [16].
Functional near-infrared spectroscopy (fNIRS) measures hemodynamic responses associated with neural activity using near-infrared light, providing moderate spatial resolution but limited temporal resolution due to the slow nature of blood flow changes [3]. Magnetoencephalography (MEG) detects magnetic fields generated by neural currents, offering both good temporal resolution and better spatial localization than EEG, as magnetic fields are less distorted by biological tissues [3]. However, MEG requires extremely sensitive sensors and heavily shielded environments, limiting its practical applications [3].
Recent advances in non-invasive BCIs have focused on overcoming these inherent limitations through improved sensor technologies and advanced signal processing. Dry electrode systems for EEG eliminate the need for conductive gels, improving usability while maintaining signal quality [3]. Most significantly, artificial intelligence and machine learning approaches are being deployed to enhance the effective performance of non-invasive systems. UCLA researchers recently demonstrated that an AI "copilot" system could improve BCI performance by a factor of 3.9 times for paralyzed participants in cursor control and robotic arm tasks, effectively compensating for inherent SNR limitations through sophisticated pattern recognition [16].
Invasive BCI Technologies
Invasive BCIs record neural signals directly from the cortical surface or within brain tissue, bypassing the signal-degrading barriers encountered by non-invasive approaches. Electrocorticography (ECoG) arrays are placed on the surface of the brain beneath the skull but do not penetrate neural tissue [3]. This approach provides higher spatial resolution and SNR than non-invasive methods while avoiding some of the long-term stability issues associated with penetrating electrodes [3] [13]. Companies like Precision Neuroscience are developing ultra-thin electrode arrays designed to be inserted through a small dural slit, conforming to the cortical surface while minimizing tissue damage [13].
Intracortical microelectrode arrays represent the most invasive and highest-performance approach, with devices like the Utah Array (Blackrock Neurotech) and Neuralink's N1 implant penetrating brain tissue to record from individual neurons [3] [13]. These systems offer exceptional spatial resolution at the sub-millimeter level, temporal resolution capable of capturing individual action potentials, and significantly higher SNR due to their proximity to neural sources [13]. The trade-offs include potential tissue response, encapsulation, and long-term stability challenges as the body reacts to foreign materials [3]. Emerging approaches like Neuralace (Blackrock Neurotech) aim to address these limitations through flexible, less invasive designs that distribute electrodes across a wider cortical area while minimizing tissue damage [13].
Endovascular approaches like Synchron's Stentrode represent an intermediate category, accessing neural signals through blood vessels rather than direct brain implantation [13]. This method offers improved safety compared to fully invasive approaches while providing higher signal quality than non-invasive alternatives, particularly for motor control applications [13].
Experimental Protocols and Methodologies
Standardized experimental protocols are essential for generating comparable data across different BCI technologies and research laboratories. This section outlines key methodologies for quantifying spatial resolution, temporal resolution, and SNR in neural interfaces.
Spatial Resolution Assessment
Spatial resolution is typically quantified by measuring the system's ability to distinguish between closely spaced neural sources or to accurately localize known activations. For invasive microelectrode arrays, spatial resolution can be assessed by measuring the distance at which two adjacent microelectrodes can discriminate signals from separate individual neurons [13]. This involves analyzing cross-correlation between channels and signal independence metrics while physically verifying electrode positions post-implantation.
For non-invasive systems like EEG, spatial resolution assessment requires specialized protocols due to the inherent limitations of scalp recordings. Researchers typically use phantom head models with simulated dipole sources at known locations [17]. The protocol involves:
- Placing current dipoles at precisely known locations within a realistically shaped head model filled with conductive solution
- Recording the resulting potentials at scalp electrode positions
- Applying source localization algorithms to estimate dipole locations from scalp recordings
- Calculating localization error as the distance between actual and estimated dipole positions [17]
Surface Laplacian (SL) transformation, also known as Current Source Density (CSD) estimation, can dramatically improve the effective spatial resolution of EEG by reducing volume conduction effects and reference electrode artifacts [17]. This mathematical transformation estimates the radial current flow through the skull at each electrode location, effectively sharpening the spatial distribution of recorded activity.
Temporal Resolution Assessment
Temporal resolution benchmarking involves measuring a system's ability to accurately track rapidly changing neural signals. The fundamental protocol involves presenting precisely timed stimuli or having participants perform tasks with known neural timing characteristics while recording neural responses [17].
A standard approach involves measuring auditory evoked potentials using precisely timed auditory stimuli and analyzing the latency and sharpness of the resulting P300 or other event-related potential components [17]. The protocol includes:
- Presenting auditory stimuli with precise millisecond timing
- Recording neural responses across multiple trials
- Averaging responses to improve SNR
- Measuring the latency and full-width at half-maximum (FWHM) of resulting peaks
- Comparing these measurements across different systems
For systems targeting motor control applications, participants may perform precisely timed movements or motor imagery tasks while researchers measure the temporal precision of associated neural correlates in motor cortex [16]. The UCLA AI copilot study employed such a protocol, having participants attempt to control a cursor or robotic arm while measuring the temporal precision of control signals [16].
Signal-to-Noise Ratio Quantification
SNR measurement protocols vary depending on the specific BCI modality but share common elements. For electrophysiological systems (EEG, ECoG, microelectrode arrays), the standard approach involves:
- Recording data during both "signal" conditions (e.g., during task performance) and "noise" conditions (e.g., during rest or baseline periods)
- Calculating signal power (P_signal) during active periods
- Calculating noise power (P_noise) during quiet periods
- Computing SNR as: SNR = Psignal / Pnoise, often converted to decibels: SNRdB = 10 × log10(Psignal / P_noise) [18]
For systems with periodic stimuli, such as visual evoked potential BCIs, phase-locking measures across multiple trials can separate signal from noise more effectively [17]. Advanced approaches may use the coefficient of variation (ratio of mean to standard deviation, μ/σ) as an alternative SNR definition, particularly when signals have non-zero means [18].
The following diagram illustrates the fundamental signal processing pipeline common to most BCI systems, highlighting where each performance metric is most critically determined:
BCI Signal Processing Pipeline and Quality Metrics
Advanced Signal Processing Techniques
Modern BCI systems employ sophisticated signal processing and machine learning techniques to overcome inherent limitations in raw signal quality. These approaches have proven particularly valuable for enhancing the performance of non-invasive systems where fundamental physical constraints limit spatial resolution and SNR.
The convolutional neural network-Kalman filter (CNN-KF) architecture represents a significant advancement in BCI signal processing [16]. This approach combines the spatial pattern recognition capabilities of convolutional neural networks with the temporal filtering properties of Kalman filters to extract meaningful control signals from noisy neural data. The CNN component identifies spatial patterns in neural activity associated with different movement intentions, while the KF component tracks the temporal evolution of these states, effectively smoothing the output and improving control stability [16].
AI "copilot" systems represent another frontier in BCI signal enhancement. Rather than simply decoding user intent, these systems actively collaborate with users to achieve task goals [16]. For example, when controlling a robotic arm, the AI copilot might interpret high-level intentions from noisy neural signals while handling lower-level details of trajectory planning and obstacle avoidance. In the UCLA study, this approach improved performance by nearly four times for paralyzed participants, enabling tasks that would otherwise be impossible with non-invasive BCIs alone [16].
Surface Laplacian transformation continues to be a valuable technique for improving effective spatial resolution in EEG-based systems [17]. By computing the second spatial derivative of scalp potentials, this method reduces the blurring effect of volume conduction, effectively sharpening the spatial distribution of recorded activity. Studies have demonstrated that SL transformation not only improves spatial resolution but also enhances temporal accuracy by providing a more faithful representation of the timing of underlying neural sources [17].
Research Reagents and Materials
The following table details essential research reagents, materials, and systems used in BCI research and development:
Table 3: Essential Research Materials and Systems for BCI Development
| Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| Electrode Technologies | Wet electrodes (Ag/AgCl), Dry electrodes, Utah Array, Neuralace [3] [13] | Neural signal acquisition | Biocompatibility, impedance, long-term stability [3] |
| Signal Acquisition Systems | Blackrock Neurotech systems, OpenBCI, Medtronic DBS systems [3] [13] [16] | Amplification and digitization of neural signals | Channel count, sampling rate, input-referred noise [3] |
| Implantable Materials | Biocompatible coatings (PEDOT, iridium oxide), Flexible substrates (polyimide) [3] | Chronic implantation interfaces | Tissue response, mechanical compliance, longevity [3] |
| Computational Platforms | NVIDIA AI platforms, Custom decoding hardware [13] [16] | Signal processing and intent decoding | Processing speed, power consumption, algorithm compatibility [16] |
| Calibration Phantoms | Head models with simulated dipoles [17] | System validation and testing | Anatomical accuracy, electrical properties matching tissue [17] |
The benchmarking analysis presented in this guide reveals a consistent trade-off between invasiveness and signal quality across BCI technologies. Invasive systems, particularly intracortical microelectrode arrays, provide superior spatial resolution, temporal resolution, and SNR—enabling complex applications such as speech decoding and dexterous motor control [13]. These performance benefits come at the cost of surgical risk, potential tissue response, and higher regulatory hurdles. Non-invasive systems offer greater accessibility and safety but face fundamental limitations in signal quality due to the biological barriers between neural sources and sensors [17].
The future of BCI technology development appears to be progressing along two parallel tracks: refinement of invasive systems for high-performance medical applications and enhancement of non-invasive systems through advanced signal processing and AI integration [16]. For researchers and clinicians, selection of appropriate BCI technology must consider the specific requirements of the target application, balancing the need for signal quality against practical constraints including safety, accessibility, and regulatory considerations.
As both invasive and non-invasive technologies continue to evolve, the boundaries of what is possible with BCIs will undoubtedly expand. However, the fundamental relationships between spatial resolution, temporal resolution, and SNR will continue to govern system performance, ensuring that these three metrics remain essential for objective comparison and benchmarking of brain-computer interfaces across research, clinical, and consumer domains.
Brain-Computer Interfaces (BCIs) represent a transformative technology that enables direct communication between the brain and external devices, offering significant promise for individuals with motor impairments and advancing human-computer interaction [13]. The core of a BCI system involves acquiring neural signals, processing them to decode the user's intent, and translating that intent into commands for external devices [11]. As the technology evolves, standardized performance metrics are essential for comparing systems, guiding development, and assessing clinical viability. This is particularly critical in the ongoing research comparing invasive BCIs (surgically implanted) with non-invasive BCIs (typically using scalp electrodes) [3] [11].
Invasive BCIs, such as those developed by Neuralink and Blackrock Neurotech, provide high-fidelity signals by placing electrodes directly on or in the brain, but they carry surgical risks and long-term biocompatibility challenges [3] [13]. Non-invasive BCIs, primarily using electroencephalography (EEG), are safer and more accessible but must contend with signal attenuation by the skull, leading to lower resolution and a poorer signal-to-noise ratio (SNR) [16] [11]. This guide introduces and contextualizes the three fundamental KPIs—Accuracy, Information Transfer Rate (ITR), and Latency—for evaluating and comparing BCI performance across these paradigms, providing researchers with a framework for objective benchmarking.
Defining the Core KPIs for BCI Evaluation
Accuracy
Accuracy measures the correctness of the BCI's output relative to the user's intent. It is typically calculated as the percentage of correct classifications or commands over the total number of attempts [9]. For example, in a speller task, if a user intends to select 100 characters and the BCI correctly identifies 95 of them, the accuracy is 95%. High accuracy is paramount for reliable communication and control, especially for assistive technologies used by paralyzed individuals [16] [9]. In discrete tasks, accuracy is a straightforward binomial measure, whereas for continuous control tasks, metrics like correlation coefficients or mean squared error are often more appropriate [9].
Information Transfer Rate (ITR)
Information Transfer Rate (ITR), also known as Bitrate, is a composite metric that balances speed and accuracy, measured in bits per minute (bpm). It quantifies the amount of information communicated per unit time. The standard formula for a system with N possible choices is: ( ITR = (\log2N + P\log2P + (1-P)\log_2\frac{1-P}{N-1}) \times (\frac{60}{T}) ) where P is classification accuracy and T is the time per selection in seconds [9] [21]. A higher ITR indicates a more efficient system. ITR is highly sensitive to both accuracy and the speed of selection, making it a crucial metric for comparing the practical throughput of different BCI systems, from spellers to prosthetic controllers [22] [21].
Latency
Latency refers to the total delay between the user's initiation of a mental command and the system's execution of the corresponding action. This encompasses the time for signal acquisition, processing, decoding, and device response [23]. Low latency is critical for real-time, closed-loop applications like controlling a robotic arm or wheelchair, where delays can disrupt control and user immersion [13] [23]. While often reported as a total system delay, latency can also be broken down into its constituent parts to identify bottlenecks in the BCI pipeline.
Performance Benchmarking: Invasive vs. Non-Invasive BCI Technologies
The table below summarizes performance data from recent studies and commercial systems, highlighting the typical performance ranges and the trade-offs between invasive and non-invasive approaches.
Table 1: Performance Benchmarking of Invasive and Non-Invasive BCI Technologies
| System / Paradigm | Reported Accuracy (%) | Reported ITR (bits/min) | Key Applications | Notes & Context |
|---|---|---|---|---|
| Invasive BCI (General) | ~99% (Speech decoding) [13] | N/A | Communication, prosthetic control | High-fidelity neural signals enable complex decoding with very high accuracy [13]. |
| Non-Invasive: c-VEP Speller | 96.71 [22] | 27.55 [22] | Spelling, communication | Integrated with Mixed Reality (MR); performance on par with traditional screens [22]. |
| Non-Invasive: SSVEP-based BCI | 94.90 [21] | 64.35 [21] | Spelling, device control | A hybrid system combining VEP and pupillary response; uses low-frequency stimuli for comfort [21]. |
| Non-Invasive: AI-Enhanced BCI | Significant improvement factor of 3.9x [16] | N/A | Cursor/robotic arm control | An AI "copilot" dramatically improved task performance for a paralyzed user [16]. |
| Non-Invasive: Motor Imagery (MI) | 86.46 (subject-dependent) [24] | N/A | Neurorehabilitation, control | Deep learning model (EEGEncoder) on a public dataset [24]. |
| Embedded SSVEP System | 93.3 [23] | N/A | Real-time human-computer interaction | Optimized CNN on an FPGA platform; latency of 0.2 ms per trial [23]. |
Analysis of Performance Gaps and Trade-offs
The data illustrates a fundamental trade-off in BCI design. Invasive systems leverage their superior signal-to-noise ratio (SNR) and access to high-frequency neural data to achieve top-tier performance in complex tasks like speech decoding [13]. In contrast, non-invasive systems have made significant strides, particularly with paradigms like SSVEP and c-VEP, which can achieve high ITRs above 60 bpm [21]. The integration of advanced AI and deep learning is a key driver in closing this performance gap, as algorithms become better at decoding noisy EEG signals [16] [25] [24]. Furthermore, hardware advancements, such as deploying models on efficient heterogeneous architectures (e.g., ARM+FPGA), are reducing latency and power consumption, making portable, high-performance non-invasive BCIs more feasible [23].
Experimental Protocols for KPI Measurement
Standardized BCI Evaluation Paradigms
To ensure comparability between studies, researchers employ standardized experimental protocols. Key paradigms include:
- Speller Tasks: Users select characters from a grid. This is common for testing P300, SSVEP, and c-VEP BCIs. Performance is measured by character-level accuracy and typing speed (ITR) [22] [9].
- Motor Imagery (MI) Tasks: Users imagine movements of specific limbs (e.g., left hand vs. right hand). Classification accuracy on datasets like BCI Competition IV-2a is a standard benchmark [24].
- Closed-Loop Control Tasks: Users control a cursor or robotic arm in real-time to reach targets. Metrics include success rate, path efficiency, and completion time, which relate to accuracy and latency [16].
A Representative Workflow: Testing an SSVEP-based BCI
The following diagram outlines a typical experimental workflow for evaluating a hybrid SSVEP-based BCI system, as described in recent literature [21].
Diagram 1: SSVEP-BCI Experimental Workflow
Detailed Methodology for a Hybrid BCI Study [21]:
- Participants: 10 healthy subjects.
- Stimuli: A 12-target speller with flickering stimuli in a low-frequency range (0.8–2.12 Hz) to simultaneously elicit Visual Evoked Potentials (VEP) and Pupillary Response (PR). This range was chosen to improve user comfort compared to traditional alpha-band SSVEP.
- Data Acquisition: EEG signals are recorded using a multi-channel cap. An eye-tracker simultaneously records pupil diameter.
- Signal Pre-processing: EEG data is bandpass filtered. Both EEG and PR signals are transformed into the frequency domain using Fast Fourier Transform (FFT) to identify the stimulus-driven oscillatory responses.
- Feature Extraction & Classification: Features from both VEP and PR are extracted. A decision fusion method combines the information from these two modalities to produce a single classification output (i.e., the target the user is attending to).
- Performance Calculation: Accuracy is calculated as the percentage of correctly identified targets. ITR is then computed using the accuracy and the time taken per selection, including all steps from stimulus onset to command output. User experience is often assessed via questionnaires on visual fatigue and usability [22] [21].
The Scientist's Toolkit: Essential Research Reagents & Materials
The following table details key hardware, software, and methodological components essential for conducting BCI performance evaluations.
Table 2: Essential Research Tools for BCI Performance Evaluation
| Tool / Solution | Function in BCI Research | Specific Examples & Notes |
|---|---|---|
| EEG Amplifier & Cap | Acquires neural signals from the scalp. | Systems from ANT Neuro, EMOTIV, OpenBCI. The number and location of electrodes (e.g., 10-20 system) are critical [3] [11]. |
| Implantable Electrode Arrays | Records high-fidelity neural signals for invasive BCIs. | Utah Array (Blackrock Neurotech), Neuralink's chip, Synchron's Stentrode, Precision's Layer 7 [13]. |
| Visual Stimulation Platform | Presents flickering stimuli to evoke SSVEP/c-VEP. | Can be a standard monitor or an MR headset. Stimulus frequency, intensity, and pattern are key parameters [22] [21]. |
| Eye-Tracking System | Monitors gaze and pupillary dynamics. | Used in hybrid BCIs to measure Pupillary Response (PR) as a complementary input to EEG [21]. |
| Signal Processing Library | Filters, cleans, and pre-processes raw neural data. | Libraries in Python (MNE, SciPy) and MATLAB are standard for handling noise and artifacts [9] [25]. |
| Deep Learning Framework | Builds and trains models for EEG decoding. | TensorFlow, PyTorch. Used to implement architectures like EEGNet, CNN-LSTM, and Transformers [25] [24] [23]. |
| Embedded Computing Platform | Enables portable, real-time BCI operation. | FPGA (e.g., Xilinx ZYNQ) and ARM-based boards are used for hardware acceleration to achieve low-latency processing [23]. |
| Standardized Datasets | Benchmarks and compares new algorithms. | Public datasets like BCI Competition IV-2a for Motor Imagery are vital for reproducible research [24]. |
The rigorous evaluation of BCI systems using Accuracy, ITR, and Latency is fundamental to advancing the field. While invasive BCIs currently hold an advantage in decoding complex intentions with high accuracy, non-invasive systems are rapidly advancing through innovations in hybrid paradigms [21], sophisticated AI-driven decoding [16] [25] [24], and optimized hardware [23]. The choice between invasive and non-invasive approaches involves a complex trade-off between performance, risk, and usability. Future progress will rely on the continued standardization of these KPIs and experimental protocols, enabling clear, objective comparisons that drive the entire field toward more effective and accessible brain-computer interfaces.
From Signals to Solutions: Decoding Methodologies and Real-World Applications in Medical Research
The evolution of Brain-Computer Interfaces (BCIs) hinges on a fundamental trade-off: the balance between performance fidelity and clinical invasiveness. On one end of the spectrum, invasive BCIs, which require neurosurgery, provide high-resolution neural data; on the other, non-invasive BCIs offer greater safety and accessibility but historically suffer from inferior signal-to-noise ratios [16]. The central thesis of modern BCI research is that this performance gap can be bridged not only through hardware improvements but increasingly through advanced decoding algorithms. These algorithms translate raw, often noisy, neural signals into precise control commands, transforming theoretical potential into practical application.
This guide objectively compares the performance of traditional and contemporary neural decoding algorithms, with a specific focus on the emerging hybrid architecture that combines convolutional neural networks with Kalman filters (CNN-KF). We frame this comparison within the broader context of invasive versus non-invasive BCI performance benchmarks, providing researchers with quantitative data and methodological details essential for evaluating the current state of neural decoding.
Fundamental Algorithms in Neural Decoding
Traditional Workhorses: The Kalman Filter
The Kalman Filter (KF) represents a classical approach to decoding continuous movement intentions from neural activity. Introduced in 1960 by Rudolf E. Kálmán, this algorithm is fundamentally a recursive method that estimates unknown variables from a series of noisy measurements over time [16]. In BCI applications, its primary function is to filter out noise in order to find the meaningful signal, effectively estimating the user's intended movement trajectory (e.g., cursor or robotic arm velocity) from noisy neural data.
The strength of the Kalman Filter lies in its recursive predictive capability. It operates in a two-step process: a prediction step, where it forecasts the next state and its uncertainty, and an update step, where it incorporates the new measurement to refine its estimate. This makes it particularly well-suited for time-series analysis in BCIs, where it can decode movement parameters like hand velocity from motor cortical neurons [26]. However, as a primarily linear decoder, its performance is limited when faced with the non-stationary and non-linear characteristics of neural signals, especially from non-invasive sources like EEG.
Modern Powerhouses: Deep Learning and Convolutional Neural Networks
Deep Learning architectures, particularly Convolutional Neural Networks (CNNs or ConvNets), represent a paradigm shift in neural decoding capability. Unlike traditional linear methods, CNNs are deep learning algorithms designed to automatically and adaptively learn spatial hierarchies of features from data [16]. Inspired by the human visual system, these networks learn features directly from the data in order to make predictions, eliminating the need for manual feature engineering.
In BCI applications, CNNs excel at identifying complex, non-linear patterns in neural data that are imperceptible to traditional algorithms. For EEG-based BCIs, specialized architectures like EEGNet have been developed to decode neural signals with remarkable accuracy [26]. The primary advantage of CNNs is their ability to model the complex, non-linear relationships between neural activity and behavior. However, they face challenges in closed-loop BCI settings, particularly regarding adaptation to neural non-stationarities, as noisy gradient descent steps during online operation are not guaranteed to improve decoder performance [26].
The Hybrid Approach: CNN-KF Architecture
The CNN-KF (convolutional neural network-Kalman filter) architecture represents a sophisticated hybrid approach that synergistically combines the strengths of both component algorithms. In this configuration, a CNN serves as a non-linear feature extractor, processing raw neural data to identify complex patterns. The outputs of this CNN then become the observation inputs for a Kalman Filter, which performs the temporal filtering and state estimation necessary for smooth, continuous control [16] [26].
This architecture is guided by two complementary principles. First, it leverages the empirically demonstrated superior performance of deep learning architectures in both offline and closed-loop experiments compared to traditional linear classifiers [26]. Second, it addresses the critical need for adaptation in response to EEG non-stationarities, which are an inherent challenge in practical BCI operation. By freezing the CNN parameters and only adapting the linear KF component during closed-loop operation, the CNN-KF achieves both non-linear decoding capability and stable real-time adaptation [26]. This approach has demonstrated particular efficacy in non-invasive BCI systems, enabling continuous control of computer cursors and robotic arms through decoded electroencephalography (EEG) signals.
Table 1: Core Algorithm Comparison in Neural Decoding
| Algorithm | Core Mechanism | Primary Strengths | Primary Limitations | Best Suited BCI Type |
|---|---|---|---|---|
| Kalman Filter (KF) | Recursive state estimation from noisy time-series data | Effective noise filtering; stable continuous trajectory decoding | Primarily linear; limited non-linear pattern recognition | Invasive & Non-invasive (simpler tasks) |
| Convolutional Neural Network (CNN) | Hierarchical non-linear feature learning from raw data | Powerful pattern recognition; no manual feature engineering needed | Online adaptation challenges; computationally intensive | Non-invasive (complex pattern decoding) |
| CNN-KF Hybrid | CNN features feed into KF for state estimation | Combines non-linear decoding with stable online adaptation | Increased architectural complexity | Non-invasive (continuous control tasks) |
Performance Benchmarks: Quantitative Comparisons
Performance in Non-Invasive BCI Systems
The integration of AI copilots with advanced decoding algorithms has yielded dramatic performance improvements in non-invasive BCIs, particularly for users with paralysis. In a landmark 2025 study, researchers implemented a CNN-KF decoder with an AI copilot in a non-invasive BCI system using a 64-channel EEG cap [16] [26]. The system was tested on three healthy participants and one participant with T5 complete paraplegia.
The results demonstrated that the AI copilot solution improved performance by a factor of 3.9 times for the paralyzed participant in both cursor control and robotic arm tasks [16]. Critically, researchers reported that the paralyzed participant would not have been able to perform these tasks without the AI copilot assistance [16]. In the cursor control task (a center-out task with eight targets), all participants achieved control across multiple sessions, with healthy participants maintaining a median success rate of 100%, while the participant with paralysis achieved a median success rate of 88% [26].
For communication applications, recent advances in speech decoding have been equally impressive. A streaming brain-to-voice neuroprosthesis developed in 2025 demonstrated the ability to decode a 1,000+ word vocabulary at 47.5 words per minute with a greater than 99% success rate, translating brain activity into audible speech in less than 80 milliseconds [27]. This represents a significant leap from earlier devices that decoded approximately 15 words per minute.
Performance in Invasive BCI Systems
Invasive BCIs continue to set the benchmark for high-performance neural decoding, particularly for motor control and communication applications. Companies like Neuralink, Blackrock Neurotech, Paradromics, and Precision Neuroscience are advancing invasive technologies with increasingly sophisticated electrode arrays and decoding approaches [13].
While specific performance metrics for these commercial systems in human trials are often preliminary, the fundamental advantage of invasive approaches lies in their signal quality. Invasive electrodes positioned directly on or in the brain provide significantly higher spatial resolution and signal-to-noise ratio compared to non-invasive methods [3]. This enables more detailed neural feature extraction, which theoretically supports higher-dimensional control and faster information transfer rates.
The Connexus BCI from Paradromics, for instance, utilizes a modular array with 421 electrodes and an integrated wireless transmitter, representing the high channel counts possible with invasive approaches [13]. Similarly, Precision Neuroscience's Layer 7 device, an ultra-thin electrode array designed to conform to the cortical surface, has received FDA clearance for commercial use with implantation durations of up to 30 days [13]. These technological advances in electrode design are complemented by increasingly sophisticated decoding algorithms that leverage the rich data streams these devices provide.
Table 2: Experimental Performance Metrics Across BCI Modalities
| BCI Type | Decoding Algorithm | Task | Performance Metric | Result | Participant Type |
|---|---|---|---|---|---|
| Non-invasive (EEG) | CNN-KF + AI Copilot | Cursor Control | Performance Improvement | 3.9x increase | Paraplegic (T5 complete) |
| Non-invasive (EEG) | CNN-KF | Center-Out 8 Task | Success Rate | 88% median | Paraplegic (T5 complete) |
| Non-invasive (EEG) | CNN-KF | Center-Out 8 Task | Success Rate | 100% median | Healthy (3 participants) |
| Invasive (ECoG) | Deep Learning | Speech Decoding | Words Per Minute | 47.5 WPM (1000+ vocab) | Paralysis from stroke |
| Invasive (ECoG) | Deep Learning | Speech Decoding | Success Rate | >99% | Paralysis from stroke |
| Visual BCI | Broadband White Noise | Information Transfer | Bit Rate | 50 bps (record) | N/A |
Experimental Protocols and Methodologies
CNN-KF Decoder Training and Validation
The development of the hybrid CNN-KF decoder follows a rigorous, multi-stage training protocol designed to ensure robust performance while avoiding decoding of confounding signals like eye movements [26].
Open-Loop Training Phase: Participants are first prompted with randomly chosen actions from four movement classes (e.g., left hand, right hand, both hands, feet for healthy participants; left leg, right leg, both legs, still for participant with paralysis). An initial "seed decoder" is trained from these data, but this is not the final decoder, as CNNs may initially decode features related to eye movement rather than sensorimotor activity [26].
Decorrelated Closed-Loop Training: To ensure performance derives from sensorimotor decoding rather than eye movements, researchers implement a critical second training task where presented targets and kinematics are decoupled from motor intent. In this phase, participants are prompted with a random motor intent (e.g., left leg) while being shown a random target position (e.g., up right). The CNN classifier is specifically trained to decode the motor intent despite the conflicting visual cue, confirming through subsequent analysis that the system decodes sensorimotor activity rather than eye movement artifacts [26].
Closed-Loop Evaluation: Performance is evaluated using a standard center-out task with eight targets, a common intracortical BCI cursor control benchmark. Participants must hold the cursor over the target for 500ms to successfully acquire it. To systematically challenge the decoder, target sizes are progressively shrunk (from 7cm diameter down to 2.9cm) as participant performance improves, with successful performance defined as acquiring targets at a rate higher than ten targets per minute with over 90% success rate [26].
Performance Measurement Standards in BCI Research
Standardized performance measurement is crucial for cross-study comparisons and field advancement. The BCI research community has developed specific guidelines for performance reporting through workshops like the 2013 International BCI Meeting at Asilomar Conference Center [9].
For discrete BCIs (e.g., P300 spellers), key metrics include:
- Accuracy: The percentage of correct classifications, with reporting of both theoretical chance level and empirical chance performance calculated through label permutation tests [9].
- Confidence Intervals: Particularly important for accuracy and correlation coefficients, acknowledging that any performance metric calculated on finite data represents one observation of a random variable [9].
- Information Transfer Rate (ITR): A speed-accuracy trade-off metric that accounts for both the rate of selection and accuracy, though calculation methods must be standardized for proper comparison [9].
For continuous BCIs (e.g., cursor control), essential metrics include:
- Success Rate: The percentage of successfully acquired targets in tasks like the center-out task [26].
- Trial Time: The average time to acquire each target, which naturally increases as target difficulty increases [26].
- Path Efficiency: The optimality of the movement trajectory compared to the most direct path [9].
General methodological reporting must include detailed equipment specifications, electrode number and location, participant demographics, experimental protocol timing, and the quantity of data used for both training and testing [9].
Visualization: Algorithmic Architectures and Workflows
CNN-KF Hybrid Decoder Architecture
BCI Performance Benchmarking Workflow
The Researcher's Toolkit: Essential Materials and Reagents
Table 3: Essential Research Reagents and Materials for BCI Decoding Research
| Item | Function/Purpose | Example Specifications |
|---|---|---|
| EEG Acquisition System | Records electrical brain activity from scalp | 64-channel cap; compatible with dry/wet electrodes |
| Electrode Types | Interface for signal acquisition; choice affects signal quality | Wet electrodes (gel); Dry electrodes; Multi-electrode arrays (invasive) |
| Signal Processing Software | Preprocessing, feature extraction, and decoding implementation | MATLAB, Python (MNE, PyTorch, TensorFlow) |
| Deep Learning Framework | Implementation of CNN and other neural network architectures | PyTorch, TensorFlow with GPU acceleration support |
| Kalman Filter Library | Implementation of state estimation algorithms | Custom implementations; Python (SciPy, NumPy) |
| Closed-Loop BCI Platform | Real-time signal processing and experimental control | BCI2000, OpenViBE, Lab Streaming Layer (LSL) |
| Validation Datasets | Benchmarking algorithm performance | Public BCI competition datasets; Laboratory-recorded data |
| Performance Metrics Package | Standardized calculation of BCI performance metrics | Custom scripts implementing ITR, accuracy, path efficiency |
The evolution from traditional algorithms like the Kalman Filter to modern deep learning approaches and their hybrid combinations represents a fundamental shift in BCI research strategy. Rather than treating signal acquisition and decoding as separate challenges, the field is increasingly adopting integrated approaches where algorithms are specifically designed to compensate for the limitations of particular recording modalities.
The CNN-KF architecture exemplifies this trend, demonstrating that hybrid approaches can extract significantly greater utility from existing recording technologies, particularly non-invasive systems. The addition of AI copilots utilizing shared autonomy further enhances this approach, leveraging task structure and contextual information to dramatically improve performance [16] [26]. As these algorithmic strategies mature, they narrow the performance gap between invasive and non-invasive BCIs, potentially expanding the applications of non-invasive systems while pushing invasive systems toward increasingly complex control capabilities.
Future progress will likely involve even tighter integration of multiple algorithmic approaches, increased personalization of decoders to individual users, and the development of standardized benchmarking frameworks that enable direct comparison across studies and laboratories. As decoding algorithms continue to advance, they will play an increasingly pivotal role in translating BCI technology from laboratory demonstrations to clinically viable and commercially successful applications.
Brain-Computer Interfaces (BCIs) represent a transformative technology for restoring motor function to individuals with paralysis, spinal cord injuries, and stroke. The field is fundamentally divided between invasive approaches, which involve surgical implantation of electrodes directly into or onto the brain tissue, and non-invasive approaches, which measure brain signals from the scalp. The core thesis of contemporary BCI research posits that this choice entails a fundamental trade-off: invasive interfaces provide superior signal quality and control fidelity at the cost of surgical risk, while non-invasive systems offer greater safety and accessibility but historically lag in performance [3] [2]. This guide provides a performance benchmark of current BCI technologies across three key applications for motor restoration: robotic arm control, exoskeleton operation, and computer cursor manipulation. By synthesizing the latest experimental data and methodologies, we offer researchers and drug development professionals a quantitative foundation for evaluating these rapidly evolving neurotechnologies.
Performance Benchmarking: Invasive vs. Non-Invasive BCIs
The following tables consolidate key performance metrics from recent studies and commercial systems, providing a direct comparison of the capabilities of invasive and non-invasive BCIs.
Table 1: Overall System Performance Benchmarks for Motor Function Applications
| Metric | Invasive BCI (Paradromics Connexus) | Non-Invasive BCI (EEG-based) | Semi-Invasive BCI (Synchron Stentrode) |
|---|---|---|---|
| Information Transfer Rate (ITR) | >200 bps (max), >100 bps (with 11ms latency) [28] | Not specified | "Orders of magnitude" lower than invasive [28] |
| Total System Latency | 56ms (for >200 bps), 11ms (for >100 bps) [28] | Not specified | Not specified |
| Typing Speed (Equivalent) | Exceeds transcribed human speech (~40 bps) [28] | Not specified | Lower than intracortical systems [28] |
| Key Applications Demonstrated | High-bandwidth communication, complex device control [28] | Robotic hand control (individual fingers), cursor control [29] [16] | Computer cursor control, text communication [2] [30] |
Table 2: Application-Specific Performance in Motor Function Restoration
| Application | BCI Type & Study | Performance Metrics | Subject Cohort |
|---|---|---|---|
| Robotic Hand (Individual Finger Control) | Non-invasive (EEG), [29] | Real-time decoding accuracy: 80.56% (2-finger task), 60.61% (3-finger task) [29] | 21 able-bodied, experienced BCI users [29] |
| Computer Cursor & Robotic Arm Control | Non-invasive (EEG) with AI Copilot, [16] | Performance improvement factor: 3.9x; Paralyzed participant could not complete tasks without AI aid [16] | 3 healthy participants, 1 participant with T5 spinal cord injury [16] |
| Forearm Exoskeleton for Stroke | Non-invasive (Low-cost EEG), [31] | Classification accuracy: >92% for SSVEP, 100% for two-state Alpha Suppression recognition [31] | Designed for subacute and chronic stroke patients [31] |
| Communication & Text Generation | Invasive (Blackrock Neurotech), [30] | Typing speed: 90 characters per minute [30] | Patients with paralysis, ALS, spinal cord injuries [30] |
Experimental Protocols and Methodologies
Non-Invasive EEG for Individual Finger Control
A landmark 2025 study published in Nature Communications demonstrated real-time, non-invasive robotic hand control at the individual finger level, a significant advance in dexterity [29].
Core Protocol:
- Task Paradigm: Participants performed both Movement Execution (ME) and Motor Imagery (MI) of individual fingers (thumb, index, pinky) on their dominant hand.
- Signal Acquisition: Brain activity was recorded using scalp Electroencephalography (EEG).
- Neural Decoding: A deep neural network architecture, EEGNet-8.2, was used for real-time decoding of the neural signals associated with each finger movement or intention [29].
- Model Adaptation: A fine-tuning mechanism was implemented to adapt the base decoding model to individual participants using same-day data, countering inter-session variability.
- Feedback & Control: Decoded outputs were converted into control commands for a robotic hand, providing participants with both visual (on-screen) and physical (robotic finger movement) feedback.
The workflow for this sophisticated decoding process is illustrated below.
AI Copilot Enhancement for Non-Invasive BCI
A study from UCLA showcased a paradigm for overcoming the signal-to-noise limitations of non-invasive BCIs by integrating an artificial intelligence (AI) copilot [16].
Core Protocol:
- Signal Acquisition: EEG signals were recorded from a 64-channel cap on three healthy participants and one paraplegic participant.
- Decoder Architecture: The core decoder used a Convolutional Neural Network-Kalman filter (CNN-KF). The CNN extracted spatial and temporal features from the neural data, while the Kalman filter provided robust state estimation from the noisy time-series data.
- AI Copilot Integration: The system incorporated a separate "AI copilot" that leveraged task structure and environmental context (e.g., location of potential targets) to change the distribution of actions.
- Shared Autonomy: This created a shared autonomy system where the user's decoded neural commands were intelligently supplemented and refined by the AI to achieve the task goal more efficiently and reliably.
The integration of the AI copilot within the BCI control loop is detailed below.
The Scientist's Toolkit: Key Research Reagents & Materials
For researchers aiming to replicate or build upon the cited studies, the following table details essential components and their functions in modern BCI systems for motor restoration.
Table 3: Essential Research Materials for BCI Motor Control Studies
| Item / Technology | Function in Experimental Protocol | Example Use Case |
|---|---|---|
| High-Density EEG Systems | Records electrical brain activity from the scalp using multiple electrodes (e.g., 64-channel). Foundation for non-invasive signal acquisition. [29] [16] | Motor imagery decoding, evoked potential studies. |
| Deep Learning Decoders (e.g., EEGNet) | Specialized neural networks for classifying EEG signals. Enable end-to-end learning from raw data, improving accuracy for complex tasks like finger control. [29] | Real-time decoding of individual finger movements. |
| AI Copilot / Shared Autonomy Algorithms | Software modules that interpret user intent and integrate environmental context to refine BCI commands. Crucial for compensating for low SNR in non-invasive BCIs. [16] | Assisting paralyzed users in controlling a cursor or robotic arm. |
| Kalman Filter | A recursive algorithm that optimally estimates the state of a dynamic system from a series of noisy measurements. Used for smoothing and predicting kinematic parameters. [16] | Trajectory decoding for cursor control or robotic arm movement. |
| Motorized Robotic Hand/Exoskeleton | A physical actuation device that provides functional output for decoded brain signals. Offers tangible feedback and enables Activities of Daily Living (ADL) tasks. [29] [31] | Restoring grasping and manipulation capabilities. |
| Utah Array / Microelectrode Array | Invasive cortical implant providing high-resolution neural signals by recording from dozens to hundreds of individual neurons. [3] [2] | High-performance communication and complex robotic control. |
| Stentrode (Endovascular Electrode) | A minimally invasive electrode array delivered via blood vessels to rest on the surface of the brain. Offers a middle ground between surface EEG and cortical implants. [2] [30] | Thought-based control of digital devices without open-brain surgery. |
The empirical data and methodologies presented in this guide underscore a clear, yet evolving, performance hierarchy. Invasive BCIs, as demonstrated by Paradromics and Blackrock Neurotech, currently set the benchmark for speed and fidelity, achieving information transfer rates that support fluent communication and complex control [28] [30]. Conversely, non-invasive approaches have made remarkable strides in dexterous applications, such as individual finger control, and are being rapidly augmented by AI copilots that effectively close the performance gap for specific tasks [29] [16]. The choice between invasive and non-invasive BCI pathways is no longer purely a trade-off between performance and safety. It is increasingly a question of aligning technology with clinical need: invasive interfaces for maximal restoration in severe paralysis, and AI-enhanced non-invasive systems for broader applications in rehabilitation and assistive technology. For the research community, the critical path forward involves standardizing benchmarking protocols like SONIC [28], validating these technologies in larger clinical cohorts, and continuing to innovate in adaptive algorithms that bridge the neural signal gap.
Brain-Computer Interface (BCI) technology represents a revolutionary approach to restoring communication for individuals with severe speech and motor impairments. By establishing a direct communication pathway between the brain and external devices, BCIs can translate neural activity into executable commands for speech synthesis or text output. This technology has particular significance for conditions such as amyotrophic lateral sclerosis (ALS), brainstem stroke, spinal cord injuries, and other neurological disorders that result in anarthria—the loss of the ability to speak.
The field has diverged into two primary technological pathways: invasive approaches that require surgical implantation of neural recording devices, and non-invasive methods that measure brain activity from outside the skull. As research advances, understanding the performance characteristics, limitations, and appropriate applications of each approach becomes critical for researchers, clinicians, and developers working in neurotechnology and assistive communication. This comparison guide examines the current state of both invasive and non-invasive BCIs for speech restoration, providing experimental data and methodological insights to inform research and development decisions.
Performance Benchmarks: Quantitative Comparison
Direct comparison of invasive and non-invasive BCIs reveals significant differences in their performance characteristics and technical capabilities. The table below summarizes key performance metrics based on current research findings:
Table 1: Performance Comparison of Invasive vs. Non-Invasive Speech BCIs
| Performance Metric | Invasive BCI | Non-Invasive BCI |
|---|---|---|
| Word Decoding Accuracy | High accuracy demonstrated (e.g., phoneme-based decoding for speech restoration) [32] | Up to 37% top-10 accuracy with 250-word retrieval set [33] |
| Temporal Resolution | ~1 ms resolution for neuronal spikes [34] | Limited by EEG/MEG systems, but sufficient for word-level decoding [33] |
| Spatial Resolution | Single neuron level with microelectrodes [34] [35] | Limited by skull/scalp, source localization challenges [11] |
| Signal-to-Noise Ratio | High SNR, robust against electrical noise [34] | Lower SNR, susceptible to artifacts and noise [16] [11] |
| Information Transfer Rate | Record-breaking speeds reported [35] | Significantly lower, improved with AI copilots (3.9x improvement shown) [16] |
| Clinical Availability | Early clinical trials (e.g., Paradromics, Neuralink) [35] | Commercially available systems (e.g., Emotiv, NeuroSky) [36] |
| Key Applications | Restoring fluent, rapid communication [32] [35] | Basic communication, cognitive research, assistive technology [37] [36] |
Market analysis indicates distinct growth trajectories for each approach. The overall BCI market is forecast to grow to over $1.6 billion by 2045, with non-invasive BCI growth rate for 2025-2033 estimated at 9.35% CAGR, compared to 1.49% CAGR for invasive BCIs [3] [16].
Invasive BCI Approaches: Methods and Protocols
Experimental Framework for Intracortical Speech Decoding
Invasive BCI approaches for speech restoration typically involve surgical implantation of microelectrode arrays directly into cortical regions responsible for speech production and processing. The following methodology is based on recent pioneering work:
Neural Signal Acquisition: Microelectrode arrays (smaller than a pea) are surgically implanted in the speech motor cortex. These arrays typically contain multiple electrodes that record neural activity at the level of individual neurons or small neuronal populations. The arrays are connected via a cable to a wireless transceiver implanted in the chest [32] [35].
Participant Training and Data Collection: Participants imagine speaking sentences presented to them while neural patterns are recorded. The training involves repeated trials where patients attempt to speak or imagine speaking words and sentences, allowing the system to learn the correspondence between neural patterns and speech sounds [32].
Decoding Algorithm Training: Machine learning algorithms, particularly deep learning models, are trained to recognize repeatable patterns of neural activity associated with phonemes—the smallest units of speech. The system learns to stitch these phonemes into complete sentences [32].
Output Generation: Decoded neural patterns are converted into either text display on a screen or real-time voice output using pre-recorded samples of the participant's own voice when available [35].
Table 2: Key Research Reagents and Materials for Invasive BCI Research
| Item | Function | Example Specifications |
|---|---|---|
| Microelectrode Arrays | Record neural signals from individual neurons | Utah Array; Paradromics array (7.5mm diameter, platinum-iridium electrodes) [34] [35] |
| Neural Signal Processors | Amplify, filter, and digitize neural signals | Miniaturized electronics with wireless transmission capabilities [35] |
| Deep Learning Algorithms | Decode neural patterns into speech elements | Phoneme recognition models, sequence-to-sequence models [32] |
| Speech Synthesizers | Convert decoded signals to audible speech | Formant synthesizers, voice banking systems [32] [38] |
Recent Advances: Inner Speech Decoding
A significant recent advancement in invasive BCI has been the decoding of "inner speech" or "inner monologue"—the imagination of speech in one's mind without any physical attempt to speak. Researchers at Stanford Medicine have demonstrated that inner speech evokes clear and robust patterns of activity in motor regions of the brain, though these patterns are smaller than those evoked by attempted speech [32].
This research has also addressed critical privacy concerns through the development of security measures. For BCIs designed to decode attempted speech, training methods can help the system ignore inner speech. For next-generation systems intended to decode inner speech directly, password-protection systems can prevent accidental decoding—for example, requiring users to imagine a rare phrase such as "as above, so below" before activation [32].
Invasive BCI Signal Pathway
Non-Invasive BCI Approaches: Methods and Protocols
Experimental Framework for EEG/MEG Speech Decoding
Non-invasive BCI approaches for speech restoration utilize external sensors to measure brain activity without surgical implantation. The following methodology is based on large-scale studies involving hundreds of participants:
Signal Acquisition Systems: Research-grade EEG systems with 64 or more electrodes positioned according to the international 10-10 system. Alternatively, Magnetoencephalography (MEG) systems provide higher signal-to-noise ratios but require specialized shielded environments [33] [38].
Experimental Paradigms: Participants either read or listen to words and sentences while neural activity is recorded. Reading paradigms often use Rapid Serial Visual Presentation (RSVP), where words are displayed one at a time at a fixed rate. For listening paradigms, participants hear spoken words or sentences [33].
Signal Preprocessing: Raw EEG/MEG signals are processed to remove artifacts including eye movements, muscle activity, and environmental noise. Advanced filtering techniques and independent component analysis are typically employed [37] [11].
Deep Learning Decoding Pipeline: State-of-the-art approaches use transformer-based architectures with contrastive learning objectives. The model is trained to decode individual words from EEG/MEG recordings by learning the alignment between neural signals and word representations [33].
Performance Validation: Decoding accuracy is evaluated using top-k accuracy metrics (e.g., top-10 accuracy), measuring whether the correct word appears among the model's top k predictions. Cross-validation across participants and datasets ensures robustness [33].
Scaling Laws and Performance Optimization
Recent research with 723 participants across nine datasets has revealed important scaling properties for non-invasive speech decoding:
- Training Data Volume: Decoding performance increases with the amount of training data, following a roughly log-linear trend without clear signs of diminishing returns [33].
- Test-Time Averaging: Accuracy substantially improves with averaging of multiple decoding predictions for the same word, with some datasets showing two-fold improvement after averaging just 8 predictions [33].
- Modality Differences: Reading paradigms yield better decoding performance than listening paradigms, and MEG generally outperforms EEG due to higher signal-to-noise ratios [33].
AI-Enhanced Performance Optimization
A significant breakthrough in non-invasive BCI performance has come from the integration of artificial intelligence copilots. Researchers at UCLA demonstrated that shared autonomy systems, where AI collaborates with BCI users, can improve performance by a factor of 3.9 times for paralyzed participants in cursor control and robotic arm tasks [16].
The AI copilot approach uses a convolutional neural network-Kalman filter (CNN-KF) architecture that takes advantage of task structure by updating decoder parameters in closed loop and changing the distribution of actions based on observations of the environment [16].
Non-Invasive BCI Signal Pathway
Comparative Analysis and Research Implications
The performance disparity between invasive and non-invasive approaches reflects fundamental differences in their underlying technology. Invasive BCIs provide direct access to neural signals with high spatial and temporal resolution, enabling more accurate and rapid speech decoding. However, this comes with significant clinical risks, surgical requirements, and higher costs [34] [35].
Non-invasive BCIs offer greater accessibility and safety, with commercially available systems that can be deployed outside laboratory settings. While their performance currently lags behind invasive approaches, advances in AI enhancement and large-scale deep learning models are rapidly closing this gap [33] [16].
For research applications, the choice between invasive and non-invasive approaches depends on multiple factors including target population, research timeline, and available resources. Invasive approaches are particularly suited for clinical populations with severe communication impairments where the risks of surgery are justified by the potential benefits. Non-invasive approaches offer broader research applicability, including basic neuroscience studies, protocol development, and assistive technology with lower regulatory barriers [3] [37].
Table 3: Research Applications and Technology Selection Guidelines
| Research Objective | Recommended Approach | Rationale | Key Considerations |
|---|---|---|---|
| Basic Speech Neuroscience | Non-invasive (EEG/MEG) | Sufficient resolution for many research questions without ethical concerns of implantation [33] [11] | Higher participant numbers, lower regulatory barriers |
| Clinical Speech Restoration | Invasive (intracortical) | Superior performance for patients with complete communication loss [32] [35] | Surgical risks justified by quality of life improvement |
| Assistive Technology Development | Non-invasive | Faster iteration cycles, easier participant recruitment [37] [36] | Balance between performance and accessibility |
| Long-Term Usage Studies | Both approaches needed | Invasive for permanent implantation, non-invasive for intermittent use [3] [11] | Device longevity, signal stability over time |
Future research directions include the development of fully implantable wireless systems for invasive BCIs, with several companies expecting such hardware to become available within the next few years [32]. For non-invasive systems, exploration of brain regions outside the motor cortex may yield higher-fidelity information for speech decoding [32]. Hybrid approaches that combine the strengths of both methodologies may ultimately provide the optimal balance of performance and accessibility for synthetic speech restoration.
The Role of AI Copilots and Shared Autonomy in Enhancing BCI Performance and Usability
Brain-Computer Interfaces (BCIs) represent a groundbreaking technology that enables direct communication between the brain and external devices, offering particular promise for restoring function to individuals with paralysis or neurological disorders [39]. A fundamental challenge in BCI development has been the performance trade-off between invasive systems, which offer high-quality neural signals but require neurosurgery, and non-invasive systems, which are safer but historically demonstrate inferior signal-to-noise ratios and lower information transfer rates [40] [16]. The emergence of artificial intelligence (AI) copilots and shared autonomy architectures now provides a transformative approach to bridging this performance gap. These systems complement traditional neural decoding by integrating contextual awareness and task structure, thereby significantly enhancing BCI performance and usability for both invasive and non-invasive platforms [41] [26].
This review objectively compares the performance of AI-enhanced BCI systems against traditional alternatives, framed within the broader research context of invasive versus non-invasive BCI performance benchmarks. We synthesize recent experimental data, detail methodological protocols, and analyze how shared autonomy is redefining the potential of neurotechnology.
Performance Benchmarking: Quantitative Comparisons
Information Transfer Rates and Latency
Information Transfer Rate (ITR), measured in bits per second (bps), and latency (delay in milliseconds) are critical metrics for evaluating BCI performance. Higher ITR and lower latency enable more fluid and complex control. The following table summarizes the performance of various BCI systems, demonstrating the significant advantage of invasive platforms and the performance envelope required for different applications.
Table 1: BCI Performance Benchmarking Based on Information Transfer Rate and Latency
| BCI System / Type | Information Transfer Rate (ITR) | Latency | Key Applications / Context |
|---|---|---|---|
| Paradromics Connexus BCI (Invasive) [28] | >200 bps (High-Accuracy Mode)>100 bps (Low-Latency Mode) | 56 ms (High-Accuracy)11 ms (Low-Latency) | Preclinical data from sheep auditory cortex. ITR exceeds transcribed human speech (~40 bps). |
| Traditional Intracortical BCIs (e.g., Utah Array, early Neuralink trials) [28] | ~10 bps | Not Specified | Established benchmark for invasive research and clinical trials. |
| Endovascular BCIs (e.g., Synchron Stentrode) [28] | ~1-2 bps | Not Specified | Minimally invasive approach; lower bandwidth. |
| AI-Enhanced Non-Invasive BCI (UCLA EEG-based) [41] [26] | Performance measured by task success rate | Not Specified | Target hit rate in cursor control improved by a factor of 3.9. |
Functional Task Performance
Beyond raw data transfer, functional performance in specific tasks is a crucial measure of real-world utility. The integration of AI copilots has shown dramatic improvements, particularly for non-invasive systems and users with paralysis.
Table 2: Functional Task Performance of BCI Systems With and Without AI Copilots
| BCI System / Type | Task | Performance Without AI Copilot | Performance With AI Copilot |
|---|---|---|---|
| Non-Invasive BCI (UCLA) [41] [26] | Cursor Control (8-target center-out task) | Baseline performance (Reference) | 3.9x higher target hit rate for a participant with paralysis. |
| Non-Invasive BCI (UCLA) [41] | Robotic Arm Pick-and-Place | Participant with paralysis unable to complete the task. | Participant completed the task in ~6.5 minutes. |
| Invasive BCIs (General) [34] | Control of robotic arms, computer cursors | Enables basic real-time control in 3D space and gripping force. | Further enhancements possible through shared autonomy, though data from specific copilot implementations is still emerging. |
Experimental Protocols and Methodologies
The UCLA AI-BCI Protocol for Non-Invasive Systems
A key study from UCLA provides a replicable experimental protocol for integrating AI copilots with a non-invasive BCI. The system uses electroencephalography (EEG) and a specific AI architecture to achieve shared autonomy [41] [26].
- Signal Acquisition: A 64-channel EEG cap is used to record brain signals from participants. The study involved three healthy participants and one with T5 complete paraplegia [26] [16].
- Decoder Architecture (CNN-KF): The core of the system is a hybrid decoder combining a Convolutional Neural Network (CNN) and a Kalman Filter (KF). The CNN, adapted from the EEGNet architecture, extracts non-linear features from the EEG data. These features are then fed into a Kalman Filter, which decodes the user's intended movement kinematics (e.g., cursor velocity). The KF parameters are adaptively updated in real-time using Closed-Loop Decoder Adaptation (CLDA) to compensate for the non-stationary nature of EEG signals [26].
- AI Copilot Implementation: Two distinct AI copilots were designed:
- Cursor Control Copilot: Trained using deep reinforcement learning, this copilot assists in moving the cursor toward inferred targets on the screen by leveraging task structure (e.g., known target locations) [26].
- Robotic Arm Copilot: This copilot uses a built-in camera (computer vision) to automatically identify objects and assist in the "pick-and-place" task by refining the user's commands to facilitate grasping and placement [41] [26].
- Task Paradigm: Performance was evaluated using a standard center-out task (for cursor control) and a sequential block moving task (for the robotic arm). The AI copilot's assistance was quantified by comparing task completion times and success rates with and without its intervention [41] [26].
The following diagram illustrates the workflow and logical structure of this AI-BCI system.
The SONIC Benchmark for Invasive BCI Performance
Paradromics has introduced the Standard for Optimizing Neural Interface Capacity (SONIC) as an application-agnostic benchmarking paradigm for invasive BCIs. This protocol provides a standardized way to measure and compare the core engineering performance of BCI systems [28].
- Objective: To measure the fundamental information transfer capacity of a BCI system, independent of any specific end-application (e.g., typing or limb control).
- Preclinical Model: Experiments are conducted using animal models (e.g., sheep).
- Stimulus Presentation: Controlled sequences of sounds (e.g., five-tone sequences mapped to characters) are presented to the subject.
- Neural Recording: The implanted BCI (e.g., Paradromics Connexus) records neural activity from the relevant cortical area (e.g., auditory cortex).
- Decoding and Analysis: The system attempts to predict which sounds were presented based solely on the recorded neural data. The mutual information between the presented and predicted sounds is calculated, yielding a robust and comparable measure of the system's ITR in bits per second (bps) [28].
This benchmark highlights the raw performance capabilities of next-generation invasive BCIs, setting a new standard against which other systems, including those enhanced by AI, can be evaluated.
The Scientist's Toolkit: Key Research Reagents and Materials
Successful BCI research, particularly in developing AI-enhanced systems, relies on a suite of specialized hardware and software tools.
Table 3: Essential Research Reagents and Materials for AI-Enhanced BCI Development
| Item / Solution | Function / Application | Example Use Case |
|---|---|---|
| High-Density EEG Systems (e.g., 64-channel caps) [26] | Non-invasive acquisition of brain electrical activity with high temporal resolution. | Primary signal source for non-invasive BCI studies, as used in the UCLA AI-BCI experiments. |
| Implantable Electrode Arrays (e.g., Utah Array, Paradromics Connexus, Neuralink) [3] [28] [13] | Invasive recording of neural signals (spikes, LFPs) with high spatial resolution and signal-to-noise ratio. | Used in high-performance invasive BCI research for applications like robotic arm control and speech decoding. |
| Convolutional Neural Network (CNN) Architectures (e.g., EEGNet) [26] | Deep learning models for extracting complex, non-linear features from raw neural data. | Employed in hybrid decoders to improve the accuracy of intent decoding from noisy EEG signals. |
| Kalman Filter & Adaptive Algorithms [26] | Statistical filtering for estimating system state from noisy data and adapting decoder parameters in real-time. | Critical for Closed-Loop Decoder Adaptation (CLDA) to maintain stable performance over time. |
| Computer Vision (CV) Platforms [41] [26] | Provides real-time environmental context (e.g., object location and identity) to the AI copilot. | Enables the robotic arm copilot to assist with "pick-and-place" tasks by identifying blocks. |
| Reinforcement Learning (RL) Frameworks [26] | Trains AI agents (copilots) to make optimal decisions to achieve long-term goals in complex environments. | Used to develop the cursor control copilot that learns to assist users in efficiently reaching on-screen targets. |
The integration of AI copilots and shared autonomy represents a paradigm shift in BCI development, offering a powerful strategy to overcome the inherent limitations of both invasive and non-invasive platforms. Experimental data confirms that AI augmentation can dramatically enhance the functional performance of non-invasive BCIs, at times enabling tasks that would otherwise be impossible for users with paralysis [41] [26]. Concurrently, advancements in invasive BCI hardware, benchmarked by rigorous standards like SONIC, continue to push the boundaries of raw information throughput, setting the stage for highly complex applications such as synthetic speech [28].
The future of high-performance BCIs appears to be a synergistic one. The ultimate solution may not be a choice between invasive or non-invasive systems, but rather a combination of strategic hardware choices and sophisticated AI-driven software. This synergy will be key to creating robust, intuitive, and clinically viable BCIs that restore function and independence to patients with neurological conditions.
Overcoming Performance Barriers: Key Challenges and Optimization Strategies in BCI Design
The development of chronic invasive Brain-Computer Interfaces (BCIs) represents a frontier in neurotechnology, offering unparalleled potential for restoring function in neurological disorders. However, their long-term performance is fundamentally constrained by a persistent biological challenge: the foreign body response (FBR). This immune reaction triggers cascading events—glial scar formation, neuronal death, and chronic inflammation—that ultimately lead to signal degradation and device failure [42]. The core of this challenge lies in the mechanical mismatch between conventional rigid implant materials and the delicate, dynamic neural tissue, which experiences continuous micromotion [43] [42]. This review objectively compares the product strategies and material innovations emerging to mitigate these issues, providing a critical analysis for researchers and development professionals navigating the complex trade-offs between invasive and non-invasive BCI paradigms.
The Biological Mechanism of Signal Degradation
Understanding the product performance of various neural implants requires a deep dive into the biological sequence that causes signal degradation. The process begins upon implantation and evolves over weeks and months, fundamentally driven by the body's attempt to isolate the foreign object.
The following diagram illustrates the key stages in the foreign body response that leads to signal degradation in chronic neural implants:
Figure 1: The cascade of the foreign body response leading to signal degradation in chronic neural implants.
The biological cascade, as illustrated, is initiated by the initial mechanical injury from implantation. This injury disrupts the blood-brain barrier (BBB), allowing serum proteins and pro-inflammatory factors to leak into the brain tissue [42]. This environment activates microglia, the brain's resident immune cells, which release neurotoxic cytokines including IL-1, TNF-α, and IL-6 [42]. The sustained presence of the implant leads to a state of chronic inflammation, characterized by ongoing oxidative stress from free radicals. In response, astrocytes become reactive and form a dense glial scar around the implant, a process known as gliosis [42]. Concurrently, the toxic environment and mechanical strain lead to progressive neuronal death in the vicinity of the electrodes. The combined effect of the glial scar and neuronal loss increases the physical distance between the electrodes and viable neurons, which raises interfacial impedance and causes a marked decay in the signal-to-noise ratio (SNR) of recorded neuronal activity, ultimately culminating in device failure [42].
Material and Design Strategies for Improved Biocompatibility
The primary engineering response to the foreign body response has been to develop implants with properties that more closely mimic those of native brain tissue. The following table compares the key material strategies being pursued to achieve this goal.
Table 1: Comparison of Material Strategies for Chronic Neural Implants
| Material Strategy | Key Materials Used | Proposed Mechanism of Action | Reported Advantages | Reported Challenges |
|---|---|---|---|---|
| Soft Conductive Polymers | PEDOT:PSS, PANI [44] | Matches tissue modulus; Ionic conduction improves interface [44] | High biocompatibility; Mixed ionic/electronic conduction [44] | Long-term stability under operational conditions [44] |
| Nanomaterial-Based Coatings | Graphene, Carbon Nanotubes (CNTs) [44] | Nanoscale topography reduces glial activation; High surface area for recording [44] | Excellent electrical & mechanical properties; Ultra-thin, conformal designs [44] | Potential long-term toxicity concerns of free CNTs [44] |
| Ultra-Soft & Flexible Substrates | Polyimide, SU-8, Silicone Elastomers [42] [44] | Reduces mechanical strain and micromotion-induced damage [42] | Significant reduction in chronic glial scarring [42] | Challenging surgical implantation without stiffening shuttles [42] |
| Bioactive Surface Modifications | Peptide sequences, Anti-inflammatory drugs [42] | Disrupts FBR signaling; Promotes neuronal integration [42] | Directly targets molecular pathways of inflammation [42] | Complex fabrication; Potential unintended effects on neural circuits [42] |
Quantitative Performance Benchmarks
Translating these material strategies into functional devices leads to quantifiable differences in performance. The data below, compiled from recent literature, provides a comparative benchmark of how different probe types fare against the traditional, rigid standards.
Table 2: Quantitative Performance Comparison of Neural Probe Technologies
| Probe Technology / Product | Typical Material/Design | Approximate Stiffness | Signal Longevity (Single-Unit) | SNR Change Over Time | Reduction in Glial Scar Thickness |
|---|---|---|---|---|---|
| Traditional Utah/Michigan Array | Silicon, Tungsten, Platinum [42] | ~100 GPa [42] | Weeks to months [42] | Rapid decay (>50% in 8 weeks) [42] | Baseline (No reduction) [42] |
| Neuropixels | Silicon CMOS with metal electrodes [42] | ~100 GPa | Months (intraoperative use) [42] | Moderate decay | Not Reported |
| Flexible Mesh Electronics | SU-8/Pt Nanocomposite [44] | ~1 GPa [44] | >1 year (in mice) [44] | Stable (<10% change) | ~70% reduction vs. tungsten [44] |
| Conductive Polymer-Coated Probes | Metal/PEDOT:PSS [44] | Varies (coating is soft) | Improved months | Initial SNR improvement ~300% [44] | ~30-50% reduction [42] |
| Synchron Stentrode (Endovascular) | Nitinol Mesh [13] | Implanted in blood vessel | 12+ months (human trial) [13] | Stable for LFP/ECoG signals | Avoids direct FBR in brain parenchyma [13] |
Experimental Protocols for Evaluating Biocompatibility and Performance
To generate the comparative data in the tables above, researchers rely on a suite of standardized and advanced experimental protocols. These methodologies are crucial for objectively assessing the performance and biocompatibility of emerging neural implants.
Figure 2: Standard experimental workflow for evaluating neural implant biocompatibility and performance.
Key Experimental Methodologies
Chronic In Vivo Electrophysiology: This is the primary functional assay. Researchers implant the device in an animal model (e.g., rodent or non-human primate) and record neural signals (spikes and local field potentials) over weeks and months [42]. The key metrics tracked include the signal-to-noise ratio (SNR), single-unit yield (the number of detectable neurons), and amplitude of action potentials [42]. A stable device will show minimal decline in these metrics over time. For instance, studies on mesh electronics have demonstrated the ability to record from the same individual neurons for over a year, a benchmark that rigid arrays consistently fail to achieve [44].
Immunohistochemical Analysis: This is the primary structural assay for quantifying the foreign body response. After a defined period, the animal is perfused, and the brain tissue is sectioned and stained for specific cell markers [42]. Critical markers include:
- GFAP (Glial Fibrillary Acidic Protein): To label reactive astrocytes and measure the thickness and density of the glial scar surrounding the probe track.
- Iba1 (Ionized Calcium-Binding Adapter Molecule 1): To label activated microglia and macrophages, indicating the level of acute and chronic inflammation.
- NeuN (Neuronal Nuclei): To quantify neuronal density and survival in the tissue directly adjacent to the implant [42]. The reduction in glial scar thickness and the preservation of nearby neurons, as cited in Table 2, are direct outcomes of this analysis.
Electrochemical Impedance Spectroscopy (EIS): This technique is used to characterize the electrode-tissue interface in vivo. It involves applying a small AC voltage across a range of frequencies and measuring the impedance. A stable or decreasing impedance over time suggests a healthy interface, while a steady increase often indicates the buildup of insulating scar tissue around the electrode [42] [44].
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and testing of next-generation neural implants rely on a specific set of materials and reagents. The following table details key items essential for research in this field.
Table 3: Essential Research Reagents and Materials for Neural Interface Development
| Tool / Material | Function / Application | Specific Examples / Notes |
|---|---|---|
| Flexible Substrate Materials | Forms the structural base of soft probes, providing mechanical compliance. | Polyimide, SU-8 photoresist, Parylene-C, Silicone elastomers (e.g., PDMS) [44]. |
| Conductive Nanomaterials | Creates the electrode sites and interconnects for signal transmission. | Graphene, Carbon Nanotubes (CNTs), Platinum/Gold Nanowires, PEDOT:PSS conductive polymer [44]. |
| Anti-Inflammatory Compounds | Used to functionalize coatings to actively suppress the foreign body response. | Ibuprofen, Dexamethasone, Peptide sequences (e.g., RGD, L1CAM) [42]. |
| Cell Type-Specific Antibodies | Essential for immunohistochemical analysis to identify and quantify the cellular FBR. | Anti-GFAP (astrocytes), Anti-Iba1 (microglia), Anti-NeuN (neurons) [42]. |
| Surgical Implantation Aids | Enables the handling and insertion of ultra-soft devices without damage. | Biodegradable sugar glass shuttles, PEG-based hydrogels, stereotaxic frames with microdrives [42]. |
| Wireless Data Acquisition Systems | Allows for untethered, long-term neural recording in freely behaving animals, critical for chronic studies. | Intan Technologies RHD chips, OpenEphys acquisition systems, custom-built telemetry modules [13]. |
The pursuit of a truly chronic and high-fidelity invasive BCI hinges on solving the biocompatibility challenge. While non-invasive BCIs offer a safer alternative for certain applications, their spatial resolution and signal quality remain fundamentally limited by the skull and scalp [45] [46]. The data clearly shows that strategies based on material and design softening—such as flexible mesh electronics and conductive polymer coatings—demonstrate quantifiable improvements in reducing glial scarring and preserving signal quality over time [42] [44]. Emerging players like Synchron, with its endovascular Stentrode, are pursuing an alternative paradigm by avoiding direct implantation into the brain parenchyma altogether, thereby sidestepping the core FBR [13].
The future of this field lies in the continued blurring of the distinction between man-made devices and natural-born organisms. This will involve the development of "self-healing" materials, fully bioresorbable implants that serve a temporary function, and closed-loop neuromodulation systems that can detect the onset of inflammation and deliver anti-inflammatory drugs on demand [43]. For researchers and drug development professionals, the critical takeaway is that the choice of material and structural design is not merely an engineering concern but a central determinant of biological integration and long-term therapeutic efficacy. The benchmarks and experimental frameworks provided here offer a roadmap for the objective evaluation of the next generation of neural implants as they move from the laboratory to the clinic.
Brain-Computer Interfaces (BCIs) represent a revolutionary technology that establishes a direct communication pathway between the human brain and external devices, offering transformative potential for individuals with motor impairments and neurodegenerative diseases [13] [11]. The fundamental challenge for non-invasive systems, particularly those using electroencephalography (EEG), lies in their inherent susceptibility to signal degradation and environmental noise compared to their invasive counterparts [11] [16]. While non-invasive BCIs offer the significant advantage of not requiring neurosurgery, the skull and scalp act as biological filters, dramatically reducing spatial resolution and signal strength [6]. This performance gap has historically limited the practical application of non-invasive systems.
However, recent advances in both hardware innovation and artificial intelligence (AI) algorithms are rapidly changing this landscape. As of 2025, the convergence of improved sensor technology, sophisticated deep learning models, and multimodal sensing approaches is systematically addressing these core limitations [47] [48] [6]. This guide provides a comparative analysis of the current hardware and algorithmic solutions that are enhancing signal fidelity in non-invasive BCIs, framing their performance within the broader context of invasive BCI benchmarks.
Performance Benchmarks: Non-Invasive vs. Invasive BCI
The performance trade-off between non-invasive and invasive BCIs centers on the balance between risk and data fidelity. The following table summarizes key performance characteristics and applications.
Table 1: Performance Comparison of Invasive vs. Non-Invasive BCIs
| Feature | Invasive BCIs | Non-Invasive BCIs |
|---|---|---|
| Spatial Resolution | Very High (single-neuron level) [13] | Low (scalp-level signals) [11] |
| Signal-to-Noise Ratio | High [16] | Low, requires significant processing [16] |
| Clinical Risk | High (requires brain surgery) [13] | Low or None [11] |
| Primary Market (2025) | Medical/Assistive Technology [3] | Medical, Consumer, Research [3] [16] |
| Key Applications | Restoring speech, motor control [13] | Communication, wheelchair control, neurorehabilitation [47] [48] |
The market and growth projections reflect these distinctions. The non-invasive BCI segment is estimated to grow at a Compound Annual Growth Rate (CAGR) of 9.35% for 2025-2033, compared to 1.49% for invasive BCIs [16]. Meanwhile, the overall BCI market is forecast to grow to over $1.6 billion by 2045 [3]. This growth is largely driven by the safer profile and broader potential applications of non-invasive systems, provided their signal quality continues to improve.
Hardware Solutions: Enhancing Signal Acquisition at the Source
Hardware innovations focus on improving the initial signal acquisition, which is a primary source of noise in the BCI pipeline.
Dry Electrodes and Advanced Sensors
Traditional wet electrodes require conductive gels that can be uncomfortable and impede long-term use. A significant hardware advancement is the development of dry electrodes that eliminate the need for gel, improving user comfort and setup time [3]. Companies like Synaptrix Labs are integrating such dry sensors into their commercial headsets to enhance practicality for daily use [47]. Furthermore, the exploration of novel sensor materials, including e-textiles and skin patches, aims to improve contact quality and signal stability [3].
Multimodal Sensing and High-Density Arrays
A powerful strategy to overcome the limitations of any single sensing modality is multimodal sensing. Combining EEG with other technologies like functional Near-Infrared Spectroscopy (fNIRS) or even emerging modalities like functional Ultrasound (fUS) provides a richer, more correlated dataset [6]. fUS, for instance, can image hemodynamics with promising spatial precision noninvasively [6]. Simultaneously, the development of high-density EEG arrays with more electrodes and better amplifier electronics increases spatial sampling and improves the signal-to-noise ratio (SNR) right at the source, providing AI decoders with more informative raw data [6].
Algorithmic Solutions: Decoding Noisy Signals with AI
Algorithmic advancements are arguably the most impactful area of progress, using AI to extract clear intent from noisy data.
AI Copilots and Shared Autonomy
A breakthrough concept in non-invasive BCI is the use of an "AI copilot" or shared autonomy. This approach does not rely solely on the user's neural signals to control a device. Instead, an AI collaborates with the user, interpreting their high-level intent and handling the precise, noisy details of execution. A UCLA study demonstrated this by using an AI copilot to help a paralyzed participant control a cursor and robotic arm, improving performance by a factor of 3.9 [16]. The participant was unable to complete the tasks without this AI assistance, highlighting its transformative potential.
Advanced Channel Selection and Feature Extraction
EEG caps have many channels, but not all provide useful information for a specific task. Redundant channels introduce noise and slow processing. Advanced channel selection algorithms are therefore critical. One study developed a hybrid method combining statistical tests with a Bonferroni correction to identify and use only the most significant channels [49]. This pre-processing step, focused on reducing dimensionality and removing irrelevant data, resulted in classification accuracies above 90% for motor imagery tasks, significantly outperforming other methods [49].
Hybrid Deep Learning Architectures
For the core task of classifying neural signals, hybrid deep learning models have shown remarkable success. These models combine the strengths of different neural network architectures. For instance, a CNN-LSTM hybrid can simultaneously extract spatial features (using the Convolutional Neural Network) and temporal dependencies (using the Long Short-Term Memory network) from EEG data [50]. One research team reported that their hybrid model achieved a 96.06% accuracy in classifying motor imagery tasks, substantially outperforming traditional machine learning models like Random Forest (91%) and individual deep learning models [50].
Table 2: Comparative Performance of Algorithmic Solutions for Motor Imagery Classification
| Algorithmic Approach | Reported Accuracy | Key Advantage | Study Details |
|---|---|---|---|
| Traditional ML (Random Forest) | 91.00% [50] | Strong baseline, less complex | Used PhysioNet EEG dataset [50] |
| Channel Selection + DLRCSPNN | >90.00% (all subjects) [49] | Reduces noise & computation | Tested on BCI Competition datasets [49] |
| Hybrid Deep Learning (CNN-LSTM) | 96.06% [50] | Captures spatial & temporal features | Used PhysioNet EEG dataset [50] |
Experimental Protocols & Workflows
To ensure reproducibility and provide a clear framework for researchers, this section details the standard experimental workflows derived from the cited literature.
Standard BCI Closed-Loop Processing Pipeline
The following diagram illustrates the standardized sequential workflow for a closed-loop BCI system, which is foundational to most modern BCI research.
This closed-loop design is the backbone of current BCI research, allowing the user to adapt their mental strategy based on real-time feedback [13]. The pipeline consists of four standard, sequential components: signal acquisition, feature extraction, feature translation, and device output [48].
Detailed Methodology for a Hybrid Deep Learning Experiment
A specific experiment achieving 96.06% accuracy provides a clear template for implementing a hybrid deep learning approach. The workflow involves a structured pipeline from data preparation to model evaluation, with key techniques applied at each stage.
Key Methodological Details:
- Dataset: The "PhysioNet EEG Motor Movement/Imagery Dataset" was used, which encompasses data from various actual and imagined movements [50].
- Pre-processing: The pipeline included normalization, band-pass filtering, and artifact removal to ensure clean, standardized signals [50].
- Feature Extraction: Advanced techniques like Wavelet Transform and Riemannian Geometry were employed to capture critical time-frequency characteristics and the intrinsic geometric structure of the EEG data [50].
- Data Augmentation: Generative Adversarial Networks (GANs) were utilized to generate synthetic EEG data, which helped balance the dataset and improve the model's ability to generalize [50].
- Model Training: The training process was optimized, with the model reaching peak accuracy within just 30–50 epochs, making it efficient for practical applications [50].
The Scientist's Toolkit: Essential Research Reagents & Materials
For researchers aiming to build upon these solutions, the following table catalogs key hardware, software, and datasets that constitute the essential toolkit for this field.
Table 3: Essential Research Toolkit for Non-Invasive BCI Development
| Tool Category | Specific Examples | Function & Application |
|---|---|---|
| Hardware Platforms | Synaptrix Labs' Neuralis Headset [47], Cognixion's Axon-R Nucleus [51], IDUN Technologies' EEG Hardware [47] | Commercial platforms featuring dry electrodes, integrated biosensing, and licensing opportunities for R&D. |
| Software & Algorithms | Convolutional Neural Network-Kalman Filter (CNN-KF) [16], Hybrid CNN-LSTM Models [50], DLRCSPNN Framework [49] | AI models for noise filtering, intent decoding, and classification of motor imagery tasks. |
| Public Datasets | BCI Competition III & IV [49], PhysioNet EEG Motor Movement/Imagery Dataset [50] | Benchmark datasets for training and validating new machine learning models and algorithms. |
| Analysis Techniques | Bonferroni Correction Channel Reduction [49], Riemannian Geometry [50], Wavelet Transform [50] | Statistical and mathematical methods for selecting relevant EEG channels and extracting robust features. |
The frontier of non-invasive BCI research is being pushed forward by a dual-track approach: refining hardware for cleaner signal acquisition and developing increasingly sophisticated AI algorithms to decode noisy data. While fundamental physical limits remain, solutions like AI copilots, hybrid deep learning models, and advanced channel selection are dramatically improving functional performance. The experimental protocols and research toolkit outlined herein provide a foundation for continued innovation. As these hardware and algorithmic solutions mature and converge, they are poised to narrow the performance gap with invasive systems for many applications, thereby democratizing access to BCI technology for a broader population in need of assistive communication and rehabilitation tools.
The transition of Brain-Computer Interfaces (BCIs) from laboratory demonstrations to real-world applications hinges on optimizing the critical trade-offs between three fundamental performance metrics: Information Transfer Rate (ITR), latency, and error rates. This comparative guide analyzes how invasive and non-invasive BCI technologies balance these parameters across medical, assistive, and research applications. Current market forecasts indicate the overall BCI sector is projected to grow to over $1.6 billion by 2045, with non-invasive technologies exhibiting particularly strong growth potential [3]. The following analysis synthesizes performance data from recent studies and clinical trials to provide researchers and development professionals with evidence-based comparisons of leading BCI approaches.
Performance Benchmarking: Invasive vs. Non-Invasive BCI Technologies
The fundamental dichotomy in BCI design—invasive versus non-invasive implementation—represents a core trade-off between signal fidelity and practical implementation constraints. The table below summarizes key performance characteristics across these categories based on current technological capabilities.
Table 1: Fundamental Performance Trade-offs in BCI Technologies
| Parameter | Invasive BCIs | Non-Invasive BCIs |
|---|---|---|
| Spatial Resolution | Micrometer scale (single neuron recording) [52] | Centimeter scale (signal averaging over large neural populations) [52] [11] |
| Temporal Resolution | Millisecond (direct neural firing capture) [13] | Millisecond (EEG); Seconds (fNIRS) [52] [45] |
| Signal-to-Noise Ratio | High (minimal signal attenuation) [16] | Lower (signal degradation through skull, muscle artifacts) [16] [11] |
| Best-Case ITR | High (≥100 bits/min in motor control) [52] | Moderate (96.1 bits/min in hybrid SSVEP-EMG systems) [53] |
| Typical Latency | Low (direct signal pathway) [13] | Variable (EEG: <0.25s with AI; fNIRS: several seconds) [16] [45] |
| Clinical Risk Profile | Higher (surgical implantation, tissue response) [13] [11] | Lower (minimal physical risk) [45] [11] |
The performance differential stems from fundamental biophysical principles. Invasive systems using Microelectrode Arrays (MEAs) capture single-neuron activity with micrometer spatial resolution, while electrocorticography (ECoG) positioned on the cortical surface measures signals averaged over thousands of neurons [52]. Non-invasive approaches like electroencephalography (EEG) detect signals averaged over significantly larger neural populations after substantial attenuation and distortion through cerebrospinal fluid, skull, and scalp tissues [52] [11].
Quantitative Performance Comparison Across BCI Paradigms
Different neural signal acquisition methods and decoding approaches yield substantially varied performance profiles. The following table synthesizes quantitative metrics from recent experimental studies across multiple BCI paradigms.
Table 2: Experimental Performance Metrics Across BCI Paradigms
| BCI Paradigm | Best Reported Accuracy | ITR (bits/min) | Latency/Response Time | Key Applications |
|---|---|---|---|---|
| Invasive Motor Decoding | Not explicitly quantified | Highly variable based on channel count [52] | Low millisecond range for signal acquisition [52] | Exoskeleton control, robotic arms, functional electrical stimulation [52] [13] |
| Non-Invasive EEG with AI | Not explicitly quantified | Not reported | <0.25 seconds with AI enhancement [16] | Cursor control, basic device manipulation [16] |
| c-VEP BCI | >97% (with sufficient calibration) [54] | Not reported | 2-3s decoding window (to achieve >95% accuracy) [54] | Communication systems, environmental control |
| Hybrid SSVEP-EMG Speller | 91.2% [53] | 96.1 [53] | 0.2s latency for SSVEP component [53] | Spelling applications, communication devices |
| fNIRS | Not explicitly quantified | Limited by hemodynamic response | Several seconds [52] [45] | Basic state monitoring, non-time-critical applications |
Performance optimization reveals counterintuitive relationships between system parameters. Research indicates a negative correlation between power consumption per channel (PpC) and Information Transfer Rate (ITR) in decoding circuits, suggesting that increasing channel count can simultaneously reduce PpC through hardware sharing while increasing ITR through additional input data [52]. This finding has significant implications for implementing BCIs in power-constrained implantable or wearable devices.
Experimental Protocols and Methodologies
Invasive BCI Motor Decoding Protocols
Invasive BCI systems typically employ microelectrode arrays (MEAs) implanted in motor cortex regions to capture neural signals associated with movement intention. The general workflow follows a standardized acquisition-processing-output pipeline:
Figure 1: Invasive BCI Motor Decoding Workflow
Hardware implementations focus on optimizing power efficiency for battery-powered or implantable devices. decoding circuits for EEG and ECoG are dominated by signal processing complexity rather than data acquisition power requirements [52]. For systems requiring high channel counts, recent designs implement hardware sharing architectures that reduce power consumption per channel while maintaining decoding performance.
Non-Invasive BCI Enhancement Protocols
AI Copilot Integration
A recent breakthrough in non-invasive BCI performance comes from UCLA research integrating artificial intelligence as a "copilot" to enhance system capabilities. The experimental protocol employed:
- Signal Acquisition: 64-channel EEG cap recording from both healthy participants and paraplegic patients with T5-level spinal cord injury [16]
- AI Architecture: Convolutional Neural Network-Kalman Filter (CNN-KF) combining pattern recognition capabilities with recursive estimation for noisy time-series data [16]
- Task Design: Cursor control and robotic arm manipulation tasks with varying complexity levels
- Performance Metric: Comparison of task completion with and without AI assistance
This approach demonstrated a 3.9x performance improvement for the paralyzed participant in both cursor control and robotic arm tasks, with the research noting that successful task completion would not have been possible without AI augmentation [16].
Hybrid SSVEP-EMG Implementation
Hybrid BCI systems combine multiple signal modalities to overcome limitations of individual approaches. A recent speller implementation achieved high ITR through:
- Stimulus Design: Nine flickering symbols with each symbol mapped to four characters controlled via EMG activation patterns [53]
- Character Encoding: Frequency-based encoding assigning simpler muscle activations to characters with higher occurrence probability [53]
- Signal Processing: Separate processing pipelines for SSVEP (occipital electrodes O1, O2, Oz) and EMG (flexor carpi radialis) [53]
- Temporal Parameters: 2-second processing window following 0.2-second SSVEP latency period [53]
Figure 2: Hybrid SSVEP-EMG BCI Architecture
Calibration-Time Optimization in c-VEP BCIs
Calibration requirements significantly impact practical BCI usability. Research on code-modulated visual evoked potential (c-VEP) systems reveals:
- Performance Trade-off: Clear relationship between calibration duration and decoding performance [54]
- Stimulus Design Impact: Binary checkerboard stimuli with spatial frequency of 1.2 c/º achieved 95% accuracy within 2s decoding window using only 7.3s calibration [54]
- Minimum Requirements: At least 1-minute calibration needed for stable c-VEP response estimation in template-matching paradigms [54]
Implementation Considerations for Real-World Deployment
Clinical and Assistive Technology Applications
The translation of BCI technologies to clinical practice requires careful consideration of application-specific requirements:
- Spinal Cord Injury Rehabilitation: Recent meta-analysis of 9 studies with 109 SCI patients found non-invasive BCI interventions significantly improved motor function (SMD=0.72), sensory function (SMD=0.95), and activities of daily living (SMD=0.85) [45]
- Communication Applications: Hybrid BCI spellers demonstrate the practical application of performance optimization, with systems achieving 20.9 characters per minute representing viable communication rates for severely paralyzed users [53]
- Neuroprosthetic Control: Invasive systems show promise for direct control of exoskeletons and robotic arms, though signal stability over time remains a challenge [13]
The Research Toolkit: Essential Components for BCI Implementation
Table 3: Essential Research Reagents and Solutions for BCI Development
| Component Category | Specific Examples | Function/Purpose |
|---|---|---|
| Signal Acquisition | EEG electrodes (wet/dry), MEA arrays, ECoG grids, fNIRS optodes | Neural signal capture with varying invasiveness and spatial resolution [3] [11] |
| Reference Algorithms | CNN-Kalman Filter, SSVEP template matching, EMG pattern classification | Signal processing and intent decoding with different computational requirements [52] [16] [53] |
| Stimulation Paradigms | Binary/Non-binary c-VEP sequences, SSVEP flicker patterns, Motor imagery tasks | Eliciting measurable neural responses for specific BCI applications [54] [53] |
| Performance Metrics | ITR, Classification Accuracy, Latency, Calibration Time | Quantitative assessment of BCI performance across multiple dimensions [52] [54] [53] |
| Hardware Platforms | Low-power ASICs, Implantable wireless systems, Mobile EEG headsets | Implementation platforms balancing performance, power consumption, and usability [3] [52] |
Selecting and optimizing BCI technologies for real-world applications requires careful balancing of the ITR-latency-error rate triangle based on specific use case requirements:
- High-Performance Motor Control: Invasive systems (MEA, ECoG) currently provide superior performance for complex tasks like robotic arm control, despite higher surgical risk and regulatory hurdles [52] [13]
- Practical Communication Systems: Hybrid non-invasive approaches (SSVEP-EMG) offer compelling performance for spelling applications with minimal user risk [53]
- Clinical Rehabilitation: Non-invasive EEG-based systems demonstrate significant functional improvements for SCI patients, particularly when enhanced with AI copilot systems [16] [45]
- Balanced Usability: c-VEP systems with optimized calibration protocols provide reasonable performance while minimizing user burden [54]
Future BCI development will continue to refine these trade-offs through advances in neural signal processing, minimally invasive implantation techniques, and adaptive AI systems that learn individual user patterns. The emerging landscape suggests a future with multiple BCI modalities optimized for specific applications rather than a single dominant technology.
The evolution of brain-computer interfaces (BCIs) and neuromodulation technologies has increasingly focused on minimizing invasiveness while maintaining high-quality signal acquisition and therapeutic efficacy. Within this landscape, two prominent approaches—endovascular and epidural interfaces—offer distinct pathways for accessing neural information. Endovascular BCIs represent a revolutionary approach that utilizes the body's natural vascular pathways to place recording electrodes within blood vessels near neural targets [55] [56]. In contrast, epidural interfaces occupy an intermediate position between non-invasive and fully invasive methods, requiring craniotomy but maintaining an external position relative to the dura mater [57]. This comprehensive analysis compares the technical capabilities, experimental performance, and clinical applications of these two emerging technologies to inform researchers, scientists, and drug development professionals working at the intersection of neurotechnology and therapeutic development.
Technical Specifications and Performance Benchmarks
Physical and Operational Characteristics
Endovascular and epidural interfaces differ fundamentally in their implantation methodology, anatomical placement, and operational parameters. Endovascular systems typically consist of stent-mounted electrode arrays deployed via catheter into cortical veins such as the superior sagittal sinus [58] [56]. These devices integrate with the vascular endothelium over time, potentially enhancing signal stability [56]. Epidural interfaces are placed above the dura mater through craniotomy, creating direct contact with the skull but maintaining separation from cortical tissue [57]. The table below summarizes key comparative characteristics:
Table 1: Physical and Operational Characteristics of Neural Interfaces
| Characteristic | Endovascular Interface | Epidural Interface |
|---|---|---|
| Implantation Method | Minimally invasive vascular catheterization | Surgical craniotomy required |
| Anatomic Position | Within blood vessels (e.g., superior sagittal sinus) | Epidural space (between skull and dura) |
| Surgical Risk Profile | Lower risk of brain tissue damage; vascular complications possible | Higher risk of infection, surgical complications; no brain penetration |
| Chronic Stability | Potential improvement through endothelialization | Subject to scar tissue formation |
| Target Engagement | Deep cortical structures accessible via vasculature | Cortical surface primarily |
| Device Examples | Stentrode | Conventional epidural grids |
Quantitative Performance Metrics
Direct comparative studies provide objective performance metrics for evaluating neural interface technologies. Research simultaneously examining endovascular, subdural, and epidural signal quality in sheep models reveals critical insights into the operational characteristics of these systems [57]. The following table summarizes key quantitative findings from comparative studies:
Table 2: Experimental Performance Metrics of Neural Interfaces
| Performance Metric | Endovascular Interface | Epidural Interface | Subdural Interface |
|---|---|---|---|
| Signal-to-Noise Ratio (SNR) | Not significantly different from epidural/subdural [57] | Not significantly different from endovascular/subdural [57] | Reference standard |
| Bandwidth | Comparable to conventional interfaces (<500 Hz for LFP) [57] | Comparable to conventional interfaces [57] | Reference standard |
| Spatial Resolution | Location and frequency dependent [57] | Location and frequency dependent [57] | 2-6 mm typical [57] |
| Decoding Accuracy | Comparable between electrode types [57] | Comparable between electrode types [57] | Reference standard |
| Signal Amplitude | 2-5 times higher than scalp EEG [56] | Higher than scalp EEG | Highest amplitude |
| Chronic Implantation | Demonstrated up to 6 months in sheep models [56] | Long-term use demonstrated in humans | Long-term use demonstrated |
Experimental Methodologies and Protocols
Endovascular BCI Implementation
Endovascular BCI research employs standardized methodologies for device validation. The typical experimental workflow involves venous access through the jugular vein, followed by catheter-based navigation of electrode arrays to target locations in the superior sagittal sinus under fluoroscopic guidance [56]. Chronic implantation studies conduct recordings over extended periods (weeks to months) to assess signal stability and biological integration [57]. Signal processing pipelines typically include bandpass filtering (0.5-500 Hz), common average referencing, and artifact removal techniques [57]. Performance validation often employs motor imagery tasks or evoked potentials with decoding algorithms such as support vector machines or deep learning networks to translate neural signals into device commands [59].
Figure 1: Endovascular BCI Experimental Workflow
Comparative Signal Quality Assessment
Rigorous comparison of neural interfaces requires controlled experimental conditions. Simultaneous recording protocols implant endovascular, epidural, and subdural arrays in the same subject to enable direct signal quality comparison [57]. Studies typically employ evoked potential paradigms (e.g., median nerve stimulation) to generate standardized neural responses across recording modalities [57]. Quantitative analysis includes frequency bandwidth assessment through power spectral density analysis, signal-to-noise ratio calculation from evoked responses, and spatial resolution estimation through cross-channel correlation analysis [57]. Classification accuracy for intended movements or stimuli provides a practical performance metric with direct relevance to BCI applications [57].
Signaling Pathways and Neural Information Access
The biological interfaces between recording technologies and neural activity fundamentally differ between endovascular and epidural approaches. Endovascular devices access neural signals through multiple tissue layers: endothelial lining of the blood vessel, vessel wall, cerebrospinal fluid (CSF), pia mater, and subarachnoid space before reaching cortical tissue [56] [57]. This multi-layer interface potentially filters high-frequency components of neural signals. Epidural interfaces record through the dura mater, CSF, and pia mater, but avoid direct vascular integration [57]. The distinctive signal transmission characteristics of these pathways explain observed differences in signal resolution and frequency content between modalities.
Figure 2: Neural Signal Pathways for Different Interface Modalities
Research Reagent Solutions and Essential Materials
The experimental and clinical implementation of endovascular and epidural interfaces requires specialized materials and technical resources. The following table details key components of the research toolkit for scientists developing and evaluating these technologies:
Table 3: Essential Research Materials for Neural Interface Development
| Component Category | Specific Examples | Research Function | Technology Application |
|---|---|---|---|
| Electrode Arrays | Stentrode, epidural grid electrodes, subdural strips | Neural signal acquisition | Both endovascular and epidural |
| Implantation Tools | Delivery catheters, Tuohy epidural needles, guidewires | Device placement and positioning | Both endovascular and epidural |
| Navigation Systems | Fluoroscopic imaging, neuromavigation systems | Anatomic targeting accuracy | Both endovascular and epidural |
| Signal Processors | Amplifiers, filters, analog-to-digital converters | Signal conditioning and digitization | Both endovascular and epidural |
| Decoding Algorithms | Support vector machines, convolutional neural networks, LSTM networks | Neural signal translation to commands | Both endovascular and epidural |
| Anti-Thrombotic Coatings | Heparin-coated stents, antiplatelet surface treatments | Prevention of vascular occlusion | Primarily endovascular |
| Biocompatible Materials | Platinum-iridium electrodes, silicone polymer insulation | Long-term tissue compatibility | Both endovascular and epidural |
Discussion and Future Directions
The comparative analysis of endovascular and epidural approaches reveals a consistent trade-off between invasiveness and signal fidelity. While both technologies provide signals superior to non-invasive alternatives like scalp EEG [56] [57], endovascular methods offer a compelling combination of minimal surgical intervention and robust signal quality comparable to more invasive interfaces [55] [57].
For researchers designing clinical trials or preclinical studies, selection between these technologies should consider specific application requirements. Endovascular approaches show particular promise for conditions requiring deep cortical access or where patient acceptance of craniotomy is low [56]. Epidural interfaces may remain preferable for applications requiring broad cortical coverage or where vascular anatomy is unsuitable for stent deployment [57].
Future development should address specific limitations of each approach. For endovascular interfaces, reducing thrombotic risk through improved device materials and anti-coagulation protocols remains a priority [56]. Enhanced electrode density and miniaturization could improve spatial resolution constrained by the vascular interface [57]. For epidural systems, reducing foreign body response and improving long-term signal stability through advanced biocompatible materials represents an important research direction [57].
The rapid advancement of minimally invasive neural interfaces holds significant implications for drug development and therapeutic testing. These technologies enable more precise monitoring of neurological drug effects and create new opportunities for closed-loop therapeutic systems that adapt to neural state changes [56] [59]. As the field progresses, standardized performance metrics and validation protocols will be essential for meaningful comparison across studies and acceleration toward clinical translation.
Validating the Hype: A Standardized Framework for Comparing BCI Technologies and Market Leaders
The field of brain-computer interfaces has experienced remarkable growth, with PubMed and Scopus showing a consistent increase in BCI-related publications [9]. However, this rapid expansion has created a significant challenge: the varied nature of BCIs, combined with the large number of laboratories conducting research, has made uniform performance reporting exceptionally difficult [9]. This lack of standardization hinders direct comparison between different BCI approaches, obscures true technological progress, and ultimately slows the translation of research into practical applications [28] [7].
The problem is particularly acute when comparing invasive and non-invasive approaches. Invasive BCIs, which involve surgical implantation, typically offer higher signal quality but carry greater clinical risk. Non-invasive BCIs provide greater accessibility and safety but generally deliver lower information throughput [29]. Without standardized benchmarks, claims about performance advantages remain difficult to verify independently.
This article examines the current landscape of BCI performance metrics, introduces the newly proposed SONIC benchmarking framework, and provides experimental methodologies and comparative data to enable objective evaluation across different BCI architectures.
Established BCI Performance Metrics and Reporting Standards
Core Performance Metrics
Before examining specific benchmarking frameworks, it is essential to understand the fundamental metrics used to evaluate BCI performance. The BCI research community has established several key quantitative measures, each capturing different aspects of system capability:
Table 1: Fundamental BCI Performance Metrics
| Metric | Description | Application Context |
|---|---|---|
| Information Transfer Rate (ITR) | Measures the speed of information transfer, typically in bits per second (bps) or bits per trial | Communication systems, spelling devices |
| Classification Accuracy | Percentage of correct classifications in discrete tasks | All BCI types, particularly for control applications |
| Latency | Delay between user intention and system response | Real-time control applications, gaming, prosthetics |
| Signal-to-Noise Ratio (SNR) | Ratio of desired signal power to noise power | Signal quality assessment across all BCI types |
| Area Under Curve (AUC) | Measure of classifier performance across all classification thresholds | Movement detection, intent recognition |
Beyond these core metrics, comprehensive evaluation should also consider usability (effectiveness and efficiency), user satisfaction (including BCI-related aspects), and system usage (match between system and user) [7]. These human-factor dimensions are particularly crucial for translating BCI technology from laboratory demonstrations to practical applications [60].
General Reporting Guidelines for BCI Research
To improve reproducibility and comparison across studies, researchers have developed checklists for reporting BCI methods and results. These guidelines help ensure that critical experimental details are not omitted:
Table 2: Essential Reporting Elements for BCI Studies
| Methods Section | Results Section |
|---|---|
| Equipment specifications (electrodes, amplifiers) | Chance performance (theoretical and empirical) |
| Sensor/electrode number and location | Confidence intervals for key metrics |
| Participant demographics and relevant medical conditions | Idle or "no-control" performance |
| Experimental protocol timing and duration | Online/closed-loop performance metrics |
| Data quantity (trials per subject for training and testing) | Individual participant results and variability |
The timing of BCI tasks deserves special attention, as the inclusion or exclusion of inter-trial intervals and other pauses can significantly affect reported performance metrics like ITR [9]. Transparent reporting of exactly which time periods are included in calculations enables meaningful cross-study comparisons.
The SONIC Benchmarking Framework: A New Standard
The SONIC (Standard for Optimizing Neural Interface Capacity) benchmark represents a significant step toward standardized performance assessment in BCI research [28]. Developed by Paradromics, this open benchmarking standard aims to provide rigorous, application-agnostic metrics that reflect underlying system properties, similar to how benchmark tests drive progress in the semiconductor industry [28].
SONIC addresses two critical limitations in previous BCI performance reporting: (1) the reliance on theoretical frameworks that make assumptions often not valid in complex neural systems, and (2) the failure to adequately account for latency in performance assessments [28]. By measuring actual information transfer while controlling for delay, SONIC provides a more honest representation of real-world BCI capabilities.
Experimental Protocol and Implementation
The SONIC benchmarking methodology employs a carefully controlled experimental design:
In preclinical validation, SONIC was implemented using sheep models with fully implanted, wireless Connexus BCI devices [28]. The benchmark involves:
Stimulus Presentation: Controlled sequences of sounds are presented to the subject. Each character is encoded as a unique five-note musical tone sequence, creating a dictionary for information transfer measurement.
Neural Recording: The implanted BCI records neural activity from the auditory cortex while sounds are presented.
Decoding Phase: The system predicts which sounds were presented based solely on the recorded neural activity.
Metric Calculation: The mutual information between presented sounds and predicted sounds is calculated to determine the true information transfer rate, while latency is simultaneously measured.
This approach specifically addresses the trade-off between speed and accuracy by testing multiple encoding schemes and reporting performance across different operating points [28].
Key Performance Achievements
Using the SONIC benchmark, Paradromics has demonstrated record-setting performance with their Connexus BCI system:
- >200 bits per second with 56ms total system latency
- >100 bits per second with 11ms total system latency [28]
These rates are notable for exceeding the information transfer rate of transcribed human speech (~40 bps), suggesting sufficient capacity for high-performance communication BCIs [28]. The benchmark also highlights the critical importance of latency - while high information transfer rates are valuable, many applications (like conversational speech) cannot tolerate long delays [28].
Comparative Performance Analysis: Invasive vs. Non-Invasive BCI Technologies
Performance Benchmarking Across BCI Modalities
Direct comparison of BCI technologies reveals significant differences in performance capabilities between invasive and non-invasive approaches:
Table 3: Comparative Performance of BCI Technologies
| BCI Technology | Max Reported ITR | Typical Latency | Key Applications | Notable Studies |
|---|---|---|---|---|
| Invasive (Intracortical) | 200+ bps [28] | 11-56 ms [28] | Communication, motor control | Paradromics Connexus, Neuralink |
| Non-invasive (EEG) | 0.5-1.5 bps [45] [29] | 100-500 ms [29] | Rehabilitation, basic control | Robotic hand control [29] |
| Non-invasive (fNIRS) | Not quantified in bps | 1-2 second delay [61] | Cognitive state monitoring | Memory prosthesis [61] |
| Non-invasive BSI | Not quantified in bps | Not specified | Motor rehabilitation | Brain-spine interface [62] |
The performance disparity between invasive and non-invasive approaches is substantial, with invasive systems demonstrating orders of magnitude higher information transfer rates. However, this raw performance advantage must be balanced against the significantly higher risk profile of surgical implantation.
Application-Specific Performance Requirements
Different BCI applications have varying performance requirements, which influences the suitability of different technological approaches:
Non-invasive BCIs have demonstrated particular promise in rehabilitation applications. For spinal cord injury patients, non-invasive BCI interventions have shown significant impacts on motor function (SMD = 0.72), sensory function (SMD = 0.95), and activities of daily living (SMD = 0.85) [45]. These systems typically achieve workable performance despite lower information transfer rates because rehabilitation applications can tolerate slower, more deliberate interactions.
Experimental Protocols and Methodologies
Protocol for Non-Invasive Motor Decoding
Recent advances in non-invasive BCI have enabled increasingly sophisticated applications, such as the real-time robotic hand control at the individual finger level demonstrated by [29]. Their experimental protocol provides an excellent example of rigorous methodology in non-invasive BCI research:
Participant Selection and Training:
- 21 able-bodied individuals with previous BCI experience
- Each participant completed one offline familiarization session and two online sessions for both motor execution and motor imagery tasks
- Subject-specific decoding models were trained using the offline session data
Signal Acquisition and Processing:
- EEG signals were acquired using standard high-density systems
- Deep learning architecture (EEGNet-8.2) was implemented for real-time decoding of individual finger movements
- Fine-tuned models were further trained from base models using same-day data to address inter-session variability
Performance Validation:
- Binary classification (thumb vs. pinky) and ternary classification (thumb vs. index vs. pinky) paradigms were tested
- Majority voting accuracy was calculated across multiple segments of each trial
- Visual feedback (color change on screen) and physical feedback (robotic finger movement) were provided simultaneously
This study achieved notable performance for a non-invasive system: 80.56% accuracy for two-finger motor imagery tasks and 60.61% for three-finger tasks [29]. The use of fine-tuning demonstrated significant performance improvements across sessions (F = 14.455, p = 0.001 for binary tasks; F = 24.590, p < 0.001 for ternary tasks) [29].
Non-Invasive Brain-Spine Interface Protocol
The development of non-invasive brain-spine interfaces represents another important application category with distinct methodological requirements [62]:
System Architecture:
- EEG system (32-channel gNautilus headset) for detecting movement intention
- Transcutaneous spinal cord stimulation (tSCS) for facilitating motor responses
- EMG recording for validation and feedback
Decoder Development:
- Linear discriminant analysis (LDA) decoder trained to detect knee extension onset
- Features focused on event-related desynchronization in central-medial cortical regions
- Multiple frequency bands utilized: μ (8-12 Hz), low β (16-20 Hz), and high β (24-28 Hz)
Experimental Paradigm:
- Cued movement tasks for initial decoder training
- Generalization testing to imagery and uncued movement conditions
- Online testing with brain-controlled stimulation delivery
This approach achieved an average area under the curve (AUC) of 0.83 ± 0.06 during cued movement tasks offline, and maintained performance (AUC = 0.81 ± 0.05) when implemented in real-time with closed-loop stimulation [62].
Essential Research Reagents and Equipment
Successful BCI research requires specialized equipment and computational tools. The following table summarizes key components used in the studies reviewed:
Table 4: Essential Research Solutions for BCI Development
| Category | Specific Tools/Solutions | Research Function | Example Implementation |
|---|---|---|---|
| Signal Acquisition | 32-channel EEG (gNautilus) [62] | Neural signal recording | Brain-spine interface [62] |
| Stimulation Hardware | Biphasic constant current stimulator (DS8R) [62] | Transcutaneous spinal cord stimulation | Non-invasive BSI [62] |
| Computational Framework | BCI2000 software [62] | General-purpose BCI research platform | Experimental control and data acquisition |
| Deep Learning Architecture | EEGNet-8.2 [29] | EEG signal decoding | Individual finger movement classification |
| fNIRS Systems | ISS Imagent device [61] | Prefrontal cortex activation monitoring | Implicit BCI for memory prosthesis |
| Performance Benchmarking | SONIC framework [28] | Standardized performance assessment | Comparative evaluation of BCI technologies |
The introduction of standardized benchmarking frameworks like SONIC represents a critical maturation point for BCI technology. By enabling direct, transparent comparison across different approaches, such standards accelerate innovation and help focus research efforts on technologies with genuine potential for clinical impact.
The comparative data clearly shows that invasive and non-invasive BCIs occupy complementary rather than competitive positions in the technology landscape. Invasive systems offer unparalleled performance for applications requiring high bandwidth and low latency, such as communication prosthetics for severely disabled individuals. Non-invasive systems provide safer, more accessible platforms for rehabilitation and basic control applications where ultimate performance is less critical than usability and deployability.
As the field progresses, comprehensive evaluation must extend beyond technical metrics like ITR and accuracy to include usability, user satisfaction, and real-world performance [7] [60]. The adoption of rigorous, standardized benchmarks will be essential for translating BCI technology from laboratory demonstrations to practical applications that improve human health and capability.
In the rapidly advancing field of brain-computer interfaces, two engineering metrics have emerged as fundamental for evaluating system performance: Information Transfer Rate (ITR) and latency. As BCIs transition from laboratory research to clinical applications, these quantitative benchmarks provide an objective framework for comparing the capabilities of various platforms, particularly in the critical comparison between invasive and non-invasive approaches [28] [13]. ITR, measured in bits per second (bps), quantifies how much data can be reliably communicated from the brain to an external device per unit of time. Latency, measured in milliseconds (ms), represents the delay between neural activity and the corresponding system output [28]. For applications such as speech restoration or real-time motor control, both metrics must be optimized simultaneously—high ITR enables complex communication, while low latency ensures natural, responsive interaction [28] [63].
This comparative analysis examines the current performance benchmarks of leading BCI platforms, focusing on their respective positions within the invasive-to-non-invasive spectrum. The data reveals significant disparities in technical capabilities, highlighting how different engineering approaches and implantation strategies yield substantially different performance characteristics with direct implications for clinical applicability [28] [13].
Performance Comparison of Leading BCI Platforms
Quantitative Benchmark Analysis
Table 1: Comparative ITR and Latency Performance of Leading BCI Platforms (2025)
| Company/Platform | Approach/Implantation | Information Transfer Rate (ITR) | Latency | Key Applications |
|---|---|---|---|---|
| Paradromics (Connexus BCI) | Invasive (Cortical Array) | 200+ bps (high-accuracy mode); 100+ bps (low-latency mode) [28] | 56 ms (200 bps mode); 11 ms (100 bps mode) [28] | Speech decoding, communication restoration [28] |
| Neuralink | Invasive (Cortical Threads) | ~10 bps (estimated from clinical trial reports) [28] | <50 ms (system specification) [63] | Motor control, cursor control, planned speech applications [63] [13] |
| Blackrock Neurotech | Invasive (Utah Array) | ~5-10 bps (comparable to Neuralink in academic studies) [28] [13] | Not specifically reported | Motor control, academic research [13] |
| Synchron (Stentrode) | Minimally Invasive (Endovascular) | ~1-2 bps (estimated from reported performance) [28] | Not specifically reported | Texting, basic device control [13] |
| Non-invasive BCI (EEG-based) | Non-invasive (Scalp EEG) | <1 bps (typical performance range) [26] | Variable, typically >100ms [11] | Basic control, rehabilitation, research [11] [45] |
Table 2: BCI Platform Characteristics and Clinical Status
| Platform | Invasiveness Level | Electrode Count/Type | Clinical Trial Status (2025) | Key Differentiators |
|---|---|---|---|---|
| Paradromics Connexus | Fully invasive | 421 electrodes, modular array [13] | First-in-human recording completed; full trial planned for late 2025 [13] | Highest demonstrated ITR; wireless; focused on speech [28] |
| Neuralink | Fully invasive | 96-1024 flexible threads [63] [13] | Human trials ongoing for motor control; speech trial planned [63] | Robotic implantation; custom ASICs; high channel count [63] [13] |
| Synchron Stentrode | Minimally invasive | Stent-based electrode array [13] | Multi-patient trials completed; pivotal trial planned [13] | Endovascular approach; no brain incision [13] |
| Blackrock Neurotech | Fully invasive | Utah array (traditional); Neuralace (developing) [13] | Multiple human trials; long-term research use [13] | Established research use; developing less invasive flexible array [13] |
| Precision Neuroscience | Minimally invasive | Layer 7 cortical surface array [13] | FDA clearance for ≤30-day implantation [13] | Cortical surface placement; minimal tissue penetration [13] |
Performance Analysis and Interpretation
The performance data reveals a clear trade-off between invasiveness and performance. Paradromics currently leads in raw performance metrics, demonstrating ITRs that are 20 times faster than initial Neuralink clinical results and 100-200 times faster than Synchron's reported performance [28]. This performance advantage comes at the cost of higher invasiveness, requiring direct cortical implantation.
Notably, Paradromics' benchmarking emphasizes the importance of measuring both ITR and latency simultaneously, as some systems can appear to achieve high transfer rates by introducing significant delays that make real-time interaction impractical [28]. Their demonstration that the Connexus BCI can achieve ITRs exceeding the information rate of transcribed human speech (~40 bps) provides strong evidence for the clinical feasibility of thought-based communication systems [28].
Experimental Protocols and Benchmarking Methodologies
The SONIC Benchmarking Standard
Paradromics has introduced the Standard for Optimizing Neural Interface Capacity (SONIC) as an open benchmarking framework designed to provide standardized, application-agnostic performance measurements for BCI systems [28]. The methodology involves:
- Stimulus Presentation: Controlled sequences of sounds (five-tone sequences mapped to characters) are presented to an animal model (sheep) [28].
- Neural Recording: The fully implanted BCI system records neural activity from the auditory cortex while stimuli are presented [28].
- Decoding and Prediction: Algorithms process the neural data to predict which sounds were presented [28].
- Mutual Information Calculation: The information transfer rate is calculated by measuring the mutual information between the presented sounds and the system's predictions, providing a rigorous measure of how much information is being transmitted through the neural interface [28].
This approach allows for preclinical benchmarking that reflects fundamental system capabilities independent of specific applications, similar to how benchmark tests accelerated progress in the semiconductor industry [28].
Shared Autonomy in Non-Invasive BCI
For non-invasive systems, recent research demonstrates an alternative approach to improving practical performance through "shared autonomy" or AI copilots [26]. The experimental protocol typically involves:
- Signal Acquisition: EEG signals are captured using scalp electrodes while participants perform specific motor imagery tasks [26].
- Hybrid Decoding: A convolutional neural network (CNN) extracts features from EEG signals, which are then processed by a Kalman filter for continuous control decoding [26].
- AI Assistance: Computer vision and task context are used to infer user goals, with the AI system providing assistance in completing tasks [26].
- Performance Measurement: Success is measured through target acquisition rates and task completion times, with demonstrations showing 3.9-times improvement in target hit rates for participants with paralysis compared to traditional BCI control [26].
This approach acknowledges the inherent bandwidth limitations of non-invasive systems but demonstrates how contextual intelligence can significantly enhance functional performance despite lower raw ITR [26].
BCI Signaling Pathways and System Architecture
The fundamental signaling pathway for BCIs follows a consistent pattern across platforms, though implementation details vary significantly between invasive and non-invasive approaches.
Figure 1: BCI System Architecture and Signaling Pathways. This diagram illustrates the core signal processing pipeline common to all BCIs, with specialized pathways for invasive and non-invasive approaches that explain their performance differences.
The signaling pathway reveals why invasive systems achieve superior performance: direct neural access provides higher signal-to-noise ratios (SNR) and richer neural data, enabling higher ITR [28] [13]. The trade-off involves surgical complexity and long-term biocompatibility requirements. Non-invasive systems sacrifice signal quality for ease of use and safety, resulting in lower ITR but broader potential applicability [11] [45].
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 3: Essential Research Tools for BCI Development and Benchmarking
| Tool/Category | Specific Examples | Function/Purpose | Relevance to Performance Benchmarking |
|---|---|---|---|
| Electrode Technologies | Utah Array (Blackrock); Flexible Threads (Neuralink); Modular Arrays (Paradromics); Stentrode (Synchron) [13] | Neural signal acquisition with varying invasiveness levels | Electrode density and proximity to neurons directly impact signal quality and achievable ITR [28] [13] |
| Signal Processing Frameworks | BCI2000; OpenViBE; NeuroPype [64] | Standardized platforms for signal processing, feature extraction, and classification | Enable reproducible benchmarking and algorithm development [64] |
| Decoding Algorithms | Kalman Filters; Convolutional Neural Networks (CNNs); ReFIT-KF [26] | Translate neural signals into control commands | Algorithm choice directly impacts decoding accuracy and latency [28] [26] |
| AI Copilots | Shared autonomy systems; Goal inference algorithms [26] | Augment BCI control using contextual information | Improve functional performance despite bandwidth limitations [26] |
| Benchmarking Standards | SONIC protocol [28] | Standardized performance measurement | Enable cross-platform comparison of fundamental capabilities [28] |
| Biocompatible Materials | Platinum-iridium contacts; Polyimide substrates [63] [13] | Ensure long-term device stability and safety | Critical for chronic implantation and signal stability over time [13] |
The current landscape of BCI platforms reveals a spectrum of engineering trade-offs between performance, invasiveness, and clinical practicality. Invasive systems like Paradromics Connexus BCI demonstrate the upper limits of achievable performance, with ITRs exceeding 200 bps—sufficient for fluent speech restoration [28]. Less invasive approaches like Synchron's Stentrode offer substantially lower risk profiles but achieve correspondingly lower information transfer rates [28] [13].
The emerging standardization of benchmarking methodologies, particularly the SONIC framework, provides the field with tools for objective comparison and targeted improvement [28]. Simultaneously, innovative approaches like AI-powered shared autonomy demonstrate how systems can optimize functional performance even within the constraints of lower-bandwidth interfaces [26].
For researchers and clinicians, selection of BCI platforms must consider the specific application requirements—where real-time communication demands both high ITR and low latency, more invasive systems currently provide unmatched performance. For less demanding applications or situations where surgical risk is prohibitive, less invasive alternatives offer viable pathways to clinical implementation. As the field progresses through 2025 and beyond, these fundamental benchmarks will continue to provide critical guidance for both engineering development and clinical application.
Brain-Computer Interfaces (BCIs) represent a revolutionary technology that creates a direct communication pathway between the brain and an external device [65]. As of 2025, the field is undergoing a pivotal transition from laboratory demonstrations to regulated clinical trials, marking what industry experts call the "translation era" [14]. This case study analysis objectively compares the performance of leading BCI technologies within the critical framework of invasive versus non-invasive approaches, providing researchers and drug development professionals with comprehensive experimental data and methodologies.
The fundamental distinction in BCI technology lies in the placement of the signal acquisition hardware. Invasive BCIs involve electrodes placed either within brain tissue (intracortical) or on the brain surface (electrocorticography), while non-invasive BCIs typically use scalp-electrodes (electroencephalography - EEG) or other external sensors [11]. This distinction creates a fundamental trade-off: invasive interfaces provide superior signal quality but require neurosurgery and carry associated health risks, whereas non-invasive systems are safer and more accessible but contend with signal attenuation from the skull and scalp [13] [11]. The following analysis examines how leading BCI companies and research institutions are addressing this trade-off through distinct technological approaches currently undergoing clinical validation.
Comparative Performance Analysis of Major BCI Platforms
Quantitative Performance Metrics Across BCI Types
Table 1: Comparative Performance Metrics of Select BCI Platforms in Clinical Trials
| Company/ Platform | BCI Type & Invasiveness | Key Performance Metrics (Reported Outcomes) | Trial Participant Data | Primary Applications Tested |
|---|---|---|---|---|
| Neuralink [13] [14] | Invasive (Intracortical microelectrodes) | 2D cursor control for gaming, online chess, texting [14]. High-bandwidth neural recording. | 5 participants with severe paralysis as of June 2025 [13]. First participant: Noland Arbaugh [14]. | Communication, digital device control for paralysis. |
| Synchron [13] [14] | Minimally Invasive (Endovascular stentrode) | Basic "switch" control for menu navigation, texting with pre-written messages [14]. | 10 volunteers (6 US, 4 Australia) with paralysis [14]. No serious adverse events at 12 months [13]. | Communication for severe paralysis. |
| Precision Neuroscience [13] | Invasive (Cortical surface array) | High-resolution surface signal recording. FDA 510(k) cleared for up to 30 days implantation [13]. | Limited public data on participant numbers. | Communication, focus on ALS patients. |
| UCLA Non-Invasive BCI [16] | Non-Invasive (64-channel EEG cap) | AI copilot improved performance by 3.9x in cursor/robial arm control. Enabled task completion for paralyzed participant. | 3 healthy participants, 1 paraplegic participant (T5 spinal cord injury) [16]. | Robotic arm control, cursor control for paralysis. |
| Paradromics [13] | Invasive (High-channel-count implant) | Ultra-fast data transmission with 421 electrodes. First-in-human recording in 2025. | Initial testing in patient undergoing epilepsy surgery [13]. | Speech restoration (planned focus). |
Table 2: Technical Signal Acquisition Characteristics Across BCI Modalities
| Signal Acquisition Method | Spatial Resolution | Temporal Resolution | Signal-to-Noise Ratio | Typical Information Transfer Rate | Key Limitations |
|---|---|---|---|---|---|
| EEG (Non-Invasive) [11] [65] | Low (centimeters) | High (milliseconds) | Low | 5-25 bits/minute [65] | Signal attenuation by skull, sensitive to artifacts |
| ECoG (Invasive Surface) [65] | Medium (millimeters) | High (milliseconds) | Medium | Not specified in results | Requires craniotomy, limited coverage |
| Microelectrode Arrays (Invasive) [65] | High (micrometers) | High (milliseconds) | Very High | Up to 200+ bits/minute [65] | Tissue response, signal stability over time |
| Endovascular (Minimally Invasive) [13] [14] | Medium to Low | Medium | Medium | Not specified in results | Limited electrode count, signal filtering through vessel wall |
Market Context and Adoption Trajectory
The global BCI market is projected to experience significant growth, with estimates ranging from $2.41 billion in 2025 to $12.11 billion by 2035, representing a compound annual growth rate (CAGR) of 15.8% [66]. Another forecast predicts the overall BCI market will grow to over $1.6 billion by 2045, with a CAGR of 8.4% since 2025 [3]. Currently, non-invasive BCIs dominate the market share due to their wider application range and greater accessibility, particularly in healthcare, gaming, and assistive technology [66]. The medical segment represents the largest end-user market, driven by rising neurological disorders and demand for advanced treatment options [66].
Detailed Experimental Protocols and Methodologies
UCLA Non-Invasive BCI with AI Copilot Protocol
A groundbreaking study from UCLA demonstrates how artificial intelligence can significantly enhance the performance of non-invasive BCIs [16]. The experimental protocol employed a structured approach to overcome the inherent signal-to-noise limitations of EEG-based systems:
- Participant Profile: The study included three healthy participants and one paraplegic participant with a T5-level spinal cord injury who had lost motor control and sensation in the legs and lower body [16].
- Signal Acquisition: Researchers recorded electroencephalogram (EEG) signals using a non-invasive 64-channel cap placed on the scalp according to standardized placement systems [16] [65].
- AI Processing Pipeline: The team developed a hybrid algorithm combining a Convolutional Neural Network (CNN) with a Kalman filter (CNN-KF). The CNN extracted features from the neural data, while the Kalman filter provided recursive estimation to filter noisy time-series data and predict intended movements [16].
- AI Copilot Implementation: Crucially, the system incorporated two AI copilots that worked in closed-loop with the user. Unlike traditional decoders that interpret neural data in isolation, these copilots integrated task structure knowledge, including the locations of potential targets, to update decoder parameters in real-time and modify the distribution of possible actions based on environmental observations [16].
- Task Validation: Participants attempted to control both a computer cursor and a robotic arm using only their neural signals. Performance was quantified by completion accuracy and smoothness of control [16].
- Results: The AI copilot solution improved performance by a factor of 3.9 times for the paralyzed participant in both cursor control and robotic arm tasks. Notably, the researchers reported that the paralyzed patient would not have been able to perform the tasks without AI assistance [16].
Neuralink N1 Implant Human Trial Protocol
Neuralink's approach represents the high-bandwidth end of the invasive BCI spectrum, with a protocol emphasizing dense neural recording:
- Participant Selection: The trial focuses on individuals with severe paralysis, such as those with cervical spinal cord injuries or amyotrophic lateral sclerosis (ALS) [13] [14].
- Surgical Implantation: A coin-sized device called the N1 is implanted in the motor cortex through a cranial opening. The implant contains multiple fine electrode threads (numbering in the thousands) inserted directly into brain tissue using a specialized robotic surgeon to minimize blood vessel damage [13].
- Signal Processing: The device records neural activity associated with movement intention from individual neurons or small neural populations. The high electrode count provides substantial data bandwidth for decoding complex intentions [13].
- Output Generation: The recorded signals are processed through decoding algorithms that translate neural patterns into commands for external devices. The system operates wirelessly, transmitting data to an external computer for processing [13].
- Functional Validation: Performance is assessed through practical tasks such as controlling a computer cursor in two dimensions, clicking interface elements, and playing video games. The first participant, Noland Arbaugh, demonstrated the ability to play games like Civilization and online chess using the interface [14].
Synchron Stentrode Endovascular BCI Protocol
Synchron's methodology offers a minimally invasive alternative that avoids open-brain surgery:
- Participant Profile: Individuals with severe paralysis who require assistive communication technologies but may not be candidates for or willing to undergo more invasive procedures [14].
- Implantation Technique: The Stentrode device is delivered via the vascular system through a catheter inserted into the jugular vein. It is navigated to and lodged in the superior sagittal sinus, a major vein that drains blood from the motor cortex [13].
- Signal Acquisition: Once in position, the stent-expandable electrode array records brain signals through the vessel wall. The approach sacrifices signal resolution for reduced invasiveness and surgical risk [13] [14].
- Decoding Methodology: The system detects characteristic neural patterns associated with specific intentions, providing what the company describes as a basic "switch" control signal rather than continuous multi-dimensional control [14].
- Functional Applications: Participants use the binary control signal to navigate software interfaces through sequential selection, operate communication software for texting, and control other digital interfaces. In trials, users have successfully controlled computers for daily communication tasks [13] [14].
- Safety Outcomes: In a four-patient trial, after 12 months of implantation, none of the patients experienced serious adverse events or blood vessel blockages, and the device remained properly positioned [13].
BCI Signaling Pathways and System Workflows
Core BCI Signal Processing Pathway
The following diagram illustrates the fundamental signal processing pathway shared by most BCI systems, from neural signal generation to device output.
AI-Enhanced BCI Processing Workflow
This diagram details the specific workflow of AI-enhanced BCI systems, such as the UCLA approach, showing how artificial intelligence integrates at multiple points to improve performance.
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Materials and Reagents for BCI Development
| Component Category | Specific Examples | Function in BCI Research |
|---|---|---|
| Electrode Materials [65] | Microelectrode arrays (silicon, platinum, iridium oxide), Flexible polymer substrates (polyimide, parylene-C), Dry electrodes with conductive materials (gold, silver, carbon nanotubes) | Neural signal acquisition; interface between neural tissue and electronics |
| Biocompatible Coatings [65] | PEDOT:PSS, Hydrogels | Improve signal quality and longevity; reduce immune response and tissue encapsulation |
| Signal Processing Hardware [65] | Amplifiers, Analog-to-digital converters, Wireless transmitters (Bluetooth) | Condition weak neural signals, convert to digital format, enable untethered operation |
| Algorithm Development Tools [16] [11] | Convolutional Neural Networks (CNN), Kalman filters, Machine learning libraries (TensorFlow, PyTorch) | Decode neural signals, translate to commands, implement shared autonomy |
| Validation Systems [16] | Robotic arms, Computer cursor control tasks, Communication software | Quantify BCI performance and functional utility in realistic scenarios |
The current clinical trial landscape reveals a clear performance continuum in BCI technologies. On one end, invasive systems like Neuralink's N1 implant offer high-bandwidth control, enabling smooth, two-dimensional cursor movement and complex digital interactions, as demonstrated by participant Noland Arbaugh playing video games [14]. In the middle, minimally invasive approaches like Synchron's Stentrode provide reliable but simpler control paradigms, functioning effectively as a neural "switch" for basic communication tasks [14]. At the other end, non-invasive systems, when enhanced with sophisticated AI copilots as in the UCLA study, can achieve functionally significant control for paralyzed users, albeit with a 3.9x performance improvement required to make tasks feasible [16].
The prevailing trend across all platforms is the integration of artificial intelligence to enhance decoding capabilities and create shared autonomy systems where AI anticipates user intent and corrects errors. As the field progresses through what researchers call the "translation era," the critical challenge remains balancing the superior signal quality of invasive approaches against the safety and accessibility advantages of non-invasive systems [14] [11]. With multiple companies planning pivotal trials in the coming years, the BCI field appears poised to transition from experimental demonstrations to regulated medical products that could substantially improve quality of life for people with severe neurological disabilities [13] [14].
The evaluation of Brain-Computer Interfaces (BCIs) hinges on selecting appropriate metrics that accurately reflect performance, safety, and clinical value. For researchers and drug development professionals, the choice between application-agnostic endpoints (generalized performance metrics) and clinical endpoints (direct measures of patient benefit) represents a critical strategic decision with profound implications for regulatory pathways and commercial translation. This dichotomy is particularly pronounced when comparing invasive versus non-invasive BCI technologies, each presenting distinct trade-offs between signal fidelity, risk profile, and intended use case.
The field of neurotechnology is experiencing accelerated growth, with the global BCI market projected to expand from $2.87 billion in 2024 to $15.14 billion by 2035, representing a compound annual growth rate (CAGR) of 16.32% [30]. This rapid translation from laboratory research to clinical application necessitates rigorous frameworks for evaluating technological performance within regulatory contexts. The U.S. Food and Drug Administration (FDA) defines surrogate endpoints as "a marker that is not itself a direct measurement of clinical benefit," which may predict clinical outcome and support traditional or accelerated approval pathways [67]. Understanding this regulatory landscape is essential for structuring BCI development programs with appropriate endpoints aligned with target indications.
BCI Technology Platforms: Performance Benchmarking
BCI systems can be broadly categorized into invasive and non-invasive approaches, each with characteristic performance specifications, applications, and regulatory considerations. The table below summarizes key technical and clinical parameters for major BCI platforms currently in development.
Table 1: Performance Benchmarks for Select BCI Platforms
| Company/Platform | Technology Approach | Key Performance Metrics | Target Applications | Clinical Trial Status (2025) |
|---|---|---|---|---|
| Neuralink [30] [13] | Invasive cortical implant with thousands of micro-electrodes | High-bandwidth neural recording; Multi-channel signal acquisition | Paralysis, communication restoration | Early human trials with 5 participants |
| Synchron Stentrode [30] [13] | Endovascular electrode array via blood vessels | Signal stability; Safety profile; Device longevity | Paralysis, digital device control | Completed feasibility studies; Preparing pivotal trial |
| Blackrock Neurotech [30] [13] | Implantable Utah array & Neuralace flexible lattice | Typing speed (90 chars/min); Robotic arm control precision | ALS, spinal cord injury, communication | Multiple human implants with long-term data |
| Paradromics Connexus [30] [13] | High-channel-count modular implant | 1,600 channel capacity; High data transmission rates | Speech restoration for paralyzed patients | First-in-human recording in epilepsy surgery |
| Precision Neuroscience Layer 7 [30] [13] | Thin cortical surface array | Minimal tissue damage; High-resolution signal mapping | Communication for ALS patients | FDA 510(k) clearance for up to 30-day implantation |
| Non-invasive EEG with AI [16] | 64-channel cap with CNN-Kalman filter decoding | Performance improvement (3.9x with AI copilot); Task completion accuracy | Cursor control, robotic arm operation for paralyzed users | Proof-of-concept with healthy and paraplegic participants |
Endpoint Selection Framework: Regulatory and Clinical Considerations
Defining Endpoint Categories for BCI Evaluation
The selection of appropriate endpoints requires understanding the distinction between application-agnostic and clinical outcome measures, each serving different purposes in the development lifecycle:
Application-Agnostic Endpoints: These generalized metrics focus on technical performance without direct linkage to specific clinical benefits. Examples include signal-to-noise ratio, spatial and temporal resolution, channel count, information transfer rate (ITR), bit rate, and decoding accuracy. Such endpoints are particularly valuable for early-stage R&D and technical optimization, allowing comparison across diverse BCI platforms irrespective of their intended application [3] [11].
Clinical Endpoints: These patient-focused metrics directly measure how a BCI intervention affects functional ability, quality of life, or disease progression. Examples include typing speed for communication, accuracy of device control, activities of daily living (ADL) scores, functional independence measures, and patient-reported outcomes. For regulatory approval, particularly through traditional pathways, these endpoints must demonstrate direct clinical benefit [67].
Surrogate Endpoints: As defined by the FDA, surrogate endpoints are biomarkers that are "reasonably likely to predict clinical benefit" and may support accelerated approval pathways [67]. In BCI development, examples might include neural signal stability, brain-plasticity markers, or neurophysiological correlates of intended function that precede observable clinical improvement.
Regulatory Pathways and Endpoint Selection
The FDA's Table of Surrogate Endpoints provides valuable guidance for drug development, outlining endpoints that have supported approvals [67]. While BCIs fall under medical device regulation, similar principles apply. The agency recognizes that "the acceptability of these surrogate endpoints for use in a particular drug or biologic development program will be determined on a case-by-case basis" [67], emphasizing the need for early regulatory consultation.
The 21st Century Cures Act mandates publication of surrogate endpoints that have formed the basis of drug approvals, creating a framework that device developers can reference when designing trials [67]. This is particularly relevant for BCIs targeting specific neurological conditions where direct clinical benefit might be difficult to demonstrate in initial studies.
Table 2: Endpoint Selection Framework for BCI Development
| Endpoint Category | Definition & Regulatory Context | BCI-Specific Examples | Appropriate Development Phase |
|---|---|---|---|
| Application-Agnostic | Technical performance metrics independent of specific clinical application | Signal-to-noise ratio; Information transfer rate; Channel count; Spatial/temporal resolution | Early R&D; Proof-of-concept; Technical benchmarking |
| Surrogate Endpoints | Biomarkers reasonably likely to predict clinical benefit (FDA Accelerated Approval) | Neural decoding accuracy; Pattern stability; Neuroplasticity markers; Functional connectivity changes | Pivotal trials for accelerated approval; Preliminary efficacy studies |
| Clinical Endpoints | Direct measurements of how a patient feels, functions, or survives (FDA Traditional Approval) | Communication rate (words/min); Limb control accuracy; ADL improvement; Quality of life measures | Pivotal trials for traditional approval; Post-market studies |
Experimental Approaches for Endpoint Validation
Protocol for Evaluating BCI Performance with AI Enhancement
Recent research demonstrates how structured experimental protocols can validate both application-agnostic and functional endpoints. A September 2025 study from UCLA established a methodology for quantifying AI enhancement of non-invasive BCI performance [16]:
- Signal Acquisition: Researchers recorded electroencephalogram (EEG) signals using a 64-channel cap from three healthy participants and one paraplegic participant with T5-level spinal cord injury.
- AI Processing Pipeline: Implemented a convolutional neural network-Kalman filter (CNN-KF) architecture to decode neural signals. The CNN extracted spatial features from multichannel EEG data, while the Kalman filter provided recursive estimation to track intended movements from noisy time-series data.
- Task Paradigm: Participants performed cursor control and robotic arm manipulation tasks using thought alone, with and without AI copilot assistance.
- Endpoint Measurement: The team quantified both application-agnostic metrics (signal quality, decoding latency) and functional metrics (task completion accuracy, speed). The AI copilot improved performance by a factor of 3.9× for the paralyzed participant, enabling task completion that was impossible without AI assistance [16].
Clinical Trial Designs for BCI Endpoint Validation
Innovative trial methodologies are essential for evaluating BCIs, particularly for technologies with tissue-agnostic or application-agnostic potential:
Basket Trials: These investigate a single targeted therapy across multiple diseases sharing a common biomarker. This design has supported tissue-agnostic drug approvals and could be adapted for BCIs targeting specific neurological deficits across different etiologies [68] [69].
Umbrella Trials: These evaluate multiple targeted therapies for a single disease type, stratified by biomarkers. For BCIs, this could involve testing different interface technologies (invasive vs. non-invasive) for specific conditions like ALS or spinal cord injury [68].
Platform Trials: These allow for flexible, ongoing evaluation of multiple interventions, with therapies entering or leaving the platform based on predefined performance criteria. This adaptive design could efficiently compare application-agnostic versus clinical endpoints for BCI validation [68].
Decision Framework for Endpoint Selection
The following diagram illustrates the strategic decision process for selecting appropriate endpoints in BCI development, incorporating both technical and regulatory considerations:
Diagram 1: Endpoint Selection Framework for BCI Development
Research Reagent Solutions for BCI Endpoint Validation
The experimental validation of BCI endpoints requires specialized tools and methodologies. The following table details essential research reagents and their applications in benchmarking BCI performance:
Table 3: Essential Research Materials for BCI Endpoint Validation
| Research Tool Category | Specific Examples | Function in Endpoint Validation |
|---|---|---|
| Signal Acquisition Systems | 64-channel EEG caps [16]; Utah & Neuralace electrode arrays [30] [13]; fNIRS headsets [3] | Capture neural signals with varying spatial/temporal resolution for signal quality metrics |
| Decoding Algorithms | Convolutional Neural Networks (CNN) [16]; Kalman Filters [16]; Machine Learning Classifiers | Translate neural signals into commands for decoding accuracy measurements |
| AI Copilot Systems | Shared autonomy algorithms [16]; Context-aware interpreters | Enhance BCI performance for functional endpoint achievement |
| Output Control Devices | Robotic arms [16]; Computer cursors [16]; Speech synthesizers | Enable quantification of functional performance metrics |
| Clinical Assessment Tools | Functional Independence Measure (FIM); Communication speed tests; Quality of life inventories | Provide validated clinical endpoints for regulatory submissions |
| Data Acquisition Software | OpenBCI software [70]; Custom signal processing pipelines | Facilitate collection of application-agnostic performance data |
The selection between application-agnostic and clinical endpoints represents a strategic balance between technical optimization and clinical validation in BCI development. For invasive BCIs targeting severe disabilities, the regulatory pathway increasingly requires demonstration of meaningful clinical benefit through functional endpoints. Conversely, non-invasive approaches may initially leverage application-agnostic metrics for early-stage development, with progression toward surrogate and ultimately clinical endpoints as technologies mature.
The evolving regulatory landscape, particularly frameworks for surrogate endpoints and tissue-agnostic therapies, provides valuable precedents for BCI validation strategies [67] [69]. As the field advances toward broader clinical application, the rigorous implementation of appropriate endpoint selection frameworks will be essential for translating technical innovation into genuine patient benefit. Future development will likely see increased standardization of both application-agnostic benchmarking and clinical outcome assessment, enabling more direct comparison across diverse BCI platforms and accelerating the translation of neurotechnology from laboratory research to clinical practice.
Conclusion
The benchmarking of BCI performance reveals a field defined by a fundamental compromise: invasive interfaces offer unparalleled signal fidelity and information rates, crucial for complex tasks like speech decoding and dexterous robotic control, while non-invasive methods provide a safer, more accessible pathway for applications where lower ITR is acceptable. The emergence of standardized benchmarking frameworks, such as the SONIC protocol, is a critical step toward transparent and objective comparison across this diverse technological landscape. For biomedical researchers and clinicians, the choice of technology must be guided by a careful balance of performance requirements, safety profile, and long-term viability. Future progress will be driven by advancements in materials science to improve biocompatibility of invasive devices, sophisticated AI to decode noisy non-invasive signals, and the maturation of minimally invasive techniques that aim to bridge the current performance gap. The ongoing clinical trials and engineering breakthroughs promise not only to restore lost neurological functions but also to redefine the boundaries of human-computer interaction.
References
- 1. How does invasive vs non-invasive BCI compare? [https://synapse.patsnap.com/article/how-does-invasive-vs-non-invasive-bci-compare]
- 2. Non-Invasive Brain-Computer Interfaces: State of the Art and Trends - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11861396/]
- 3. Brain Computer Interfaces 2025-2045: Technologies, ... [https://www.idtechex.com/en/research-report/brain-computer-interfaces/1024]
- 4. 5-Year Follow-up of a Fully Implanted Brain-Computer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12600028/]
- 5. Improving brain-computer interface performance with ... [https://www.sciencedirect.com/science/article/abs/pii/S1350453325001110]
- 6. Outlook for Noninvasive Brain-Computer Interfaces for AI [https://etcjournal.com/2025/09/21/outlook-for-noninvasive-brain-computer-interfaces-for-ai/]
- 7. Comprehensive evaluation methods for translating BCI into ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2024.1429130/full]
- 8. Human visual skills for brain computer interface use: A tutorial [https://pmc.ncbi.nlm.nih.gov/articles/PMC7529781/]
- 9. Performance Measurement for Brain-Computer or ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4185283/]
- 10. Signal acquisition of brain–computer interfaces: A medical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11955058/]
- 11. The history, current state and future possibilities of the non- ... [https://www.sciencedirect.com/science/article/pii/S2590093525000049]
- 12. Defining Surgical Terminology and Risk for Brain ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2021.599549/full]
- 13. BCIs in 2025: Trials, Progress, and Challenges [https://andersenlab.com/blueprint/bci-challenges-and-opportunities]
- 14. Brain-computer interfaces face a critical test [https://www.technologyreview.com/2025/04/01/1114009/brain-computer-interfaces-10-breakthrough-technologies-2025/]
- 15. A Review of Brain-Computer Interface Technologies [https://arxiv.org/html/2503.16471v1]
- 16. AI Copilot Boosts Brain-Computer Interface's Performance [https://www.psychologytoday.com/us/blog/the-future-brain/202509/ai-copilot-boosts-brain-computer-interfaces-performance]
- 17. Spatial and temporal resolutions of EEG - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC4548479/]
- 18. Signal-to-noise ratio [https://en.wikipedia.org/wiki/Signal-to-noise_ratio]
- 19. Spatial resolution, signal-to-noise and information capacity ... [https://opg.optica.org/oe/abstract.cfm?uri=oe-24-15-17168]
- 20. Temporal vs. spatial resolution and Consumer Neuroscience [https://medium.com/@Pedro_R_Almeida/temporal-vs-spatial-resolution-and-consumer-neuroscience-ca6b360c5890]
- 21. A Hybrid Brain-Computer Interface Based on Visual ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8850273/]
- 22. Toward the integration of mixed reality and brain-computer ... [https://www.sciencedirect.com/science/article/pii/S0208521625000518]
- 23. A high performance heterogeneous hardware architecture ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11703782/]
- 24. Advancing BCI with a transformer-based model for motor ... [https://www.nature.com/articles/s41598-025-06364-4]
- 25. A time-frequency feature fusion-based deep learning ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1679451/full]
- 26. Brain–computer interface control with artificial intelligence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12599856/]
- 27. Brain-computer interface restores natural speech after ... [https://www.nih.gov/news-events/nih-research-matters/brain-computer-interface-restores-natural-speech-after-paralysis]
- 28. Think Fast | Setting new standards and new records for ... [https://www.paradromics.com/blog/bci-benchmarking]
- 29. EEG-based brain-computer interface enables real-time ... [https://www.nature.com/articles/s41467-025-61064-x]
- 30. Top 10 Brain-Computer Interface Companies in 2025 [https://www.sphericalinsights.com/blogs/top-10-companies-leading-the-brain-computer-interface-market-in-2025-key-players-statistics-future-trends-2024-2035]
- 31. A brain–computer-interface driven forearm exoskeleton ... [https://www.sciencedirect.com/science/article/pii/S1110016825010324]
- 32. Study of promising speech-enabling interface offers hope for ... [https://med.stanford.edu/news/all-news/2025/08/brain-computer-interface.html]
- 33. Towards decoding individual words from non-invasive ... [https://www.nature.com/articles/s41467-025-65499-0]
- 34. Modulating Brain Activity with Invasive Brain–Computer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9856340/]
- 35. A brain implant that could rival Neuralink's enters clinical ... [https://www.nature.com/articles/d41586-025-03849-0]
- 36. The Best Brain Computer Interface Kit: A Guide [https://www.emotiv.com/blogs/news/brain-computer-interface-kit-guide?srsltid=AfmBOopu6ih8FmHv_xc8ERHceD6sYgXl4R9GKUKDrJWBrrDLXt5BKybO]
- 37. A Comprehensive Survey of Brain–Computer Interface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12180781/]
- 38. A Non-Invasive Brain-Computer Interface for Real-Time ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5906041/]
- 39. Brain–computer interfaces: the innovative key to unlocking ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11392146/]
- 40. Invasive vs. Non-Invasive Neuronal Signals for Brain- ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2016.00295/full]
- 41. AI Co-Pilot Boosts Noninvasive Brain-Computer Interface ... [https://samueli.ucla.edu/ai-co-pilot-boosts-noninvasive-brain-computer-interface-by-interpreting-user-intent/]
- 42. Material and design strategies for chronically-implantable ... [https://www.nature.com/articles/s41427-025-00613-8]
- 43. Autonomous Implants - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12617072/]
- 44. Engineering aspects and materials for next generation ... [https://www.sciencedirect.com/science/article/pii/S1877117325001279]
- 45. The impact of non-invasive brain-computer interface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12642192/]
- 46. Flexible brain electronic sensors advance wearable ... [https://www.nature.com/articles/s44385-025-00029-7]
- 47. Noise To Signal: Cuban Backs Synaptrix Labs Non- ... [https://www.forbes.com/sites/naveenrao/2025/10/24/noise-to-signal-cuban-backs-synaptrix-labs-non-invasive-bci-platform/]
- 48. Advancing Brain-Computer Interface Closed-Loop Systems for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12588595/]
- 49. A novel channel reduction concept to enhance the ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0335511]
- 50. Enhanced EEG signal classification in brain computer ... [https://www.nature.com/articles/s41598-025-07427-2]
- 51. Non-Invasive Brain-Computer Interface Trial Balances ... [https://www.clinicalleader.com/doc/non-invasive-brain-computer-interface-trial-balances-regulatory-requirements-with-patient-needs-0001]
- 52. Comparison metrics and power trade-offs for BCI motor ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1547074/full]
- 53. A Modified Hybrid Brain-Computer Interface Speller Based ... [https://www.imrpress.com/journal/JIN/23/4/10.31083/j.jin2304073/htm]
- 54. Reevaluating performance in c-VEP BCIs: The impact of ... [https://www.sciencedirect.com/science/article/pii/S0208521625000816]
- 55. Advances in endovascular brain computer interface [https://www.sciencedirect.com/science/article/pii/S0165027025001128]
- 56. minimally invasive brain–computer interface by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11671944/]
- 57. Signal quality of simultaneously recorded endovascular, ... [https://www.nature.com/articles/s41598-018-26457-7]
- 58. Comparative outcomes of endovascular vs. surgical ... [https://www.sciencedirect.com/science/article/pii/S0022510X2500019X]
- 59. Review of Performance Improvement of a Noninvasive Brain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10984355/]
- 60. Comprehensive evaluation methods for translating BCI into ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11188342/]
- 61. Visualization and workload with implicit fNIRS-based BCI [https://www.frontiersin.org/journals/neuroergonomics/articles/10.3389/fnrgo.2025.1550629/full]
- 62. Development and evaluation of a non-invasive brain-spine ... [https://jneuroengrehab.biomedcentral.com/articles/10.1186/s12984-025-01628-6]
- 63. Neuralink's 2025 Speech Implant Trial - Applying AI [https://applyingai.com/2025/09/neuralinks-2025-speech-implant-trial-a-business-focused-deep-dive/]
- 64. Neural–Computer Interfaces: Theory, Practice, Perspectives [https://www.mdpi.com/2076-3417/15/16/8900]
- 65. BCI Research Documentation Standards for 2025 [https://editverse.com/bci-research-documentation-standards-for-2025/]
- 66. Brain Computer Interface (BCI) Research Report 2025-2035: [https://www.globenewswire.com/news-release/2025/10/01/3159276/0/en/Brain-Computer-Interface-BCI-Research-Report-2025-2035-EEG-MEG-and-fMRI-Integration-Enhances-Neural-Signal-Detection-and-Drives-Global-Growth.html]
- 67. Table of Surrogate Endpoints That Were the Basis of Drug ... [https://www.fda.gov/drugs/development-resources/table-surrogate-endpoints-were-basis-drug-approval-or-licensure]
- 68. Tumor-Agnostic Therapies in Practice - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC11899253/]
- 69. Real-world evidence provides clinical insights into tissue ... [https://www.nature.com/articles/s41467-025-57941-0]
- 70. Non Invasive BCI Market Report 2025 (Global Edition) [https://www.cognitivemarketresearch.com/non-invasive-bci-market-report]