Advanced EEG Artifact Removal and Noise Filtering: A Comprehensive Guide for Biomedical Research and Drug Development
This article provides a comprehensive overview of modern electroencephalogram (EEG) artifact removal and noise filtering techniques, tailored for researchers and drug development professionals.
Advanced EEG Artifact Removal and Noise Filtering: A Comprehensive Guide for Biomedical Research and Drug Development
Abstract
This article provides a comprehensive overview of modern electroencephalogram (EEG) artifact removal and noise filtering techniques, tailored for researchers and drug development professionals. It covers the foundational knowledge of physiological and technical artifacts that contaminate neural signals, explores a wide array of methodological approaches from traditional algorithms to advanced deep learning models, addresses practical troubleshooting and optimization strategies for real-world data quality challenges, and presents rigorous validation frameworks for comparative performance analysis. The content synthesizes current research to offer practical guidance for improving EEG signal integrity in clinical trials, pharmacodynamic modeling, and neuroscience research, enabling more reliable data interpretation and analysis.
Understanding EEG Artifacts: Sources, Characteristics, and Impact on Neural Signal Interpretation
FAQ: Understanding EEG Artifacts
Q: What is an EEG artifact? An EEG artifact is any recorded signal that does not originate from neural activity within the brain. These unwanted signals contaminate the neurophysiological data and can obscure or mimic genuine brain activity, complicating analysis and interpretation [1].
Q: Why is artifact removal critically important in EEG research? Artifacts can significantly reduce the signal-to-noise ratio of EEG recordings, potentially leading to misinterpretation of brain signals [1]. In clinical settings, this can result in misdiagnosis, such as confusing artifacts with epileptiform activity [1]. Furthermore, improper cleaning techniques can artificially inflate effect sizes in event-related potentials and functional connectivity analyses, while also biasing source localization estimates [2].
Q: What are the main categories of EEG artifacts? EEG artifacts are broadly classified into two categories based on their origin [1]:
- Physiological Artifacts: Originate from the patient's own body (e.g., ocular activity, muscle movement, cardiac activity).
- Non-Physiological Artifacts: Originate from the external environment or equipment (e.g., power line interference, electrode issues, cable movement).
Q: Which artifact removal techniques are considered state-of-the-art? Modern artifact management leverages both traditional and advanced computational methods. Independent Component Analysis (ICA) is a widely used traditional technique [2] [1]. Recently, deep learning models have shown remarkable performance, including transformer-based architectures like the Artifact Removal Transformer (ART) [3] and hybrid models like CLEnet, which combines convolutional neural networks (CNN) with Long Short-Term Memory networks (LSTM) to extract both morphological and temporal features from EEG data [4].
Q: How do wearable EEG systems pose specific challenges for artifact management? Wearable EEG systems often use dry electrodes, have reduced scalp coverage (typically fewer than 16 channels), and are used in mobile, real-world conditions. These factors make the signals more susceptible to specific artifacts, particularly motion-related ones, and limit the effectiveness of traditional source separation methods like ICA that perform better with high-density electrode arrays [5].
Troubleshooting Guide: Common Artifact Problems and Solutions
Table: Identifying and Resolving Common EEG Artifact Issues
| Problem Symptom | Potential Artifact Type | Recommended Solution |
|---|---|---|
| Slow, large deflections in frontal channels | Ocular (EOG) - from blinks or eye movements [1] | Apply ICA or use a targeted approach that cleans only the artifact periods of eye movement components [2]. |
| High-frequency, "spiky" noise across multiple channels | Muscle (EMG) - from jaw clenching, talking, or facial movements [1] | Apply ICA or frequency-targeted removal. For deep learning, use models like CLEnet effective against EMG [4]. |
| Rhythmic, sharp waveforms synchronized with pulse | Cardiac (ECG) - from heartbeats [1] | Use canonical correlation analysis (CCA) or other BSS methods. Models like CLEnet have also shown efficacy in removing ECG artifacts [4]. |
| 50 Hz or 60 Hz sinusoidal noise across all channels | Power Line Interference [1] | Apply a notch filter at the specific frequency (50/60 Hz). Ensure proper grounding and shielding of equipment. |
| Sudden, large-amplitude spikes in a single channel | Electrode Pop from poor contact [1] | Check impedance for the affected electrode and reapply if necessary. The signal segment can often be rejected or interpolated. |
| Signal drift or slow shifts | Perspiration or Respiration [1] | Use high-pass filtering at a suitable cutoff (e.g., 0.5 Hz). For deep learning, train models on data containing these artifacts. |
Technical Specifications and Performance Data
Table: Quantitative Performance Comparison of Advanced Artifact Removal Models
| Model Name | Architecture Type | Key Advantage | Reported Performance Metrics |
|---|---|---|---|
| CLEnet [4] | Dual-scale CNN + LSTM with attention | Effectively removes multiple artifact types (EMG, EOG, ECG) from multi-channel EEG. | SNR: 11.498 dB (mixed artifacts)CC: 0.925RRMSEt: 0.300 |
| ART (Artifact Removal Transformer) [3] | Transformer | End-to-end denoising; captures millisecond-scale EEG dynamics; outperforms other DL models. | Surpasses other deep-learning models in signal reconstruction (MSE, SNR) and improves BCI performance. |
| AnEEG [6] | LSTM-based GAN | Adversarial training helps generate artifact-free signals that maintain original neural information. | Lower NMSE/RMSE and higher CC, SNR, and SAR values compared to wavelet techniques. |
| RELAX [2] | Enhanced ICA | Targeted cleaning reduces effect size inflation and source localization bias vs. full component rejection. | Effectively cleans artifacts while better preserving neural signals and minimizing analytical biases. |
Experimental Protocols for Artifact Management
Protocol 1: Independent Component Analysis (ICA) with Targeted Cleaning
This protocol refines the standard ICA workflow to minimize the unintended removal of neural data [2].
- Data Preprocessing: Filter the continuous EEG data (e.g., 1-100 Hz bandpass) and apply a notch filter (50/60 Hz). Segment data into epochs if needed.
- ICA Decomposition: Run ICA (e.g., using EEGLAB) to decompose the EEG signal into statistically independent components.
- Component Classification: Use automated tools like ICLabel to classify components as brain or artifact (ocular, muscle, etc.).
- Targeted Removal: Instead of subtracting entire artifact components, apply a method like the RELAX pipeline, which:
- For ocular components: Removes activity only during the specific periods of eye movements or blinks.
- For muscle components: Removes activity only in the high-frequency bands where the artifact dominates.
- Signal Reconstruction: Reconstruct the EEG signal from the processed components.
Protocol 2: Deep Learning-Based Removal with CLEnet
This protocol outlines an end-to-end deep learning approach for robust, multi-artifact removal [4].
- Data Preparation: Prepare a dataset of paired noisy-clean EEG signals. This can be a semi-synthetic dataset (clean EEG artificially contaminated with EOG/EMG) or a real dataset cleaned with a trusted method.
- Model Training:
- Architecture: Use the CLEnet model, which features a dual-branch design.
- Branch 1 (Morphological Features): Employs dual-scale convolutional kernels and an EMA-1D attention module to extract spatial features at different scales.
- Branch 2 (Temporal Features): The extracted features are passed through LSTM layers to capture long-term temporal dependencies in the EEG.
- Loss Function: Train the model using Mean Squared Error (MSE) between the output and the clean reference signal.
- Model Application: Feed novel, artifact-contaminated EEG data into the trained CLEnet model to generate the cleaned output.
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Computational Tools and Datasets for EEG Artifact Research
| Tool / Resource | Type | Function and Application |
|---|---|---|
| EEGLAB | Software Plugin | A collaborative, open-source signal processing environment for EEG data; provides the foundation for running ICA and hosting other plugins like RELAX [2]. |
| RELAX Pipeline | Software Plugin | An EEGLAB plugin that implements the targeted artifact reduction protocol to minimize neural signal loss and analytical bias [2]. |
| EEGdenoiseNet | Benchmark Dataset | A semi-synthetic dataset providing clean EEG segments and recorded EMG/EOG artifacts, enabling standardized training and testing of artifact removal algorithms [4]. |
| HBN-EEG Dataset | Large-Scale Dataset | A large, publicly available dataset with over 3,000 participants, useful for training complex deep learning models and evaluating cross-task generalization [7]. |
| Transformer Architectures | Algorithm | Deep learning models (e.g., ART) that use self-attention mechanisms to capture global temporal dependencies in EEG signals, leading to state-of-the-art denoising performance [3]. |
| Ensemble Kalman Filter (EnKF) | Algorithm | A data assimilation method used for tracking time-varying changes in cortical excitation-inhibition balance from EEG, demonstrating the value of clean signals for neurophysiological insight [8]. |
Electroencephalography (EEG) is designed to record cerebral activity, but it also captures electrical activities arising from other sites in the body. These unwanted signals, known as physiological artifacts, can significantly obscure genuine brain signals and compromise data interpretation [9] [1]. Physiological artifacts originate from the patient's own body and include activities such as eye movements, muscle contractions, cardiac activity, and perspiration [9] [10]. Because EEG signals are typically measured in microvolts, they are exceptionally susceptible to these sources of contamination, which often have much larger amplitudes than neural signals [1]. Proper identification and removal of these artifacts is therefore crucial for accurate analysis in both clinical and research settings, including drug development studies where precise neurophysiological data is paramount.
Comprehensive Artifact Classification and Characteristics
The table below provides a detailed overview of the primary physiological artifacts, including their origins, temporal and spectral characteristics, and topographical distribution on the scalp.
Table 1: Characteristics of Major Physiological Artifacts in EEG
| Artifact Type | Biological Origin | Typical Morphology & Time-Domain Effect | Frequency-Domain Effect | Primary Electrode Distribution |
|---|---|---|---|---|
| Ocular (EOG) | Corneo-retinal dipole (eye as an electrical dipole); Eyelid movement [9] [1] [11] | High-amplitude, slow deflections (up to 100-200 µV) [1] [11] | Dominates delta (0.5-4 Hz) and theta (4-8 Hz) bands [1] | Frontal electrodes (Fp1, Fp2, F7, F8) [9] [1] |
| Muscle (EMG) | Contraction of head, face, neck, or jaw muscles [9] [1] | High-frequency, sharp, irregular waveforms [9] | Broadband noise, dominates beta (13-30 Hz) and gamma (>30 Hz) bands [1] | Temporal electrodes, widespread depending on muscle group [9] |
| Cardiac (ECG/Pulse) | Electrical activity of the heart (ECG) or pulsation of scalp vessels [9] [1] | Rhythmic, sharp transients synchronized with heartbeat; Pulse artifact has ~200-300 ms delay after QRS complex [9] | Overlaps multiple EEG bands; sharp peak at heart rate frequency | Central, temporal electrodes; depends on individual anatomy [9] [1] |
| Perspiration (Sweat) | Electrochemical changes from sweat glands altering skin-electrode impedance [9] [1] | Very slow baseline drifts and sways [9] [1] | Contaminates very low frequencies (delta band) [1] | Widespread, often most prominent in electrodes with poor adhesion |
Detailed Artifact Profiles
Ocular Artifacts
The eyeball functions as a dipole with a positive cornea and a negative retina [9]. When the eye moves or blinks, this dipole rotates, generating a large electrical field that is easily detected by frontal EEG electrodes [9] [1]. Blinks typically produce symmetric, high-amplitude slow waves frontally, while lateral eye movements create opposing polarities at electrodes F7 and F8 [9]. A special type of ocular artifact, the glossokinetic artifact, originates from the tongue (which also acts as a dipole) and produces broad, delta-range potentials that are maximal inferiorly [9].
Muscle Artifacts (EMG)
Myogenic potentials are among the most common EEG artifacts [9]. They are generated by the contraction of skeletal muscles, particularly the frontalis and temporalis muscles from jaw clenching, frowning, or talking [9] [1]. EMG artifacts are characterized by their high-frequency, sharp morphology, which can sometimes mimic cerebral activity, such as the rhythmic 4-6 Hz sinusoidal artifacts seen in essential tremor or Parkinson's disease [9].
Cardiac Artifacts
Cardiac artifacts manifest in two primary forms: the ECG artifact, which is the direct pickup of the heart's electrical signal, and the pulse artifact, which is a mechanically induced waveform caused by the pulsation of scalp arteries under an electrode [9] [1]. The pulse artifact occurs with a slight delay (200-300 milliseconds) after the QRS complex of the ECG [9]. In simultaneous EEG-fMRI recordings, the pulse artifact (PA) is a significant concern, with research indicating that cardiac-pulse-driven head rotation is its dominant source [12].
Perspiration and Other Artifacts
Sweat artifacts are caused by the interaction of sweat (sodium chloride and lactic acid) with the metal of the electrodes, resulting in very slow baseline drifts that can obscure underlying brain activity [9] [1]. Respiration can also cause rhythmic, slow-wave artifacts synchronous with breathing, often due to body movement or impedance changes [9] [13].
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: Why can't I just discard EEG segments that contain large artifacts? While discarding data is sometimes necessary, it is often not feasible because physiological artifacts like eye blinks occur frequently (12-18 times per minute) [11]. Removing all contaminated segments would result in a significant and unacceptable loss of neurophysiological data, especially for event-related potential (ERP) studies where time-locked signals are critical.
Q2: An electrode is showing a persistent "popping" artifact. What should I do? Electrode "pops" are caused by abrupt changes in impedance at the skin-electrode interface [9] [1]. To troubleshoot:
- Inspect and Reapply: Gently adjust the offending electrode and check for dryness. Reapply conductive gel if using wet electrodes and ensure stable contact [13] [14].
- Check Impedance: Verify that the electrode impedance is low and stable (typically below 20 kΩ) [13].
- Swap Electrodes: If the problem persists, replace the electrode to rule out a hardware fault [14].
Q3: I have followed all setup procedures, but my reference electrode impedance remains high. What could be wrong? This is a complex issue. Follow a systematic approach to isolate the problem [14]:
- Check the Ground: A faulty ground (GND) electrode can affect all channels, including the reference. Reapply the ground electrode and try alternative placements (e.g., hand, sternum) [14].
- Check the Entire Chain: Systematically verify the recording software, computer, amplifier, headbox, and electrode cap connections. Try swapping components like the headbox to isolate the fault [14].
- Participant-Specific Factors: Rarely, individual factors like a participant's skin type or static electricity can cause "oversaturation" of the reference. Using a different ground location (e.g., the experimenter's hand) can be a temporary workaround to confirm the issue [14].
Experimental Protocol for Artifact Handling
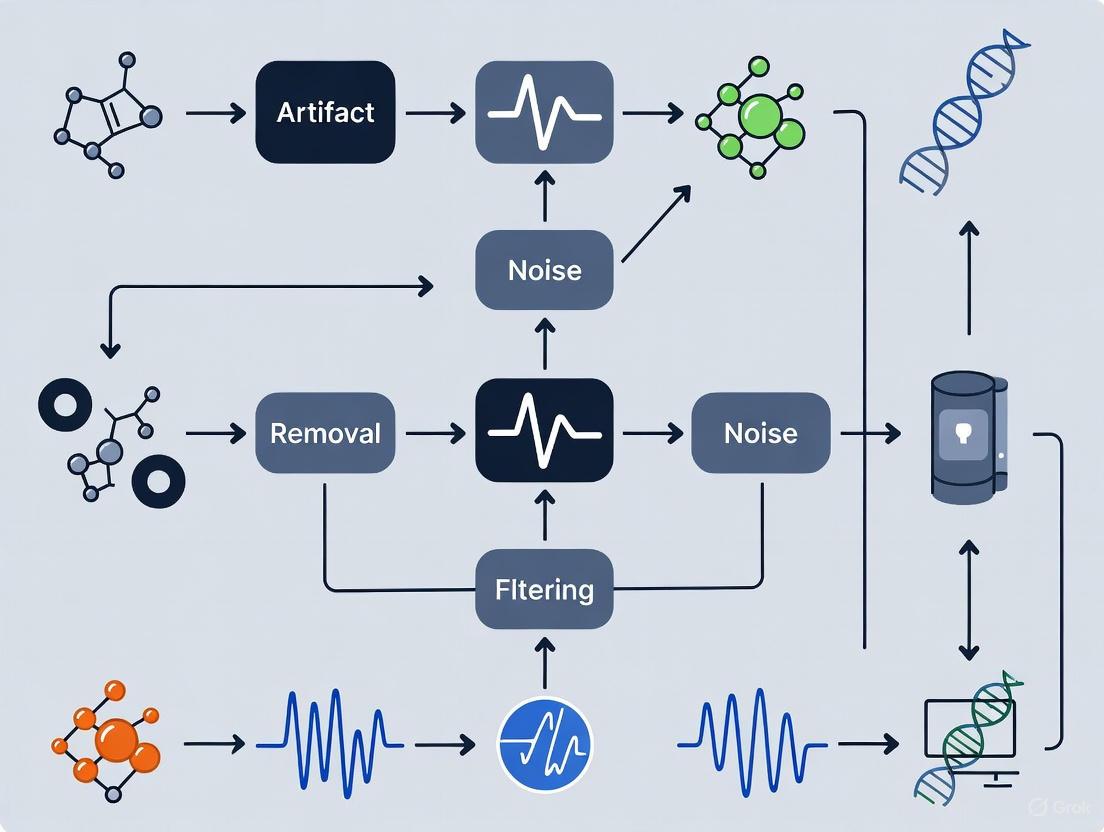
The following workflow provides a systematic guide for managing physiological artifacts during EEG experimental design and data collection.
Diagram 1: Artifact management workflow.
The Scientist's Toolkit: Research Reagents & Solutions
Table 2: Essential Tools for Physiological Artifact Management
| Tool / Reagent | Type | Primary Function in Artifact Handling |
|---|---|---|
| High-Density EEG Cap (e.g., 64+ channels) | Hardware | Enables the use of spatial filtering techniques (e.g., ICA) for effective artifact separation [11]. |
| Electrode Conductive Gel/Paste | Material | Ensures stable, low-impedance connection between electrode and scalp, minimizing electrode pop and movement artifacts [13] [14]. |
| Electrooculogram (EOG) Electrodes | Hardware | Placed near the eyes to record a reference signal of ocular activity, which can be used for regression-based removal methods [11]. |
| Electrocardiogram (ECG) Sensor | Hardware | Provides a reference channel for heartbeats, aiding in the identification and removal of cardiac artifacts [9]. |
| EEGLab (with ICA and ASR) | Software | A widely used software environment that provides implementations of Independent Component Analysis and Artifact Subspace Reconstruction for artifact removal [13] [11]. |
| Explorepy API / Impedance Checker | Software | Allows for real-time verification of electrode impedance during setup, which is critical for preventing technical artifacts [13]. |
| Deep Learning Models (e.g., CNN, SSM) | Software | Advanced methods, such as Convolutional Neural Networks and State Space Models, show high efficacy in removing complex artifacts, including those from transcranial electrical stimulation [15] [16]. |
Advanced Methodologies for Artifact Removal
Protocol for Ocular Artifact Removal using Regression
Regression-based methods are foundational for correcting ocular artifacts [11]. The following protocol is based on the Gratton and Cole algorithm:
- Data Acquisition: Record EEG alongside a dedicated EOG channel or use frontal EEG channels (e.g., Fp1, Fp2) as a surrogate EOG template.
- Filtering: Apply a band-pass filter (e.g., 1-50 Hz) to the raw EEG to remove slow drifts and high-frequency noise. Low-pass filter the EOG signal (cut-off at 15 Hz) to eliminate high-frequency components not related to the blink [11].
- Calibration and Coefficient Estimation: Use a segment of data containing spontaneous blinks to estimate the regression coefficient (( \beta_{ei} )) for each EEG electrode. This coefficient represents the degree to which the EOG signal influences that specific channel [11].
- Artifact Subtraction: For the main EEG data, subtract the EOG signal, scaled by the calculated ( \beta_{ei} ) for each channel, from the corresponding EEG channel [11].
Corrected_EEG_{ei}(n) = Raw_EEG_{ei}(n) - β_{ei} * EOG(n)
Protocol for Generalized Artifact Removal using Independent Component Analysis (ICA)
ICA is a blind source separation technique highly effective for multi-channel EEG data [13] [1] [11].
- Prerequisites: Ensure you have a high-density EEG recording (typically >40 channels is recommended for best results) [11].
- Preprocessing: Band-pass filter the data and optionally re-reference to a common average reference.
- Decomposition: ICA algorithmically decomposes the multichannel EEG data into a set of independent components (ICs). Each IC has a fixed scalp topography and a time course of activation [13].
- Component Identification: Visually or automatically classify ICs as "brain" or "non-brain" based on their topography, time course, and frequency spectrum. For example:
- Ocular ICs: Have strong, frontal topographies and large, low-frequency time-course deflections corresponding to blinks [1].
- Muscle ICs: Exhibit high-frequency bursts in their time course and topographies focused over temporal muscles [1].
- Cardiac ICs: Show a rhythmic time course locked to the heartbeat [1].
- Reconstruction: Remove the artifact-related ICs and project the remaining "clean" components back to the sensor space to obtain the artifact-corrected EEG.
Emerging Techniques: Deep Learning and State Space Models
Recent research has demonstrated the power of deep learning models for artifact removal. For instance, one study benchmarked eleven methods and found that performance is highly stimulation-dependent [15] [16]. Complex Convolutional Neural Networks (CNNs) excelled at removing tDCS artifacts, while multi-modular networks based on State Space Models (SSMs) were most effective for the more complex tACS and tRNS artifacts [15] [16]. These data-driven approaches represent the cutting edge in artifact cleaning technology.
Troubleshooting Guides
Frequently Asked Questions (FAQs)
Q1: What does an "electrode pop" look like on my EEG recording, and what causes it?
An electrode pop artifact appears as a sudden, sharp, high-amplitude deflection with a very steep upslope in a single electrode channel, often with little to no spread to surrounding channels [17] [18] [19]. It is typically caused by a loose electrode, inadequate application with poor skin contact, drying of the conductive gel, a dirty electrode, or physical pressure or pull on the electrode [17] [20]. This sudden shift occurs due to a momentary change in the electrical contact between the electrode and the scalp [19].
Q2: How can I prevent or eliminate electrode pop artifacts during my experiment?
To prevent and eliminate electrode pops:
- Ensure Secure Application: Before recording, ensure all electrodes are fixed tightly to the scalp and make good contact [19].
- Check Impedance: Verify electrode impedances are low and stable before starting the measurement [13] [19].
- Re-apply Electrodes: If a pop occurs, the most direct solution is to change or re-apply the problematic electrode [17].
- Re-reference Signals: As an offline processing step, you can re-reference the affected EEG leads to a different, stable reference (e.g., from M1 to M2) to eliminate the artifact from the signal [17].
Q3: My EEG data shows high-frequency, monotonous noise. Is this AC interference, and how do I remove it?
Yes, high-frequency, monotonous noise at 50 Hz or 60 Hz is characteristic of AC power line interference [20] [19]. This environmental artifact originates from electromagnetic fields emitted by power lines and electronic equipment in the vicinity [13].
- Prevention: Use an electromagnetically isolated room or a Faraday cage if available [13]. Employ EEG systems with active shielding on cables, which minimizes capacitive coupling from mains interference [19]. Keep participants and equipment away from obvious noise sources.
- Removal: In post-processing, a notch filter at 50 Hz or 60 Hz can be applied to selectively remove this frequency component [20] [19].
Q4: What causes cable movement artifacts, and how can they be reduced?
Cable movement artifacts are caused by triboelectric noise, which is generated by the friction of a cable's internal components or the motion of the conductor within a magnetic field [19]. These artifacts appear as sudden, high-amplitude changes or slow drifts in the signal [20] [19].
- Reduction Strategies:
- Use shorter cables and secure them to the EEG cap or the participant using velcro or putty to minimize movement [13].
- Select equipment with special low-noise cable components designed to reduce friction [19].
- Utilize systems with active shielding, which eliminates capacitive coupling and makes the signal less susceptible to cable sway [19].
Q5: How does a bad reference electrode connection affect my entire dataset?
A bad connection in the reference electrode can have severe consequences because the reference signal is used in the calculation of all other channels [19]. If the reference is unstable, its noise and artifacts will be introduced into every single EEG channel during re-referencing [20]. This can manifest as widespread, non-EEG signals, slow drifts, or pops throughout the entire recording [19]. Always ensure the reference electrode has a secure connection and low impedance before recording [19].
Table 1: Characteristics and Solutions for Technical and Environmental Artifacts
| Artifact Type | Visual Characteristics on EEG | Common Causes | Prevention Strategies | Post-Processing Removal/Correction |
|---|---|---|---|---|
| Electrode Pop [17] [18] [19] | Sudden, sharp, high-amplitude deflection; very steep upslope; localized to a single channel. | Loose electrode, poor skin contact, drying electrolyte, dirty electrode. | Secure electrode application; check impedances before recording. | Re-referencing to a different electrode; manual rejection of contaminated epochs; interpolation of bad channels [17] [20]. |
| Cable Movement [20] [19] | Sudden, high-amplitude changes or slow drifts; may have oscillatory patterns if cable is swinging. | Triboelectric effect from cable friction; motion of conductor in magnetic field. | Use short, secured cables; select low-noise cables; use active shielding. | Artifact rejection; high-pass filtering for slow drifts [20]. |
| AC (Mains) Interference [20] [19] | High-frequency (50/60 Hz), monotonous, rhythmic waves present across multiple channels. | Electromagnetic fields from power lines and electronic equipment. | Use Faraday cage; active shielding; remove AC equipment; increase distance from noise sources. | Notch filter at 50/60 Hz [20] [19]. |
| Bad Reference Electrode [20] [19] | Widespread, non-EEG signals, drifts, or pops affecting all channels in the montage. | Poor connection, high impedance, or loose placement of the reference electrode. | Ensure secure and correct placement of reference electrode; check its impedance. | Change re-referencing scheme; interpolate or discard the bad reference channel. |
Experimental Protocols for Artifact Handling
Protocol 1: A Standardized Pre-Recording Checklist to Minimize Technical Noise
This protocol aims to prevent technical artifacts before data collection begins.
- Electrode Preparation: Clean electrodes according to manufacturer guidelines to prevent contamination-related pops.
- Cap Fitting: Ensure the EEG cap fits snugly without being too tight. Part the hair to ensure direct scalp contact for each electrode.
- Impedance Check: Measure impedance for every electrode, including ground and reference. Aim for impedances below 20 kΩ, ensuring they are stable and balanced across channels [13] [19].
- Cable Management: Secure the headbox and bundle cables to the participant's clothing using velcro straps. Use a cape or net to minimize cable sway [13].
- Environment Scan: Turn off or move non-essential electronic devices. Ensure the participant is not sitting directly under or near strong sources of AC power.
Protocol 2: Offline Processing Workflow for Technical Artifact Removal
This protocol outlines a systematic approach for cleaning recorded data from technical artifacts.
- Visual Inspection: Manually inspect the raw data to identify obvious artifacts like electrode pops, cable movements, and bad channels [13].
- Filter Application:
- Bad Channel Handling: Identify persistently noisy or flat-lined channels. These can be rejected and their data interpolated from surrounding good channels using methods like spherical spline interpolation [20].
- Artifact Rejection: For episodic artifacts like large cable movements, mark and reject the contaminated epochs or trials to prevent them from affecting downstream analysis like ERP averaging [20].
- Re-referencing: Finally, re-reference the data to a stable reference (e.g., average reference, linked mastoids) to mitigate the impact of localized noise in the original reference [17].
The Scientist's Toolkit: Essential Materials and Reagents
Table 2: Key Research Reagent Solutions for EEG Experimentation
| Item | Function in Research |
|---|---|
| High-Conductivity Electrolyte Gel | Forms a stable, low-impedance electrical bridge between the scalp and electrode, crucial for preventing electrode pops and ensuring high-quality signal acquisition [17] [13]. |
| Abrasive Electrode Prep Gel/Skin Prep | Gently exfoliates the scalp's outer layer (stratum corneum) to lower initial skin impedance, improving signal quality and stability from the start of the recording. |
| Electrode Cleaning Solution | Used to disinfect and remove gel/salt residue from electrodes after use, preventing contamination and ensuring longevity, which helps avoid artifacts in future sessions [17]. |
| Active Shielded EEG Cables | Cables with built-in active shielding technology that minimizes the pickup of environmental electromagnetic noise (e.g., AC interference) and reduces artifacts from cable movement [19]. |
| Notch Filter (50/60 Hz) | A standard digital signal processing tool applied during data analysis to selectively attenuate the specific frequency of AC power line interference without significantly affecting broader brain signals [20] [19]. |
The Critical Importance of Signal-to-Noise Ratio (SNR) in Research and Clinical Applications
Frequently Asked Questions (FAQs)
Q: What is the signal-to-noise ratio (SNR) in EEG, and why is it a critical challenge? A: The signal-to-noise ratio (SNR) is the ratio of meaningful brain activity you want to measure ("signal") to all other contaminating activity ("noise") [21]. It is critical because EEG measures minuscule electrical signals (on the order of millionths of a volt) that are easily obscured by noise from the body and environment [21]. A poor SNR can lead to incorrect, misleading results and potentially bias clinical diagnoses by masking genuine neural signals or making artifacts look like brain activity [21] [1].
Q: What are the most common types of artifacts that degrade SNR in EEG? A: Artifacts are categorized by their origin [22] [1]:
- Physiological Artifacts: Originate from the participant's body.
- Ocular Artifacts: From eye blinks and movements, causing high-amplitude, low-frequency deflections over frontal electrodes [22] [1].
- Muscle Artifacts (EMG): From jaw clenching, swallowing, or frowning, producing high-frequency, broadband noise [22] [1].
- Cardiac Artifacts (ECG/ Pulse): From heartbeats, appearing as rhythmic waveforms in the EEG [22] [1].
- Non-Physiological Artifacts: Originate from external sources.
Q: My ERP results are noisy. How can I improve the SNR during data collection? A: Improving SNR starts with a well-designed experimental protocol [21]:
- Minimize External Noise: Use a high-quality EEG system, remove sources of electromagnetic noise (e.g., cables, cell phones) from the recording area, and employ a notch filter for power line interference [21].
- Minimize Participant Noise: A trained technician should create a relaxed atmosphere. Use a protocol that keeps the participant focused and engaged, with frequent, scheduled breaks for blinking and moving placed between experimental trials [21].
- Maximize Signal Averaging: For Event-Related Potentials (ERPs), ensure a sufficient number of trials. The averaging process assumes the neural response to a stimulus is constant, while noise is random. Averaging multiple trials suppresses random noise, allowing the signal to stand out [21].
Q: What are the standard methods for removing artifacts during data post-processing? A: Several established methods are used for artifact removal [21] [22]:
- Blind Source Separation (BSS) / Independent Component Analysis (ICA): These are among the most common algorithms. They decompose the EEG signal into statistically independent components, allowing you to identify and remove components associated with artifacts (like blinks or muscle activity) while preserving neural signals [21] [22].
- Regression Methods: These use reference channels (e.g., EOG for eye artifacts) to estimate and subtract the artifact contribution from the EEG data [22].
- Manual Cleaning: An expert visually examines the data and manually removes sections contaminated by large artifacts. This is often used alongside automated methods [21].
- Filtering: High-pass, low-pass, and band-stop filters can be used to remove noise outside the frequency range of interest (e.g., slow drifts from sweat or high-frequency muscle noise) [22].
Troubleshooting Guides
Problem 1: Persistent Ocular (Eye Blink) Artifacts
Symptoms: Large, low-frequency, high-amplitude deflections in frontal channels (e.g., Fp1, Fp2) that are time-locked to blinks [1].
Step-by-Step Resolution:
- Prevention Check: Review your experimental design. Were participants given adequate and scheduled breaks to blink naturally between trials? [21]
- Reference Channel Verification: Ensure you have recorded clean EOG signals from dedicated vertical and horizontal EOG electrodes. These are crucial for many correction methods [22].
- Post-Processing Correction: Apply a validated artifact removal algorithm. Independent Component Analysis (ICA) is highly effective for isolating and removing blink artifacts [21] [22].
- Validation: Before finalizing your analysis, visually compare the data before and after correction to ensure blinks were removed without distorting the underlying neural signal.
Problem 2: Excessive Muscle Artifact (EMG) Contamination
Symptoms: High-frequency, broadband noise superimposed on the EEG signal, often visible as increased activity in the beta (>13 Hz) and gamma (>30 Hz) frequency ranges [1].
Step-by-Step Resolution:
- Protocol Review: Check if the participant was appropriately instructed to relax their jaw, face, and neck muscles during the recording.
- Source Identification: Use the data's topographic maps to identify which channels are most affected. This can help locate the source of the muscle tension (e.g., jaw vs. neck) [1].
- Algorithmic Removal: Muscle artifacts are often successfully removed using Blind Source Separation (BSS) methods like ICA, as EMG signals are statistically independent from EEG [22] [1].
- Filtering (as a last resort): If high-frequency neural activity is not the focus of your study, applying a low-pass filter (e.g., cutting off below 40-50 Hz) can reduce EMG. However, this will also remove genuine high-frequency brain activity [22].
Problem 3: Continuous 50/60 Hz Power Line Interference
Symptoms: A persistent, high-frequency oscillation at 50 Hz (e.g., Europe) or 60 Hz (e.g., North America) visible in the power spectrum of all channels [21] [1].
Step-by-Step Resolution:
- Inspection & Prevention: Before starting the recording, check that all electrode impedances are low and balanced. Visually inspect for loose or unshielded cables [1].
- Environment Scan: Remove or move the participant away from obvious noise sources like computer monitors, power cables, and fluorescent lights [21].
- Apply a Notch Filter: Use a 50/60 Hz notch (band-stop) filter during data acquisition or in post-processing. This is a standard and effective solution for this specific type of noise [21].
- Re-reference: If the problem persists, try re-referencing the data to a different, clean reference channel.
Experimental Protocols for SNR Enhancement
Protocol 1: Event-Related Potential (ERP) Averaging
Objective: To isolate a consistent neural response to a specific stimulus from background brain noise [21].
Detailed Methodology:
- Stimulus Presentation: Present a discrete stimulus (e.g., an image, sound, or word) to the participant. The onset of this stimulus is marked as time "zero."
- EEG Recording: Record continuous EEG while presenting the stimulus multiple times (dozens to hundreds of repetitions).
- Epoching: Segment the continuous EEG data into individual "epochs" or "trials" surrounding each stimulus presentation (e.g., from -200 ms before to 800 ms after stimulus onset).
- Baseline Correction: For each epoch, subtract the average voltage of the pre-stimulus period (e.g., -200 to 0 ms) from the entire epoch to remove slow drifts.
- Artifact Rejection: Automatically or manually reject epochs that contain extreme voltages (e.g., > ±100 µV) indicating large artifacts.
- Averaging: Calculate the average voltage across all remaining epochs for each time point. The underlying principle is that the brain's time-locked response to the stimulus will be consistent and remain in the average, while random, non-time-locked neural noise will average toward zero [21].
The workflow below illustrates this multi-step process for obtaining a clean ERP signal.
Protocol 2: Independent Component Analysis (ICA) for Artifact Removal
Objective: To separate and remove source-localized artifacts (e.g., from eyes, heart, muscles) from EEG data without relying on a reference channel [22].
Detailed Methodology:
- Data Preprocessing: First, apply standard preprocessing steps: filtering (e.g., high-pass at 1 Hz, low-pass at 40-50 Hz) and bad channel removal/interpolation.
- Data Decomposition: The ICA algorithm is applied to the preprocessed data. It mathematically decomposes the multi-channel EEG signal into a set of "independent components." Each component has a fixed scalp topography and a time course of activation.
- Component Classification: The researcher classifies each component to identify its origin. This is done by examining the component's topography (does it look like frontal eye fields?), time course (is it rhythmic with the heartbeat?), and power spectrum (is it high-frequency like muscle noise?). Machine learning classifiers can assist with this step [1].
- Artifact Component Removal: The components identified as artifacts are projected out of the data.
- Data Reconstruction: The EEG signal is reconstructed from the remaining brain-based components, now largely free of the identified artifacts.
The following diagram outlines the core ICA workflow for separating mixed signals into independent sources.
The Scientist's Toolkit: Essential Research Reagents & Materials
The following table details key materials and software tools essential for conducting high-quality EEG research with a strong focus on SNR.
| Item Name | Function / Explanation |
|---|---|
| High-Density EEG System | An EEG recording system with a sufficient number of electrodes (e.g., 64, 128, or more). Higher spatial sampling improves the ability of algorithms like ICA to separate neural signals from artifacts [21] [1]. |
| Electrooculogram (EOG) Electrodes | Dedicated electrodes placed near the eyes to record eye movements and blinks. This provides a reference signal for detecting and correcting ocular artifacts using regression or other methods [22]. |
| Abrasive Electrolyte Gels | Conductive gel applied to lower the impedance between the scalp and EEG electrodes. Low and stable impedance (< 10 kΩ) is critical for obtaining a high-quality signal with less vulnerability to noise [1]. |
| Electromyogram (EMG) Electrodes | Surface electrodes placed on relevant muscles (e.g., masseter, trapezius) to record muscle activity. This provides a ground-truth reference for identifying and validating the removal of EMG artifacts [22]. |
| Electrocardiogram (ECG) Electrode | A single electrode placed to record the electrical activity of the heart. This is used as a reference for identifying and removing cardiac artifacts from the EEG data [22]. |
| Electrically Shielded Room | A room designed to block external electromagnetic interference. This is the gold-standard for minimizing environmental noise, though it is not always available [21]. |
| Blind Source Separation (BSS) Software | Software toolkits (e.g., EEGLAB, FieldTrip) that implement algorithms like Independent Component Analysis (ICA) to separate artifact sources from brain sources in the recorded signal [21] [22]. |
| Notch Filter | A standard feature in EEG acquisition software that applies a narrow band-stop filter to remove the fundamental frequency of power line interference (50/60 Hz) and its harmonics [21]. |
Artifact Impact on Pharmacodynamic Modeling and Drug Effect Assessment
Electroencephalogram (EEG) is a powerful, non-invasive tool for measuring brain activity with high temporal resolution, making it valuable for assessing central nervous system (CNS) drug effects in Pharmacodynamic (PD) modeling [22]. PD modeling describes how a drug's concentration relates to its biological effect, going beyond whether a drug works to explain how much, how fast, and for how long it works [23]. However, EEG signals are measured in microvolts and are extremely susceptible to contamination from various sources of noise, known as artifacts [1]. These unwanted signals can obscure underlying neural activity and compromise data quality, potentially leading to misinterpretation of a drug's true PD effects [1] [22]. In clinical research, artifacts might imitate cognitive or pathologic activity and therefore bias visual interpretation and diagnosis [22]. This technical support guide addresses how these artifacts impact PD modeling and provides methodologies for their identification and removal.
Troubleshooting Guides & FAQs
FAQ: Core Concepts and Impact
Q1: What is the fundamental relationship between pharmacokinetics (PK), pharmacodynamics (PD), and EEG artifacts?
A1: PK describes how a drug moves through the body (Absorption, Distribution, Metabolism, Excretion), resulting in a concentration-time profile. PD describes the biochemical and physiological effects of the drug, relating its concentration at the target site to the observed response [24]. EEG is often used as a direct or indirect measure of this PD response for CNS-active drugs. Artifacts are any components of the EEG signal that do not originate from the brain [1]. They contaminate the PD endpoint (the EEG signal), thereby distorting the established relationship between drug concentration and effect, which can lead to incorrect conclusions about a drug's efficacy and potency [1] [22].
Q2: Why are EEG artifacts particularly problematic for population PD modeling?
A2: Population PD models use nonlinear mixed-effects modeling to understand drug action and variability across individuals [25]. Artifacts introduce uncontrolled, non-physiological variability into the data. This can bias parameter estimates for the typical population response (e.g., Emax, EC50) and inflate estimates of between-subject variability (BSV), making it difficult to identify true biological or clinical sources of variability [25]. Furthermore, if the likelihood of artifact contamination is correlated with an underlying disease state (e.g., more movement artifacts in a restless patient population), the dropout or missing data mechanism becomes "informative," which cannot be ignored in the modeling process [25].
Q3: How can a simple ocular artifact lead to a mis-specified PD model?
A3: Ocular artifacts (blinks, movements) generate high-amplitude, low-frequency signals in the frontal EEG channels [1] [20]. If unaddressed, these can be misinterpreted as a drug-induced change in slow-wave brain activity (e.g., delta or theta power). A model might incorrectly attribute this "effect" to the drug concentration, leading to a flawed concentration-effect relationship. The model's parameters would be biased, and subsequent simulations for dose selection would be unreliable. This is a specific example of how artifact can lead to model misspecification.
Q4: What are the challenges in removing muscle artifacts (EMG) from EEG for PD analysis?
A4: Muscle artifacts are particularly challenging because they are broadband, generating noise that overlaps the entire EEG frequency spectrum, including beta and gamma bands which are often relevant for cognitive PD endpoints [1] [22]. Unlike ocular artifacts, they can originate from many muscle groups (jaw, neck, forehead) and lack a single, simple topographic distribution on the scalp. Their statistical independence from neural signals makes techniques like Independent Component Analysis (ICA) a potential solution, but automated removal is difficult without potentially removing genuine neural signals of interest [22].
Troubleshooting Guide: Common Scenarios
Problem 1: Erratic Dose-Response Relationship
- Symptoms: The PD model fits poorly. The plot of drug concentration vs. EEG response shows high scatter and no clear trend, or a trend that contradicts pre-clinical findings.
- Potential Artifact Cause: Intermittent, high-amplitude artifacts like electrode "pops," cable movement, or gross body movements are adding large, unpredictable variance to the EEG response variable [1] [20].
- Solutions:
- Inspect Raw Data: Visually inspect the continuous EEG for all subjects to identify and mark epochs with large transients.
- Apply Artifact Rejection: Implement a robust artifact rejection policy to exclude contaminated epochs from the analysis. The threshold for rejection should be justified based on the amplitude distribution of the clean data.
- Check Electrode Integrity: For persistent problems on specific channels, investigate the possibility of faulty electrodes or poor impedance [20].
Problem 2: Apparent Delay (Hysteresis) in Drug Effect
- Symptoms: A counterclockwise loop is observed in the plot of concentration vs. response, suggesting a delay between plasma concentration and effect, but this is not supported by the drug's known mechanism.
- Potential Artifact Cause: Slow drifts in the EEG baseline caused by subject sweating or poor electrode contact can mimic a delayed PD response [20]. The slow drift alters the signal independently of the drug concentration, creating a hysteresis-like loop.
- Solutions:
- Apply High-Pass Filtering: Use a zero-phase high-pass filter with an appropriate cutoff (e.g., 0.5 Hz) to remove the very slow drifts without distorting the slower EEG frequencies of interest [20].
- Investigate Sensors: Review impedance logs and data from the reference/ground electrodes, as drift can often originate from them [20].
Problem 3: Inflated Between-Subject Variability
- Symptoms: The population PD model estimates unusually high BSV for one or more parameters, making it difficult to identify covariate relationships (e.g., effect of age or renal function).
- Potential Artifact Cause: Systematic differences in artifact load between subjects. For example, one subject group (e.g., older patients) might have more tremors or eye movements, while another (e.g., anxious patients) might have more muscle tension, introducing group-wise biases that the model interprets as random variability [25] [22].
- Solutions:
- Stratified Analysis: Compare the average artifact load (e.g., number of rejected epochs, EMG power) across suspected subgroups.
- Advanced Cleaning: Use artifact source separation methods like ICA to remove specific artifacts (like eye blinks or persistent muscle noise) uniformly from all subjects before model fitting [1] [22].
- Incorporate Artifact Metrics: Include a quantitative measure of data quality (e.g., percentage of clean data) as a covariate in the population model to account for its influence on BSV.
Table 1: Common EEG Artifacts and Their Impact on PD Modeling Parameters
| Artifact Type | Origin | EEG Frequency Band | Key Impact on PD Modeling |
|---|---|---|---|
| Ocular (EOG) | Eye blinks & movements [1] | Delta, Theta [1] | Biases estimation of E0 (baseline) and Emax; can mimic drug effect on slow waves [25] |
| Muscle (EMG) | Head, jaw, neck muscle activity [1] | Broadband, especially Beta/Gamma [1] | Inflates residual variability; obscures drug effects on high-frequency oscillations [22] |
| Cardiac (ECG) | Heartbeat & pulse [22] | ~1.2 Hz (Pulse), broader (ECG) [1] | Introduces rhythmic, non-drug-related signal; can be confounded with pathological brain rhythms [20] |
| Electrode Pop | Sudden change in electrode-skin impedance [1] | Broadband [1] | Causes large, spurious outliers; severely biases individual PD parameter estimates [25] |
| Sweat/Skin Potential | Changes in skin conductivity [20] | Very slow drifts (<0.5 Hz) [20] | Creates baseline drift; can be mistaken for tolerance development (clockwise hysteresis) [25] |
| Line Noise | AC power interference [1] | 50 Hz or 60 Hz [1] | Adds structured noise at a specific frequency; can interfere with spectral PD analysis. |
Table 2: Artifact Removal Techniques Comparison
| Technique | Principle | Advantages | Limitations | Suitability for PD Modeling |
|---|---|---|---|---|
| Artifact Rejection | Manually or automatically excluding contaminated data segments [22] | Simple to implement; guarantees clean data. | Reduces data quantity; potentially introduces selection bias. | High for large, transient artifacts. Poor if artifacts are pervasive. |
| Filtering | Attenuating frequency bands dominated by artifacts. | Effective for line noise and slow drifts. | Can distort genuine EEG signals; phase shifts can be critical for ERPs. | Moderate. Useful as a preprocessing step but cannot remove artifacts overlapping with EEG. |
| Regression | Uses reference channels (EOG, ECG) to subtract artifact from EEG [22]. | Conceptually straightforward. | Requires additional reference recordings; can remove genuine brain signal correlated with reference [22]. | Low to Moderate. Risk of over-correction and signal loss. |
| Independent Component Analysis (ICA) | Blind source separation; identifies and removes artifact components [1] [22]. | Powerful for removing ocular, muscle, and cardiac artifacts without reference channels. | Computationally intensive; requires manual component inspection; performance depends on data length/quality. | High. The method of choice for many physiological artifacts in PD studies. |
| Wavelet Transform | Decomposes signal into time-frequency components for selective removal. | Good for non-stationary, transient artifacts. | Complex parameter selection; reconstruction can introduce artifacts. | Moderate. Can be effective for specific artifacts like electrode pops. |
Experimental Protocols
Protocol 1: Standardized Preprocessing Pipeline for EEG-based PD Modeling
Objective: To provide a robust, standardized methodology for cleaning EEG data prior to PD model development.
Materials:
- Raw continuous EEG data from a clinical trial.
- Software capable of performing filtering, ICA, and artifact rejection (e.g., EEGLAB, BrainVision Analyzer, MNE-Python).
Procedure:
- Data Import and Channel Setup: Import raw data. Assign proper channel locations and identify reference/ground electrodes.
- Filtering:
- Bad Channel Identification and Interpolation: Identify channels with consistently poor signal quality (e.g., flat-lined, excessively noisy) and interpolate them using data from surrounding good channels.
- Re-referencing: Re-reference the data to a robust average reference or a specific reference (e.g., linked mastoids).
- Independent Component Analysis (ICA):
- Run ICA on the filtered and cleanly referenced data.
- Manually inspect the resulting components. Identify artifact components based on their topography (e.g., frontal for eye blinks), time course (e.g., spike-like for cardiac), and frequency spectrum (e.g., broadband for muscle) [1] [22].
- Remove identified artifact components from the data.
- Epoching (if applicable): Segment the continuous data into epochs around relevant events (e.g., stimulus presentation).
- Final Artifact Rejection: Apply an automated amplitude threshold (e.g., ±100 µV) to reject any remaining epochs containing large artifacts that were not fully captured by previous steps.
- Data Export: Export the final, cleaned EEG metrics (e.g., power in a specific band, ERP amplitude) for subsequent PK-PD analysis.
Protocol 2: Handling Informative Dropout Due to Artifacts
Objective: To mitigate bias in population PD parameters when data loss (dropout) is related to the underlying PD response.
Context: In a study of a sedative drug, subjects with a high PD response (deep sedation) may be more likely to produce movement artifacts or drop out, making the data "Not Missing at Random" (NMAR) [25].
Procedure:
- Characterize the Dropout: Document the reason and timing for every subject dropout or epoch rejection. Correlate these with the observed (or imputed) PD response and drug concentration.
- Implement a Model-Based Approach: Use modeling software (e.g., NONMEM) that allows for joint modeling of the PD response and the dropout process.
- Develop a base PD model (e.g., an
Emaxmodel) for the observed data. - Develop a model for the time to dropout (e.g., a time-to-event model), where the hazard of dropout is a function of the underlying PD effect predicted by the PD model.
- Develop a base PD model (e.g., an
- Estimate Parameters Jointly: Simultaneously estimate all parameters of the PD model and the dropout model. This accounts for the fact that the probability of a data point being missing is related to the unobserved value the model is trying to predict.
- Compare with Naive Methods: Compare the results (parameter estimates, standard errors) from the joint model with those from a model that simply ignores the missing data to illustrate the potential for bias [25].
Signaling Pathways and Workflows
Diagram 1: Impact of Artifacts on PD Modeling Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Tools for EEG-based PD Studies
| Item / Solution | Function / Application | Key Consideration for PD Modeling |
|---|---|---|
| High-Density EEG System (e.g., 64+ channels) | Records brain electrical activity from the scalp. | More channels improve spatial resolution and the efficacy of source separation techniques like ICA for artifact removal [22]. |
| Active Electrode Systems | Amplifies signal at the scalp to reduce cable movement artifacts [20]. | Crucial for mobile or long-duration studies (e.g., sleep PD studies) to maintain signal fidelity. |
| Electrooculogram (EOG) & Electrocardiogram (ECG) Electrodes | Records eye movement and cardiac activity as reference channels [22]. | Provides dedicated data to facilitate regression-based removal of ocular and cardiac artifacts, improving PD signal purity. |
| Conductive Electrode Gel & Abrasive Prep Gel | Ensures stable, low-impedance connection between electrode and scalp. | Critical for minimizing slow drifts (sweat) and transient pops; stable impedance is foundational for reliable data [20]. |
| Software with ICA Capability (e.g., EEGLAB, BrainVision Analyzer) | Provides tools for blind source separation to identify and remove artifact components [1] [22]. | A non-negotiable tool for modern EEG preprocessing. Allows removal of major artifacts without distorting the underlying neural signal. |
| Population PK/PD Software (e.g., NONMEM) | Performs nonlinear mixed-effects modeling to relate drug exposure to the cleaned PD endpoint [25]. | The final step; requires a high-quality, clean EEG-derived PD metric as input to build a valid drug-disease model. |
| Line-Field Confocal Optical Coherence Tomography (LC-OCT) | A non-invasive technique to capture skin parameters (e.g., epidermal thickness) as a PD endpoint [26]. | Represents an alternative, non-EEG PD biomarker that is less susceptible to classical EEG artifacts, useful for topical drugs. |
EEG Denoising Techniques: From Traditional Algorithms to Advanced Deep Learning Approaches
Frequently Asked Questions
Q: What is the main challenge when working with EEG signals? EEG signals are measured in microvolts, making them extremely sensitive to contamination from both internal body processes (like blinking or heartbeats) and external interference (like cable movement or AC power lines). This introduces noise, known as artifacts, which can distort or mask genuine neural signals [1].
Q: Why is artifact removal a critical preprocessing step? Artifacts can obscure underlying neural activity, compromise data quality, and lead to misinterpretation or even clinical misdiagnosis. For instance, artifacts might imitate cognitive or pathologic activity and bias visual interpretation and diagnosis in clinical research [22] [1]. Effective removal is essential for accurate analysis and reliable applications, especially in brain-computer interfaces [22].
Q: What are the most common types of physiological artifacts? The primary physiological artifacts are:
- Ocular Artifacts: From eye blinks and movements, producing sharp, high-amplitude deflections, especially in frontal electrodes [22] [1].
- Muscle Artifacts (EMG): From muscle contractions (e.g., jaw clenching, swallowing), producing high-frequency, broadband noise [22] [1].
- Cardiac Artifacts (ECG): From heartbeats, appearing as rhythmic waveforms in the EEG [22] [1].
Q: How do Regression, Filtering, and BSS approaches fundamentally differ?
- Regression relies on reference channels (e.g., EOG) to estimate and subtract artifact contributions from EEG signals [22].
- Filtering uses frequency-based separation to remove noise components outside the typical EEG bands (e.g., using a high-pass filter to remove drifts) [22] [27].
- Blind Source Separation (BSS), such as ICA, decomposes the multi-channel EEG signal into statistically independent components, allowing for the identification and removal of artifact-related components without prior knowledge of the sources [28].
Troubleshooting Guides
Issue 1: My EEG data is contaminated with eye blinks and movements.
Problem: Ocular artifacts manifest as large, low-frequency deflections, predominantly in frontal channels (like Fp1, Fp2), which can mask genuine neural signals in the delta and theta bands [22] [1].
Solutions:
- Regression Method: Use a vertical EOG reference channel to estimate and subtract the ocular artifact from the EEG data.
- Protocol: Calculate the transmission factors (γ, δ) between the EOG and EEG channels. Apply the correction:
EEG_corrected = EEG_raw - γ*F(VEOG)[22]. - Troubleshooting Tip: Be aware that regression can be affected by bidirectional interference, where the EEG signal also contaminates the EOG reference, potentially leading to an over-correction and removal of genuine brain signals [22].
- Protocol: Calculate the transmission factors (γ, δ) between the EOG and EEG channels. Apply the correction:
- ICA (a BSS method): This is a highly effective and commonly used approach for ocular artifacts.
- Protocol:
- Apply ICA to decompose your multi-channel EEG data into independent components (ICs).
- Visually inspect the components' topography (frontally dominant), time course (large, infrequent spikes corresponding to blinks), and power spectrum (low-frequency focus) to identify artifact-related ICs [28].
- Remove the identified artifact components.
- Reconstruct the clean EEG signal from the remaining components [28].
- Protocol:
Issue 2: My signal has high-frequency noise from muscle activity (EMG).
Problem: Muscle artifacts introduce high-frequency, broadband noise that overlaps with and can obscure beta and gamma band neural oscillations, which are critical for studying active thinking and motor activity [22] [1].
Solutions:
- Filtering: Apply a low-pass filter with a cut-off frequency around 30-40 Hz to attenuate high-frequency EMG noise. However, this will also remove genuine high-frequency neural activity [1].
- ICA (a BSS method): Often the preferred method as it can separate EMG artifacts based on their statistical independence from EEG.
- Advanced/Combined Methods: For challenging cases, consider hybrid methods like Wavelet-ICA, where the signal is first decomposed using wavelets to isolate noisy elements, which are then processed with ICA [22].
Issue 3: I am dealing with multiple types of artifacts simultaneously.
Problem: Real-world EEG data is often contaminated by a mixture of ocular, muscle, cardiac, and technical artifacts, making single-method approaches insufficient [5].
Solutions:
- Sequential Pipeline: Combine multiple methods in a logical sequence.
- Protocol:
- Basic Filtering: Start with a band-pass filter (e.g., 1-40 Hz) to remove very low-frequency drifts and high-frequency line noise [27].
- BSS/ICA: Apply ICA to decompose the filtered data and remove components corresponding to various artifacts (ocular, muscular, etc.) [28].
- Advanced Denoising: For residual noise, consider using an automated algorithm like the Generalized Eigenvalue De-Artifacting Instrument (GEDAI), which uses a leadfield model of the brain to separate neural from non-neural activity [29].
- Protocol:
- Use a Robust, Integrated Tool: Explore modern deep-learning-based tools like the Artifact Removal Transformer (ART), which is designed to remove multiple artifact sources in one go using an end-to-end transformer architecture [3].
Table 1: Comparison of Traditional Artifact Removal Methods
| Method | Core Principle | Key Advantages | Key Limitations | Ideal Use Case |
|---|---|---|---|---|
| Regression [22] | Uses a reference channel (e.g., EOG) to estimate and subtract artifacts. | Conceptually simple; requires a dedicated reference channel. | Assumes a constant relationship between reference and EEG; suffers from bidirectional contamination (EEG signal can be subtracted). | Situations with a clean, dedicated reference channel for the artifact. |
| Filtering [22] [27] | Removes signal components based on frequency. | Simple and fast; effective for removing noise outside the EEG frequency band (e.g., line noise). | Cannot remove artifacts whose frequency overlaps with neural signals (e.g., EMG in beta/gamma bands). | Initial preprocessing to remove DC drift, high-frequency noise, and power-line interference. |
| Blind Source Separation (BSS) - ICA [22] [28] | Decomposes multi-channel EEG into statistically independent sources. | Does not require reference channels; effective for separating various physiological artifacts. | Computationally intensive; requires manual component inspection in traditional form; performance can degrade with low channel count. | Multi-channel datasets for removing ocular, muscle, and cardiac artifacts. |
| Blind Source Separation (BSS) - PCA [28] | Decomposes signals into orthogonal components based on variance. | Reduces data dimensionality; can be faster than ICA. | Artifacts are not always the highest variance sources; can be less effective than ICA for physiological artifacts. | As a preprocessing step for dimensionality reduction or for specific artifact types. |
Table 2: Common EEG Artifacts and Their Signatures
| Artifact Type | Origin | Key Characteristics in EEG Signal [22] [1] |
|---|---|---|
| Ocular (EOG) | Eye blinks and movements. | Time-domain: High-amplitude, slow deflections, frontally dominant.Frequency-domain: Dominates delta/theta bands. |
| Muscle (EMG) | Head, jaw, or neck muscle contractions. | Time-domain: High-frequency, low-amplitude "spiky" activity.Frequency-domain: Broadband, dominates beta/gamma bands. |
| Cardiac (ECG) | Electrical activity from the heart. | Time-domain: Rhythmic, spike-like pattern synchronized with heartbeat.Frequency-domain: Can overlap with multiple EEG bands. |
| Electrode Pop | Sudden change in electrode-skin impedance. | Time-domain: Abrupt, high-amplitude transient often isolated to a single channel. |
| Power Line | Electromagnetic interference from AC power. | Frequency-domain: Sharp peak at 50 Hz or 60 Hz. |
Experimental Protocols
Protocol 1: Removing Ocular Artifacts using Regression
Objective: To subtract ocular artifacts from contaminated EEG signals using a recorded EOG reference.
Materials:
- Raw multi-channel EEG data.
- Simultaneously recorded vertical EOG (VEOG) data.
Procedure:
- Data Inspection: Visually identify segments of data with prominent ocular artifacts (large, low-frequency waves in frontal channels).
- Calculate Transmission Factors: Determine the scaling factors (e.g., γ) that define the amplitude relationship between the VEOG channel and each EEG channel. This often involves a calibration phase where the relationship between the EOG and the contaminated EEG is established [22].
- Apply Correction: For each EEG channel, subtract the scaled VEOG signal:
EEG_corrected = EEG_raw - γ * VEOG[22]. - Validation: Inspect the corrected EEG to ensure artifact reduction and verify that neural signals have not been disproportionately removed due to bidirectional interference [22].
Protocol 2: Isolating and Removing Artifacts using ICA
Objective: To decompose EEG signals and manually remove components representing artifacts.
Materials:
- Multi-channel EEG data (minimum recommended channels for effective ICA is >19, but it can be attempted with fewer) [5].
- Software with ICA capabilities (e.g., EEGLAB).
Procedure:
- Preprocessing: Apply a high-pass filter (e.g., 1 Hz cut-off) to remove slow drifts that can impede ICA performance [27].
- ICA Decomposition: Run an ICA algorithm (e.g., Infomax, FastICA) on the preprocessed data. This produces a mixing matrix (A) and a set of independent components (ICs) [28].
- Component Classification: Inspect the ICs to identify artifacts. Key features of artifactual components include [28]:
- Ocular ICs: Large, infrequent deflections in the time course; topography focused on frontal poles.
- Muscle ICs: High-frequency, chaotic activity; topography focused on temporal areas.
- Cardiac ICs: Regular, pulsatile peaks at the heart rate.
- Remove Artifact Components: Select and remove the identified artifactual components.
- Reconstruct EEG: Project the remaining (clean) components back to the sensor space to obtain the artifact-corrected EEG signal [28].
Workflow for Traditional EEG Artifact Removal
The following diagram outlines a general, effective workflow for cleaning EEG data using the discussed methods.
The Scientist's Toolkit
Table 3: Essential Research Reagents & Materials
| Item | Function in EEG Research |
|---|---|
| Multi-channel EEG System | Acquires electrical brain activity from the scalp. A higher number of channels improves spatial resolution and the efficacy of source separation methods like ICA [30]. |
| Reference Electrodes (EOG, ECG) | Provide dedicated recordings of potential artifact sources (eye movement, heartbeats), which are crucial for regression-based removal methods [22]. |
| Conductive Gel/Saline Solution | Ensures good electrical contact between the electrode and the scalp, reducing impedance and preventing electrode pop artifacts [1]. |
| EEGLAB (MATLAB Toolbox) | A foundational software environment providing extensive functions for EEG processing, including ICA, filtering, and visualization [28]. |
| Leadfield/Head Model | A computational model that describes how electrical currents in the brain project to electrodes on the scalp. It is essential for source localization and advanced denoising methods like GEDAI [29]. |
| Wavelet Toolbox | Provides algorithms for performing wavelet transforms, useful for time-frequency analysis and denoising non-stationary signals like EMG artifacts [31]. |
Independent Component Analysis (ICA) and Spatial Filtering Implementation
Frequently Asked Questions (FAQs)
1. What is the primary purpose of using ICA on EEG data? ICA is used as a blind source separation technique to decompose multi-channel EEG data into statistically independent components (ICs) [32]. The primary goals are to:
- Attenuate artifacts (e.g., from eye blinks, muscle activity, heart beats) without removing the contaminated data portions, thereby preserving the original amount of data for analysis [33] [32].
- Identify and analyze statistically independent cortical processes by isolating the activity of partially synchronized cortical domains, even if their scalp projections overlap [34].
2. How much data do I need for a stable ICA decomposition?
A general rule is that finding N stable components (from N-channel data) requires more than k*N² data sample points (at each channel), where N² is the number of weights in the unmixing matrix that ICA is trying to learn and k is a multiplier [34].
- Example: For 32-channel data, a dataset with 30,800 sample points gives approximately
30 points/weight(30800/32²) [34]. - High-Channel Count Note: The value of
kappears to increase with the number of channels. For high-density arrays (e.g., 256 channels), even 30 points per weight may be insufficient, indicating a need for substantial recording durations [34].
3. My ICA components look different each time I run it on the same data. Is this a problem?
Slight variations between runs on the same data are expected and typically not a problem [33]. This occurs because the Infomax ICA algorithm (runica) starts with a random weight matrix and randomly shuffles the data order in each training step, leading to slightly different convergence paths [33]. Features that do not remain stable across decompositions should not be interpreted. For a formal assessment of reliability, you can use the RELICA plugin for EEGLAB, which performs bootstrapping on the data [33].
4. What are the key differences between ICA and PCA for EEG analysis? While both are linear decomposition methods, they have fundamental differences [34]:
| Feature | Independent Component Analysis (ICA) | Principal Component Analysis (PCA) |
|---|---|---|
| Goal | Find maximally temporally independent sources [34]. | Find components that explain maximum variance [34]. |
| Spatial Constraint | Spatially unconstrained; components can have overlapping scalp maps [34]. | Spatially orthogonal; later components often look like "checkerboards" [34]. |
| Variance Accounted For | Component contributions are relatively homogeneous (e.g., ~5% down to ~0%) [34]. | First component accounts for most variance (e.g., 50%), with contributions dropping sharply [34]. |
5. Which ICA algorithm should I choose? Several algorithms are available, and their performance can be similar on clean, simulated data [34]. Here is a comparison of common options:
| Algorithm | Key Characteristics & Notes |
|---|---|
Infomax ICA (runica) |
Recommended and stable for up to hundreds of channels (with sufficient data). Uses a combination of higher-order moments. The default runica implementation in EEGLAB finds components with super-Gaussian distributions; use the 'extended' option to also detect sub-Gaussian sources (e.g., line noise) [34] [33]. |
Jader (jader) |
Uses 4th-order moments. Becomes impractical for datasets with more than ~50 channels due to massive storage requirements for the moment matrices [34]. |
FastICA (fastica) |
Can compute components one-by-one, but the order is not known in advance. A full decomposition is not necessarily faster than Infomax. May be less stable than Infomax for high-dimensional data [34]. |
| AMICA | Considered one of the most powerful algorithms currently available. Includes an integrated, iterative function to reject bad samples based on model log-likelihood during decomposition, which can improve results [35]. |
| SOBI | Good for data epochs. Available in the standard EEGLAB distribution [33]. |
Troubleshooting Guides
Problem: Poor or Unstable ICA Decomposition
Potential Causes and Solutions:
Insufficient Data:
- Symptom: Components do not represent clear physiological processes; decomposition fails or is highly variable.
- Solution: Ensure you have enough data points. Follow the
k*N²rule and consider increasing recording duration for high-density arrays [34].
Inadequate Data Cleaning Before ICA:
- Symptom: A few extreme artifacts (e.g., large transient spikes from electrode movement) dominate the decomposition.
- Solution: Apply moderate, automatic data cleaning before running ICA.
- Recommended Protocol: Use the built-in sample rejection of the AMICA algorithm. It rejects samples AMICA cannot easily account for, thus retaining artifacts that ICA itself can decompose (like blinks). A moderate cleaning strength (e.g., 5 to 10 iterations) is recommended for most datasets [35].
- General Workflow: Clean raw data → Run ICA on cleaned data → Apply the resulting ICA weights to the original, uncleaned data for final analysis [35].
Incorrect Data Referencing:
- Symptom: Poor component scalp topographies that are difficult to interpret.
- Solution: Re-reference the data appropriately. A study on functional connectivity found that using the Current Source Density (CSD) re-referencing method, in combination with ICA artifact reduction, provided the best results for detecting meaningful effects and high test-retest reliability [36].
Problem: Identifying and Classifying Independent Components
Methodology: Use a multi-faceted approach to classify components as brain-related or artifactual. The recommended workflow is to inspect the component's topography, time course, and power spectrum [33].
ICA Component Identification Workflow
Artifact Classification Criteria: The table below summarizes criteria for common artifact types. Use the "Plot → Inspect/label components by maps" function in EEGLAB for this analysis [33].
| Component Type | Scalp Topography | Time Course | Power Spectrum |
|---|---|---|---|
| Ocular (Blinks) | Strong, smooth frontal projection [33]. | Slow, large-amplitude deflections time-locked to blinks [33]. | Smoothly decreasing (typical of eye artifacts) [33]. |
| Muscle (EMG) | Focal, often over temporal muscles [22]. | High-frequency, irregular, "spiky" activity [34]. | Broadband, high-frequency power (above ~20 Hz) [22]. |
| Heart (ECG) | Maximal projection near neck/vessels. | Regular, pulsatile pattern (∼1.2 Hz) [22]. | Peak at heart rate frequency. |
| Neural (Brain) | Focal, bilateral, or patchy, following neuroanatomical and volume conduction principles [34]. | Oscillatory activity in known frequency bands (e.g., alpha, theta) [34]. | Peaks in classic EEG bands (e.g., Alpha: 8-13 Hz) [34] [22]. |
Problem: ICA Fails to Run or Converge
Potential Causes and Solutions:
Too Many Channels for the Data Length:
- Solution: If you have a high number of channels (N) but a short recording, use the
'pca'option in EEGLAB to reduce the dimensionality and find fewer than N components. This is often the only good option with insufficient data [33].
- Solution: If you have a high number of channels (N) but a short recording, use the
Data is Not Rank-Deficient:
- Symptom: Algorithm errors related to matrix rank.
- Solution: Remove the mean from each channel and from the data as a whole during preprocessing. EEGLAB's
pop_runicafunction typically handles this by default [33].
The Scientist's Toolkit: Key Research Reagents & Materials
| Item | Function in ICA for EEG |
|---|---|
| High-Density EEG System | Provides a sufficient number of spatial samples (channels) for ICA to effectively separate sources. A minimum of 32 channels is common, but 64+ is preferred for high-quality decompositions [35]. |
| EEGLAB | A collaborative, open-source MATLAB environment that serves as the primary platform for running and analyzing ICA decompositions. It provides a unified interface for multiple ICA algorithms and visualization tools [34] [33]. |
| ICA Algorithm (e.g., Infomax, AMICA) | The core computational engine that performs the blind source separation. The choice of algorithm (runica, amica, etc.) can impact the quality and interpretation of the components [34] [35]. |
| Channel Location File | A critical file that defines the spatial coordinates of each electrode on the scalp. This is essential for computing and visualizing the scalp topography of each independent component [33]. |
| Reference EOG/ECG Channels | While not strictly required for ICA, recording dedicated electrooculogram (EOG) and electrocardiogram (ECG) channels can provide valuable reference signals to help validate and confirm components identified as ocular and cardiac artifacts [22]. |
Table 1: ICA Algorithm Performance Comparison
| Algorithm | Decomposition Stability | Scalability (Channel Count) | Key Characteristic |
|---|---|---|---|
Infomax (runica) |
Stable with up to hundreds of channels [34]. | High [34]. | Recommended for general use; extended option handles sub-Gaussian sources [34] [33]. |
| AMICA | High; considered one of the most powerful [35]. | High [35]. | Includes iterative sample rejection for improved decomposition [35]. |
Jader (jader) |
Near-equivalent on low-dim. data [34]. | Low (>50 channels impractical) [34]. | Uses 4th-order moments; storage-intensive [34]. |
FastICA (fastica) |
Near-equivalent on low-dim. data; less stable for high-dim. data [34]. | Medium [34]. | Component order not known in advance [34]. |
Table 2: ICA Data Requirements (Rule of Thumb)
| Parameter | Guideline | Example (32 channels) |
|---|---|---|
| Minimum Data Points | > k × N² (k increases with N) [34]. | N² = 1024; k=30 → ~30,000 points [34]. |
| Effect of Insufficient Data | Unstable components; decomposition failure [34] [33]. | - |
| Recommended Action | Use PCA reduction if insufficient data [33]. | Use 'pca' option to find <32 components [33]. |
Wavelet Transform and Time-Frequency Domain Processing Techniques
Troubleshooting Guide & FAQs
This technical support resource addresses common challenges researchers face when applying wavelet transform and time-frequency techniques for artifact removal and noise filtering in EEG signals.
Frequently Asked Questions (FAQs)
Q1: Why is Wavelet Transform often preferred over traditional Fourier Transform for EEG artifact removal?
Wavelet Transform is superior to the traditional Fourier Transform for EEG analysis because of its good time-frequency localized properties and multi-resolution analysis capabilities [37]. Unlike Fourier Transform, which provides only global frequency-domain information, Wavelet Transform enables the extraction of transient information and can characterize both macroscopic patterns and microscopic fluctuations in non-stationary EEG signals effectively [37] [38]. This makes it particularly adept at handling sudden, short-duration artifacts like eye blinks while preserving the underlying neural activity.
Q2: How do I select the appropriate mother wavelet for my EEG denoising experiment?
Selecting the optimal mother wavelet often requires empirical testing, as its effectiveness can depend on the specific characteristics of your EEG data and the artifacts you are targeting. The core principle is to choose a wavelet whose shape closely matches the morphology of the signal features you wish to preserve or the artifacts you aim to remove. A common and practical approach is to begin testing with well-known wavelet families, such as Daubechies (dbN) or Symlets (symN), which are frequently used in EEG research due to their orthogonal properties and similarity to EEG waveforms [39] [40]. The quantitative performance metrics from your initial tests, such as those listed in the table below, will guide you toward the final selection.
Q3: What are the common pitfalls in setting the threshold for wavelet-based denoising?
A major pitfall is using a single, universal threshold that may remove valuable neural signals along with noise, leading to over-smoothing or loss of critical information. Artifacts and neural signals often occupy overlapping frequency bands, making separation difficult. To mitigate this, employ adaptive or level-dependent thresholding schemes. These methods apply different thresholds to the wavelet coefficients at different decomposition levels, allowing for a more nuanced removal of noise. Always validate the thresholding impact by comparing the time-series of the cleaned signal with the original and by checking if known neural signatures (like event-related potentials) are preserved post-processing.
Q4: My processed EEG signal appears distorted after wavelet denoising. What could be the cause?
Signal distortion typically arises from an overly aggressive denoising strategy. Key areas to investigate are:
- Excessive Decomposition Level: Decomposing the signal too deeply might remove not just noise but also clinically relevant low-frequency brain rhythms (e.g., delta or theta waves).
- Overly Strict Threshold: A threshold value that is too high will classify weak but genuine neural signals as noise and remove them.
- Mismatched Mother Wavelet: The chosen mother wavelet may be a poor fit for the morphology of the neural signals of interest in your experiment. We recommend a systematic approach: start with a conservative (lower) decomposition level and a milder threshold, then gradually adjust parameters while quantitatively monitoring performance metrics like Signal-to-Noise Ratio (SNR) and Percent Root mean square difference (PRD) to find an optimal balance.
Q5: Can wavelet transform be combined with other methods for better artifact removal?
Yes, hybrid methods that combine Wavelet Transform with other techniques often yield superior results and are an active area of research. A prominent example is Discrete Wavelet Transform based Scalar Quantization (DWTSQ), which hybridizes a wavelet transform with a quantizer to both denoise and compress EEG signals, improving SNR while reducing data size for efficient transmission [39]. Other common hybrid approaches include using Wavelet Transform with Independent Component Analysis (ICA), where wavelets perform initial denoising before ICA separates out independent source components, including artifacts [22] [41].
Experimental Protocols & Methodologies
Protocol 1: Basic Wavelet-Based Denoising of Ocular Artifacts
This protocol provides a step-by-step methodology for removing ocular artifacts (eye blinks and movements) from a single-channel EEG recording using Discrete Wavelet Transform (DWT).
1. Signal Acquisition and Input:
- Acquire the raw EEG signal. A noisy EEG signal with ocular artifacts can be modeled as
ns = S + AN, whereSis the clean EEG andANis the additive noise [39].
2. Wavelet Decomposition:
- Select a mother wavelet (e.g., Daubechies 'db4') and a decomposition level (e.g., 5-8 levels for EEG sampled at 250 Hz).
- Decompose the noisy signal
nsusing DWT to obtain approximation coefficients (cA) and detail coefficients (cD) at each level. This step separates the signal into different frequency sub-bands [39] [40].
3. Thresholding of Coefficients:
- Apply a thresholding function (e.g., soft thresholding) to the detail coefficients (cD) at each level. These coefficients often contain the high-frequency information associated with transient artifacts.
- The approximation coefficients (cA), which represent the signal's broad shape and lower frequencies, are typically left unmodified.
4. Signal Reconstruction:
- Reconstruct the EEG signal using the original approximation coefficients and the thresholded detail coefficients via the Inverse Discrete Wavelet Transform (IDWT).
- The output is a denoised version of the original EEG signal with ocular artifacts significantly reduced [40].
The following workflow diagram illustrates this process:
Wavelet Denoising Workflow
Protocol 2: Advanced Denoising and Compression using DWTSQ
For applications like Brain-Computer Interfaces (BCIs) that require efficient data transmission, this hybrid protocol denoises and compresses EEG signals.
1. Preprocessing and Decomposition:
- Follow Steps 1 and 2 of Protocol 1 to acquire the EEG signal and decompose it using DWT.
2. Scalar Quantization:
- The wavelet coefficients from decomposition are fed into a scalar quantizer.
- The quantizer approximates the continuous range of coefficient values with a finite set of discrete levels, reducing the number of bits needed to store or transmit the data [39].
- This step is a lossy compression that effectively removes fine detail noise.
3. Encoding and Transmission:
- The quantized values are further compressed using lossless coding methods like Huffman coding.
- For robust transmission, the data stream can be encoded with a Convolutional coder, modulated (e.g., using QAM), and sent through a channel (e.g., an AWGN channel) [39].
4. Reception and Decoding:
- On the receiver side, the signal is demodulated and decoded (e.g., using a Viterbi decoder appropriate for the convolutional code).
- The signal is then dequantized and reconstructed via the Inverse DWT to obtain the denoised EEG signal [39].
The following transceiver architecture illustrates this advanced system:
DWTSQ Transceiver Architecture
Performance Data & Benchmarking
The table below summarizes quantitative performance data from key studies and techniques to help researchers set realistic expectations and benchmark their own results.
Table 1: Performance Metrics of Wavelet-Based EEG Denoising Techniques
| Technique / Study | Key Metric | Reported Value / Outcome | Application Context |
|---|---|---|---|
| DWT with Thresholding [40] | Peak Signal-to-Noise Ratio (PSNR) | ~17.8 dB | Removal of eye-blink artifacts from recorded EEG. |
| DWTSQ Method [39] | Signal-to-Noise Ratio (SNR) | Increased SNR | General denoising and compression for BCI applications. |
| Immune Feature Weighted SVM (Post-DWTSQ) [39] | Classification Accuracy | Better accuracy for different EEG features | Feature classification after denoising. |
| Wavelet + Spiking Neural Networks (SpikeWavformer) [38] | Classification Accuracy | Superior performance & high energy efficiency | Emotion recognition and auditory attention decoding tasks. |
The Scientist's Toolkit: Research Reagent Solutions
This table details key computational tools and methodological "reagents" essential for implementing wavelet-based EEG analysis.
Table 2: Essential Research Reagents for Wavelet-Based EEG Analysis
| Item Name | Function / Purpose | Specifications / Examples |
|---|---|---|
| Mother Wavelet | The basis function for decomposition; its shape determines feature extraction efficacy. | Daubechies ('db4'), Symlets ('sym4'); chosen to match signal morphology [40]. |
| Discrete Wavelet Transform (DWT) | Decomposes the signal into frequency sub-bands for multi-resolution analysis. | Implemented via MATLAB wavedec function or Python pywt.wavedec. |
| Thresholding Function | Removes noise by shrinking small-magnitude wavelet coefficients. | Soft thresholding (smoother) or Hard thresholding; often level-dependent. |
| Inverse DWT (IDWT) | Reconstructs the time-domain signal from the (thresholded) wavelet coefficients. | The final step of the denoising pipeline (e.g., MATLAB waverec). |
| Performance Metrics | Quantifies the effectiveness of the denoising algorithm. | Signal-to-Noise Ratio (SNR), Peak SNR (PSNR), Percent Root mean square difference (PRD). |
Troubleshooting Guide: Frequently Asked Questions
This guide addresses common challenges researchers face when implementing deep learning models for EEG signal processing, specifically for artifact removal and noise filtering.
Model Architecture and Selection
Q1: My CNN-LSTM model for EEG artifact removal is not converging. What could be the issue?
A1: Non-convergence in CNN-LSTM models often stems from data preprocessing or architectural configuration problems. Ensure you have properly normalized your EEG and reference EMG signals, as was done in the hybrid CNN-LSTM approach for muscle artifact removal where simultaneous facial and neck EMG recordings were essential for success [42]. Consider these specific checks:
- Input Normalization: Apply z-score or min-max scaling to all input channels (both EEG and any auxiliary signals like EMG) to ensure stable gradient propagation.
- Reference Signals: Verify the quality and synchronization of any reference signals (e.g., EMG) used to guide the artifact removal process. The hybrid CNN-LSTM approach relies on these additional recordings for precise artifact elimination [42].
- Temporal Context: Ensure your LSTM layers have sufficient sequence length to capture the temporal dynamics of both artifacts and neural signals. Studies suggest that inadequate sequence length can fail to model long-range dependencies in EEG [43] [44].
Q2: When should I choose a State Space Model (SSM) over a CNN-LSTM for EEG denoising?
A2: The choice depends on the nature of the artifact and computational constraints. Recent benchmarks indicate that while CNNs excel with certain artifacts, SSMs show superior performance for others [15].
- CNN-LSTM: Ideal for artifacts with strong spatial and temporal patterns that can be learned from data, such as muscle artifacts (EMG) when combined with reference signals [42]. It effectively combines spatial filtering (CNN) with temporal modeling (LSTM).
- State Space Models (SSMs): Particularly effective for handling long-range dependencies and specific artifact types like those from transcranial Alternating Current Stimulation (tACS) and transcranial Random Noise Stimulation (tRNS). A 2025 study found that a multi-modular SSM network (M4) outperformed other methods, including CNNs, for these complex, oscillatory artifacts [15]. SSMs also offer computational efficiency advantages for long sequences compared to the quadratic complexity of Transformers [45].
The following table summarizes the performance of different architectures on various artifact types based on recent research:
Table 1: Performance Comparison of Deep Learning Architectures on EEG Artifact Removal
| Architecture | Best For Artifact Type | Key Strength | Reported Performance Metric |
|---|---|---|---|
| CNN-LSTM with EMG reference [42] | Muscle artifacts (EMG) | Utilizes additional biosignal recording for precise removal | Effectively preserves SSVEP responses while removing artifacts |
| Complex CNN [15] | tDCS artifacts | Spatial feature extraction | Best RRMSE* performance for tDCS artifacts |
| Multi-modular SSM (M4) [15] | tACS, tRNS artifacts | Modeling long-range dependencies, computational efficiency | Best RRMSE* performance for tACS and tRNS artifacts |
| DWT-CNN-BiGRU [31] | General noise filtering for classification | Combines wavelet denoising with spatial-temporal learning | 94% accuracy in alcoholic vs. control subject classification |
| RRMSE: Root Relative Mean Squared Error, a common metric for denoising performance. |
Data Quality and Preprocessing
Q3: How can I improve my model's generalization when I have limited labeled EEG data?
A3: Data augmentation is crucial for training robust deep learning models with limited data. Several effective techniques have been demonstrated in recent literature:
- Noise Injection: Adding controlled random noise to training samples to improve model robustness [46].
- Conditional Variational Autoencoders (cVAE): Generating synthetic EEG data that retains the statistical properties of the original dataset [46].
- Advanced Generative Models: Using methods like conditional GAN with Wasserstein loss and gradient penalty (cWGAN-GP) to create high-quality synthetic EEG epochs [46]. One study on Motor Imagery classification found that such augmentation methods helped a CNN-LSTM model achieve 79.06% accuracy [46].
Q4: What is the most effective preprocessing pipeline for EEG before feeding it into a deep learning model?
A4: A robust preprocessing pipeline is foundational. The exact steps can vary, but the following workflow, synthesized from multiple studies, covers essential stages for artifact removal tasks:
Table 2: Essential Research Reagent Solutions for EEG Deep Learning Experiments
| Item / Technique | Function in Experiment | Application Example |
|---|---|---|
| High-density EEG System (e.g., 64-electrode setup) | Captures spatial distribution of brain electrical activity and artifacts. | Used in alcoholic/control classification studies to provide sufficient spatial information for CNN layers [31]. |
| Auxiliary Biosignal Amplifiers (EMG, EOG) | Records reference signals for specific artifacts (muscle, eye movement). | Critical for the hybrid CNN-LSTM approach to muscle artifact removal, providing the noise reference [42]. |
| Discrete Wavelet Transform (DWT) | Preprocessing technique for noise filtering and signal denoising. | Achieved the highest accuracy (94%) when used before a CNN-BiGRU model for classifying alcoholic subjects [31]. |
| Semi-synthetic Datasets | Enables controlled evaluation by adding known artifacts to clean EEG. | Used to benchmark 11 artifact removal techniques for tES artifacts, providing a known ground truth [15]. |
| Genetic Algorithm (GA) | Optimizes model hyperparameters and selects relevant multimodal features. | Used to optimize CNN-LSTM architecture and feature selection for coma outcome prediction, enhancing performance [43]. |
Diagram 1: Standard EEG Preprocessing Workflow
Training and Optimization
Q5: How do I balance artifact removal with the preservation of neurologically relevant signals in my model?
A5: This is a core challenge in EEG denoising. Implement a loss function or validation strategy that explicitly measures the preservation of neural information.
- Targeted Validation Metrics: Use metrics relevant to your end goal. For instance, in a study involving Steady-State Visually Evoked Potentials (SSVEP), the increase in Signal-to-Noise Ratio (SNR) of the SSVEP response after cleaning was used as a key performance indicator, ensuring the neural response of interest was preserved [42].
- Structured Training Data: Train your model using data where the neural signal of interest is present (e.g., during visual stimulation for SSVEP) and absent (e.g., during rest or artifact-only periods). This helps the model learn to discriminate between noise and signal [42].
- Hybrid Loss Functions: Combine a loss term for artifact reduction (e.g., Mean Squared Error with a clean target) with a term that penalizes the distortion of known neural features.
Q6: What strategies can I use to optimize the hyperparameters of a complex CNN-LSTM-SSM architecture?
A6: Leverage automated search strategies, as manual tuning for such complex models is inefficient.
- Genetic Algorithms (GAs): GAs are highly effective for joint optimization of architecture and hyperparameters. One study proposed a GA to select relevant multimodal features and optimize CNN-LSTM hyperparameters simultaneously, leading to improved performance for coma outcome prediction [43]. The GA can explore a vast search space of layer sizes, learning rates, and feature subsets.
- Structured Search Spaces: Define your search space logically. For a hybrid model, you might search over the number of CNN filters, LSTM units, the presence or type of SSM layer, and the fusion method for multimodal data.
Performance and Evaluation
Q7: What are the key quantitative metrics to report when evaluating an EEG artifact removal model?
A7: A comprehensive evaluation should include metrics in both time and frequency domains to assess different aspects of performance.
- Temporal Domain Fidelity: Use Root Relative Mean Squared Error (RRMSE) and Correlation Coefficient (CC) between the cleaned signal and a ground-truth clean signal. These were used in a 2025 benchmark of tES artifact removal [15].
- Spectral Domain Preservation: Calculate the RRMSE in the frequency domain to ensure oscillatory components are not distorted [15].
- Task-Specific Performance: If the cleaned EEG is for a downstream task (e.g., classification), report the task's performance metrics (e.g., accuracy, F1-score). For instance, classification accuracy was the final metric for evaluating the DWT-CNN-BiGRU model [31].
The following diagram illustrates a complete experimental workflow for developing and evaluating an EEG denoising model, integrating the key steps discussed in this guide:
Diagram 2: End-to-End Experimental Workflow for EEG Denoising
Frequently Asked Questions (FAQs)
Q1: What is a hybrid methodology in the context of EEG artifact removal? A hybrid methodology combines two or more signal processing techniques to leverage their individual strengths and overcome their limitations. For instance, a framework might integrate Fixed Frequency Empirical Wavelet Transform (FF-EWT) for decomposing a single-channel EEG signal with a Generalized Moreau Envelope Total Variation (GMETV) filter to remove EOG artifacts. This approach is more effective at isolating and removing artifacts while preserving the underlying neural information compared to using any single method alone [47].
Q2: Why does muscle fatigue degrade EMG-based intention detection in rehabilitation robots, and how can a hybrid system help? Muscle fatigue alters the spectral properties of EMG signals (a peripheral measure), leading to significant degradation in classification performance for rehabilitation robots. A hybrid EMG-EEG system addresses this by adaptively fusing the EMG signals with EEG (a central measure). A real-time fatigue estimator can dynamically adjust the contribution of each modality within a Bayesian fusion framework. As fatigue increases, the system weights the more stable EEG signals more heavily, thereby maintaining robust detection of user intention throughout prolonged rehabilitation sessions [48].
Q3: My cross-session EEG classification accuracy is poor. What feature engineering approach can improve generalizability? Poor cross-session performance often stems from features that are not robust to session-to-session variability. A hybrid feature learning framework that integrates multiple feature types can significantly enhance generalizability. You should extract not only standard channel-wise spectral features (e.g., using Short-Time Fourier Transform) but also functional and structural connectivity features (e.g., Phase Locking Value) to capture inter-regional brain interactions. Implementing a two-stage feature selection process (e.g., correlation-based filtering followed by random forest ranking) will further refine the feature set, leading to substantially improved cross-session and inter-subject classification accuracy [49].
Q4: What is the advantage of using a knowledge transfer approach in a hybrid CNN model for EEG Motor Imagery (MI) classification? A hybrid CNN that uses knowledge transfer addresses the challenge of inter-subject variability, which often degrades the performance of subject-independent models. For example, the AMD-KT2D framework employs a guide-learner architecture. A pre-trained guide model (e.g., on a large-scale dataset) extracts high-level spatial-temporal features. These features are then transferred to a learner model, with an Adaptive Margin Disparity Discrepancy (AMDD) loss function ensuring feature alignment. This process reduces domain disparity, allowing the model to generalize effectively to new subjects without needing extensive retraining [50].
Troubleshooting Guides
Issue 1: Persistent Ocular (EOG) Artifacts in Single-Channel EEG Recordings
Problem: Eye-blink artifacts are contaminating your single-channel EEG data, and traditional filters are removing crucial low-frequency brain signals along with the artifact.
Solution: Implement an automated decomposition and filtering pipeline.
Methodology:
- Decomposition: Use Fixed Frequency Empirical Wavelet Transform (FF-EWT) to adaptively decompose the contaminated single-channel EEG signal into six Intrinsic Mode Functions (IMFs) based on the signal's spectral characteristics [47].
- Identification: Automatically identify artifact-related IMFs using a combination of metrics like Kurtosis (KS), Dispersion Entropy (DisEn), and Power Spectral Density (PSD). EOG artifacts typically have high amplitude and are concentrated in low frequencies [47].
- Filtering: Apply a Generalized Moreau Envelope Total Variation (GMETV) filter specifically to the contaminated IMFs. This filter is finely tuned to suppress the artifact while preserving the signal's integrity [47].
- Reconstruction: Reconstruct the clean EEG signal from the processed IMFs.
Verification: Validate the method on a segment of data with known artifacts. A successful removal will show a high Signal-to-Artifact Ratio (SAR) and a low Mean Absolute Error (MAE) on real EEG recordings, and a low Relative Root Mean Square Error (RRMSE) on synthetic data [47].
Issue 2: Performance Degradation in Hybrid EMG-EEG Intention Detection Due to Fatigue
Problem: Your hybrid rehabilitation system's classification accuracy drops during prolonged use as the user's muscles become fatigued, corrupting the EMG signals.
Solution: Integrate a real-time, fatigue-aware adaptive fusion mechanism.
Methodology:
- Fatigue Estimation: In real-time, extract spectral features (e.g., median frequency) from the EMG signals. Input these features into a k-Nearest Neighbors (k-NN) model that has been pre-trained to output a continuous fatigue score,
f(x), between 0 (no fatigue) and 1 (high fatigue) [48]. - Unimodal Classification: Simultaneously, run independent classifiers on the EMG and EEG streams. A Support Vector Machine (SVM) is often used for EMG, while a Common Spatial Pattern (CSP) with SVM is common for EEG [48].
- Adaptive Bayesian Fusion: Fuse the probability outputs from the two classifiers using a Bayesian framework. Critically, modulate the fusion weight
αdynamically based on the fatigue score [48]:α(f) = 0.2 + 0.6 * f(x)This formula ensures that as fatiguef(x)increases, the system relies more heavily on the more stable EEG signals to maintain accurate intention detection.
Verification: Compare the classification accuracy of the adaptive system against a static fusion baseline (e.g., a simple average) during a prolonged experiment. The adaptive system should show significantly less performance drop in high-fatigue conditions [48].
Issue 3: Low Accuracy in Cross-Session Mental Attention State Classification
Problem: Your model, trained on EEG data from one session, fails to generalize to new recording sessions from the same subject, likely due to non-stationary EEG signals.
Solution: Adopt a hybrid feature learning and selection framework.
Methodology:
- Hybrid Feature Extraction: Move beyond a single type of feature. Extract a comprehensive set of features:
- Spectral Features: Compute power in standard frequency bands (Delta, Theta, Alpha, Beta) using STFT [49] [22].
- Connectivity Features: Calculate both functional (e.g., coherence) and effective connectivity (e.g., Phase Locking Value) between different brain regions to capture the network dynamics of attention [49].
- Two-Stage Feature Selection: To avoid overfitting and reduce dimensionality, use a rigorous selection process:
- Stage 1 (Filtering): Remove features with very high inter-feature correlations.
- Stage 2 (Wrapper): Use a Random Forest algorithm to rank the remaining features by their importance and select the top-performing subset [49].
- Classification: Train a classifier such as a Support Vector Machine (SVM) on this robust, selected feature set for final mental state classification (e.g., focused, unfocused, drowsy) [49].
Verification: Evaluate the model using a cross-validation strategy where the test set is from a different session than the training set. This framework has been shown to achieve high accuracy (e.g., over 86%) in such challenging cross-session scenarios [49].
Experimental Protocols & Data
Table 1: Performance Comparison of Artifact Removal Techniques
This table summarizes key metrics for evaluating the effectiveness of different artifact removal methods on synthetic and real EEG data.
| Method | Relative Root Mean Square Error (RRMSE) | Correlation Coefficient (CC) | Signal-to-Artifact Ratio (SAR) | Mean Absolute Error (MAE) |
|---|---|---|---|---|
| FF-EWT + GMETV (Proposed) | Lower | Higher | Improved | Lower [47] |
| Empirical Mode Decomposition (EMD) | Higher | Lower | Less Improved | Higher [47] |
| Singular Spectrum Analysis (SSA) | Moderate | Moderate | Moderate | Moderate [47] |
| Independent Component Analysis (ICA) | Not Effective for SCL | Not Effective for SCL | Not Effective for SCL | Not Effective for SCL [47] |
Table 2: Hybrid vs. Unimodal Classification Performance
This table compares the classification accuracy of hybrid systems against unimodal systems under different conditions.
| System Configuration | Condition | Classification Accuracy | Key Parameters |
|---|---|---|---|
| Hybrid EMG-EEG with Adaptive Fusion [48] | Normal Operation | 94.5% | Bayesian fusion, Fatigue-aware weighting |
| EMG-Only Baseline [48] | Normal Operation | 88.5% | SVM classifier |
| Hybrid EMG-EEG with Adaptive Fusion [48] | High Fatigue | 91.4% | Bayesian fusion, Fatigue-aware weighting |
| EMG-Only Baseline [48] | High Fatigue | 83.1% | SVM classifier |
| Hybrid Feature Learning (EEG) [49] | Cross-Session | 86.27% (Dataset 1) | STFT + Connectivity features, Two-stage selection |
| Hybrid Feature Learning (EEG) [49] | Cross-Session | 94.01% (Dataset 2) | STFT + Connectivity features, Two-stage selection |
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item Name | Function / Application in Research |
|---|---|
| Emotiv Epoc Flex System | A 32-channel saline-based EEG headset used for acquiring motor imagery (MI) signals. Its compatibility with the 10-20 system allows for consistent electrode placement across studies [50]. |
| OpenBCI Headset | A popular, accessible platform for acquiring multi-channel EEG data, often used in hybrid BMI systems for capturing signals from the motor cortex [48]. |
| Surface EMG (sEMG) Armband | A wearable device used to capture electrophysiological signals from muscles (e.g., biceps, triceps) for intention detection in rehabilitation robotics [48]. |
| Support Vector Machine (SVM) | A robust classifier frequently employed in both EEG and EMG pipelines due to its efficiency and strong generalization performance with physiological signals [49] [48]. |
| Common Spatial Pattern (CSP) | A spatial filtering algorithm essential for enhancing the signal-to-noise ratio of EEG data before motor imagery classification [48]. |
| Fixed Frequency EWT (FF-EWT) | A signal decomposition technique used to automatically break down a single-channel EEG signal into components for targeted artifact removal [47]. |
Experimental Workflow Diagrams
https://www.nature.com/articles/s41598-025-10276-8
https://www.nature.com/articles/s41598-025-24831-w
Real-time Artifact Removal Solutions for Clinical and Research Settings
Frequently Asked Questions (FAQs)
Q: What is the primary challenge with real-time EEG artifact removal? A: The core challenge lies in the low signal-to-noise ratio of EEG signals, which are measured in microvolts and are highly susceptible to contamination from various sources. Effective removal must be achieved without losing meaningful neural data, and in real-time scenarios, this must be accomplished with minimal processing delay, often under 250 ms, to be practical for applications like neurofeedback or BCI [1] [51].
Q: Which artifact removal method is best for my real-time application? A: The "best" method depends on your specific requirements for accuracy, speed, and hardware setup. There is no single optimal solution for all scenarios [52]. The table below summarizes the pros and cons of common real-time suitable methods.
| Method | Best For | Key Advantage | Key Limitation |
|---|---|---|---|
| Artifact Subspace Reconstruction (ASR) | Real-time BCI, scenarios with large artifacts [53] | Fast, works well even in real-time applications [53] | Requires multi-channel EEG data [11] |
| Regression-Based Methods | Scenarios with available EOG/ECG reference channels [11] [22] | Simple, computationally efficient [11] | Requires additional reference channels, risks over-correction [22] [52] |
| Adaptive Filtering | Applications where a clean reference signal is available [52] | Dynamically adjusts to changing signal and noise properties [11] | Performance depends entirely on the quality of the reference signal [52] |
| Deep Learning (e.g., LSTM) | Complex artifacts in EEG-fMRI setups [51] | Can learn complex noise patterns; no need for reference signals [54] [6] | Requires large datasets for training and significant computational resources [54] |
Q: Can I perform real-time artifact removal without extra reference channels (EOG/ECG)? A: Yes. Methods like Artifact Subspace Reconstruction (ASR) and some deep learning models are designed to work without separate EOG or ECG channels. They identify artifacts based on the statistical properties of the multi-channel EEG data itself or from pre-learned models of neural and artifactual signals [11] [54] [53].
Q: How do I validate the performance of a real-time artifact removal algorithm? A: Validation can be challenging due to the lack of a "ground truth" clean EEG signal. Common strategies include:
- Semi-Simulation: Adding known artifacts to clean EEG segments and evaluating the algorithm's ability to remove them [54] [6].
- Performance Metrics: Using quantitative metrics like Signal-to-Noise Ratio (SNR), Signal-to-Artifact Ratio (SAR), and Root Mean Square Error (RMSE) on processed data [6].
- Downstream Task Accuracy: For BCI, the most critical validation is whether the artifact removal improves the classification accuracy of intended commands [52].
Troubleshooting Guides
Problem: Poor Performance After Real-Time Ocular Artifact Removal
Symptoms: Attenuated brain signals in frontal channels, distorted Event-Related Potentials (ERPs), or introduction of new noise.
| Potential Cause | Solution | Underlying Principle |
|---|---|---|
| Over-correction by regression method | Switch to a component-based method like ICA or ASR if computationally feasible. | Regression assumes a linear relationship and can remove neural activity that is correlated with the artifact [22] [52]. |
| Inaccurate artifact template | Ensure the calibration run for template creation uses spontaneous, natural blinks rather than forced ones. | Spontaneous and imposed eye movements are physiologically different, leading to an inaccurate template [11]. |
| Prolonged processing delay | For BCI, simplify the algorithm (e.g., use a filter with a shorter window) to meet real-time constraints. | The algorithm's processing time may exceed the system's maximum allowable latency, making it impractical [52]. |
Problem: Algorithm Not Keeping Up with Real-Time Data Stream
Symptoms: Data buffer overflows, system lag, or dropped data packets.
| Potential Cause | Solution | Underlying Principle |
|---|---|---|
| Computationally expensive algorithm | Choose a more efficient method (e.g., ASR or adaptive filtering) over more complex ones (e.g., some deep learning models) for resource-constrained systems. | Deep learning models like CNNs and RNNs require significant computational power which can cause delays [54]. |
| Inefficient code implementation | Use optimized, compiled libraries (e.g., in C++ or via MNE-Python's optimized functions) rather than pure interpreted code. | The mathematical implementation and programming language directly impact processing speed. |
| Insufficient hardware resources | Allocate a dedicated processing unit for the artifact removal algorithm to ensure consistent performance. | Other processes running on the same machine can consume CPU cycles and memory, starving the real-time algorithm. |
Guide to Method Selection for Real-Time Applications
This diagram outlines a logical workflow for selecting an appropriate real-time artifact removal method based on your experimental constraints and goals.
Experimental Protocols & Methodologies
Protocol 1: Real-Time Ocular Artifact Correction using Regression
This protocol is adapted from the Gratton and Cole algorithm, a time-domain regression method [11].
1. Preprocessing:
- Apply a band-pass filter (e.g., 1–50 Hz) to the raw EEG to eliminate slow drifts and high-frequency noise.
- Apply a low-pass filter (cut-off at 15 Hz) to the EOG reference signal.
2. Calibration Phase (to estimate regression coefficients):
- Collect a segment of data where the participant engages in spontaneous blinking.
- For each EEG channel, calculate the transmission coefficient (β) that defines the linear relationship between the EOG signal and the artifact in that EEG channel.
3. Correction Phase (real-time execution):
- For each new data sample, subtract the scaled EOG artifact from each EEG channel using the formula:
Corrected_EEGᵢ(t) = Raw_EEGᵢ(t) - βᵢ * EOG(t)- Where
irepresents the i-th EEG channel.
Protocol 2: Real-Time Artifact Removal with Artifact Subspace Reconstruction (ASR)
ASR is an advanced technique that detects and reconstructs portions of the data contaminated by artifacts [11] [53].
1. Calibration Phase:
- Record a short segment (1-2 minutes) of clean, resting-state EEG data from the participant. This "calibration data" represents the normal, non-artifactual brain activity for that specific subject and setup.
- Use this data to compute a covariance matrix that defines the "clean" EEG subspace.
2. Processing Phase (real-time execution):
- For each new data chunk (e.g., a short time window), the algorithm:
- Detects: Compares the statistical properties of the incoming data to the calibrated "clean" subspace.
- Reconstructs: Identifies data channels and time points that exceed a threshold deviation from the clean subspace, flagging them as artifacts.
- Corrects: Replaces the identified artifact-contaminated segments by reconstructing them based on the clean data from other channels and time points.
Protocol 3: Real-Time Artifact Handling in EEG-fMRI using NeuXus
This protocol uses the open-source NeuXus toolbox to handle severe gradient and pulse artifacts in simultaneous EEG-fMRI recordings [51].
1. Setup:
- Ensure ECG is recorded alongside EEG for optimal pulse artifact correction.
2. Real-Time Processing Pipeline:
- Gradient Artifact Removal: Uses Average Artifact Subtraction (AAS). The system maintains a running average of the artifact template, synchronized with the MR scanner's volume triggers, and subtracts it from the EEG signal.
- Pulse Artifact Removal: Employs a Long Short-Term Memory (LSTM) network for robust R-peak detection in the ECG signal. Once detected, a similar average subtraction method is used to remove the pulse artifact from the EEG.
- This combined approach has been validated to execute in under 250 ms, making it suitable for real-time neurofeedback [51].
The Scientist's Toolkit: Essential Software & Tools
| Tool Name | Type / Function | Key Features & Applications |
|---|---|---|
| NeuXus [51] | Open-source Python toolbox | Specialized for real-time artifact reduction in EEG-fMRI; uses LSTM for R-peak detection; hardware-independent. |
| ASR (in EEGLAB) [53] | Plug-in / Algorithm | Real-time artifact removal; effective for large-amplified artifacts in BCI; works on multi-channel data. |
| ICA (in MNE-Python, EEGLAB) | Algorithm / Built-in function | Excellent for ocular and persistent muscle artifact removal; separates data into independent components for manual/automatic rejection. |
| LSTM Networks [51] [6] | Deep Learning Architecture | Models temporal dependencies; effective for complex artifacts like pulse in EEG-fMRI or in generative models (GANs). |
| GANs with LSTM [6] | Deep Learning Architecture | Generates artifact-free EEG signals; the generator creates clean data, while the discriminator judges its quality. |
| DWT-CNN-BiGRU Model [31] | Hybrid Deep Learning Model | DWT for noise filtering; CNN for spatial features; BiGRU for temporal dependencies; high classification accuracy post-denoising. |
Optimizing EEG Data Quality: Practical Solutions for Common Artifact Challenges
Frequently Asked Questions (FAQs) on EEG Artifact Prevention
Why is proactive artifact minimization more effective than post-hoc removal? While many advanced techniques exist to remove artifacts from recorded data, they all have limitations. Proactive design prevents the contamination of neural data from the outset. Artifact rejection reduces the number of trials available for analysis, which can be detrimental to decoding performance, and correction methods may not fully eliminate all confounds, sometimes even artificially inflating results [55]. Preventing artifacts at the source preserves more data and yields a cleaner, more reliable signal.
What are the most common physiological artifacts and their characteristics? The primary physiological artifacts originate from the participant's own body. Understanding their properties is the first step in preventing them. The table below summarizes the key artifacts you must account for in your experimental design [22].
Table 1: Common Physiological Artifacts in EEG Recordings
| Artifact Type | Primary Source | Frequency Range | Key Characteristics |
|---|---|---|---|
| Ocular Artifacts | Eye blinks and movements | Similar to EEG (low frequency) | Very high amplitude; can be many times greater than neural signals [22]. |
| Muscle Artifacts (EMG) | Head, jaw, neck muscle activity | 0 Hz to >200 Hz [22] | Broad frequency distribution; particularly challenging to remove due to overlapping frequencies with neural signals [6]. |
| Cardiac Artifacts | Heartbeat (ECG and pulse) | ~1.2 Hz (pulse) [22] | Regular, repeating pattern; can be introduced via electrodes placed near blood vessels. |
How can I minimize ocular artifacts in my paradigm? Ocular artifacts are among the most common and significant contaminants. Key strategies include:
- Instruction and Fixation: Provide clear, simple instructions for participants. Use a centralized fixation cross on the screen to discourage eye movements during critical trial periods.
- Paradigm Timing: Design your stimulus presentation to include intervals where visual stimuli are absent, giving participants natural breaks to blink without losing data. For example, instruct participants to blink during the inter-trial interval.
- Subject Selection and Habituation: When possible, screen for participants who can comfortably comply with the task requirements. A brief practice session allows them to habituate to the environment and task demands.
What experimental design choices can reduce muscle artifacts? Muscle artifacts are often related to participant movement and tension.
- Comfortable Setup: Ensure the participant's chair is comfortable and they are in a relaxed, supported posture to minimize neck and back strain.
- Minimize Verbal Responses: Where possible, use button presses instead of verbal responses to prevent jaw and face muscle activity.
- Breaks: Incorporate regular, mandatory breaks to prevent fatigue and fidgeting.
How does electrode choice and placement affect data quality? Proper setup is a critical line of defense.
- Electrode Type: Select high-quality, non-irritating electrodes. Ensure they are clean and applied according to manufacturer specifications.
- Skin Preparation: Properly prepare the scalp by cleaning to reduce impedance. High impedance can increase sensitivity to environmental noise and artifacts.
- Secure Placement: Ensure all electrodes are securely attached and wires are fastened to prevent movement during the recording.
Experimental Protocols for Artifact Prevention
The following workflow provides a visual summary of a comprehensive, proactive strategy for minimizing artifacts, from participant preparation to paradigm design.
Diagram 1: Proactive artifact minimization workflow.
Detailed Methodology for a Low-Artifact EEG Study
This protocol outlines the specific steps to implement the proactive design strategy shown in the workflow.
1. Participant Briefing and Preparation
- Informed Consent: Begin with a clear explanation of the study procedures, emphasizing the importance of minimizing movement.
- Instruction Script: Read from a standardized script: "During the experiment, please remain as still as possible. You will see a fixation cross in the center of the screen. Try to minimize your eye movements and blinks while the stimuli are presented. Feel free to blink naturally during the breaks between trials."
- Comfort Adjustment: Adjust the chair and any supports to ensure the participant is comfortable for the duration of the session.
2. Hardware and Electrode Setup
- Skin Preparation: Clean the scalp at each electrode site with a mild abrasive gel or solution until the impedance is consistently below 10 kΩ.
- Electrode Application: Apply electrodes using conductive paste, ensuring a firm connection. Use a cap or tape to secure wires and prevent cable sway.
- Signal Check: Before starting the experiment, visually inspect the live EEG signal to check for obvious artifacts from tension, blinking, or loose electrodes.
3. Experimental Paradigm Programming
- Fixation: Program your experiment to display a clear fixation cross (e.g., "+") in the center of the screen throughout all trial periods.
- Trial Structure: Design trials with a clear structure: a pre-stimulus baseline period, the stimulus presentation, and a post-stimulus response period, followed by an inter-trial interval (ITI) of 1-2 seconds. The ITI is the designated blink period.
- Response Method: Configure the task to use a button box or keyboard for participant responses instead of verbal answers.
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential materials and solutions for ensuring high-quality, low-artifact EEG data collection.
Table 2: Essential Materials for High-Quality EEG Research
| Item Name | Function / Purpose | Key Consideration |
|---|---|---|
| Abrasive Skin Prep Gel | Reduces impedance by removing dead skin cells and oils at the electrode site. | Essential for achieving stable impedances below 10 kΩ, which reduces environmental noise. |
| High-Quality Electrolyte Gel | Facilitates stable electrical conduction between the scalp and the electrode. | Prevents signal drift and maintains a stable connection throughout the recording session. |
| Electrode Cap (Ag/AgCl) | Holds electrodes in standardized positions (10-20 system). | A well-fitted cap ensures consistent electrode placement across participants and minimizes movement. |
| Chinstrap | Gently stabilizes the jaw and reduces muscle artifacts from head movement. | Particularly useful in long recordings or with populations that may have difficulty remaining still. |
| Button Box Response Unit | Allows for participant input without introducing verbal or large motor artifacts. | Prevents the muscle artifacts associated with speaking or using a standard keyboard. |
| Stimulus Presentation Software | Precisely controls the timing and presentation of visual/auditory stimuli. | Must allow for precise millisecond timing and the programming of fixation crosses and break screens. |
Frequently Asked Questions (FAQs)
What is the primary goal of maintaining low and stable electrode impedance? The primary goal is to ensure high-quality EEG data acquisition by maximizing the signal-to-noise ratio. Low impedance provides a clearer path for the brain's electrical signals to reach the recording equipment, while stable impedance prevents signal drift during extended recordings, which is crucial for both short-term experiments and long-term monitoring. [56] [57] [58]
Why is high impedance problematic in EEG recordings? High impedance acts as a barrier to the weak electrical signals generated by the brain. This leads to increased signal attenuation and a poor signal-to-noise ratio, making the recorded data more susceptible to contamination from environmental noise, such as 50/60 Hz power line interference. [57] Furthermore, at high frequencies, the signal can be shunted away from the amplifier through stray capacitances that form in the wiring, severely degrading data quality. [57]
How does electrode technology impact impedance and stability? The choice of electrode technology directly defines the initial impedance and its stability over time.
- Traditional Wet Electrodes: Require conductive gel and skin preparation to achieve very low impedance. However, impedance can change significantly as the gel dries, leading to signal instability during long recordings. [59] [58]
- Dry Electrodes: Eliminate the need for gel, offering greater comfort and quicker setup. Their impedance is inherently higher, but modern designs use ultra-high impedance amplifiers (e.g., >47 GΩ) to overcome this, making signal quality comparable to wet electrodes without the risk of gel drying. [58]
- Advanced Hydrogel & Hairlike Electrodes: New form factors, like 3D-printed hydrogel electrodes, use bioadhesive ink to stick directly to the scalp. This minimizes the electrode-skin gap, improving signal quality and maintaining stable contact for over 24 hours of continuous wear without degradation. [56]
What are the consequences of unstable impedance during longitudinal studies? Unstable impedance introduces unwanted variability in EEG measures over time, which can be mistaken for genuine neural changes. Research has shown that both linear (e.g., band powers) and nonlinear EEG measures require high test-retest reliability to be useful in longitudinal research. Impedance instability can mask the excellent long-term stability these measures can exhibit in stable recording conditions, potentially invalidating study results. [60]
Troubleshooting Guides
Problem: Persistent High-Frequency Noise and Unstable Baseline
Possible Cause: Stray capacitance and inductance in cabling, or environmental noise pickup.
Solution:
- Inspect Cables: Check for long, unguarded wires, weak contacts, or corroded connectors. Even a simple 10 cm wire can have ~0.98 pF of stray capacitance, which shunts high-frequency signals. [57]
- Minimize Lead Length: Use the shortest cables possible and ensure they are securely connected. Adding a 4 mm banana plug to a 10 cm wire can increase stray capacitance from 0.98 pF to 2.1 pF. [57]
- Use a Faraday Cage: Place the subject and setup in an earthed Faraday cage to block external electromagnetic noise, especially 50/60 Hz power line interference. Be aware that this also increases capacitance to the ground. [57]
- Twist Cables: For low-impedance samples, twist current-carrying leads together to minimize the loop area and reduce magnetic coupling, which can manifest as a fake inductive component in the measurements. [57]
Problem: Rapid Signal Quality Degradation During Long-Term Recordings
Possible Cause: Traditional wet electrode gel drying out, or physical movement of the electrode.
Solution:
- Consider Dry Electrodes: Switch to dry electrode systems, which are specifically designed for long-term stability as they do not rely on hydrating gels. Studies show they maintain stable performance over 4-8 hours. [58]
- Use Advanced Hydrogel Adhesives: For applications requiring direct skin contact, 3D-printed hydrogel electrodes with bioadhesive ink have demonstrated stable performance for over 24 hours of continuous wear. [56]
- Ensure Secure Mounting: Use electrode caps or headbands that provide a secure and comfortable fit. Patented mechanical isolation designs in some dry electrodes help stabilize them against movement artifacts. [58]
Problem: Variable Data Quality Across Sessions in a Longitudinal Study
Possible Cause: Inconsistent electrode application placement or impedance between sessions.
Solution:
- Standardize Application Protocol: Create a detailed, step-by-step protocol for electrode placement to ensure consistency across different operators and sessions. [60]
- Document Impedance Values: Record the impedance values for each electrode at the start of every recording session. This creates a quality control log that can be correlated with data quality.
- Use Semi-Permanent Electrodes: For critical long-term studies, consider novel electrodes that do not require removal and replacement between sessions. Research shows that hairlike hydrogel electrodes can be left in place, eliminating the risk of inconsistent data due to positional shifts or changing impedance across sessions. [56]
Experimental Protocols & Data
Table 1: Quantitative Comparison of Electrode Technologies
| Electrode Type | Typical Initial Impedance | Stability Over Time | Setup Time | Key Advantages & Disadvantages |
|---|---|---|---|---|
| Traditional Wet | Very Low (with gel) | Poor: Degrades as gel dries [58] | ~6.36 minutes [58] | Adv: Gold standard for low impedance.Disadv: Messy, time-consuming, skin irritation. [59] [58] |
| Dry Contact | High (1-2 MΩ) [58] | Excellent: No gel to dry out [58] | ~4.02 minutes [58] | Adv: Quick setup, suitable for long-term use.Disadv: Requires high-impedance amplifiers. [58] |
| MEMS Microneedle | Low [59] | Good (penetrates stratum corneum) [59] | Not Specified | Adv: Low impedance, minimal preparation.Disadv: Complex fabrication, potential discomfort. [59] |
| Hairlike Hydrogel | Not Specified | Excellent: >24 hours stable performance [56] | Not Specified | Adv: Discreet, comfortable, stable long-term contact.Disadv: Currently wired, still in development. [56] |
Table 2: Impedance-Related Artifacts and Corrective Actions
| Problem | Symptom in EEG Signal | Corrective Action |
|---|---|---|
| High Electrode-Skin Impedance | Increased 50/60 Hz noise, overall low signal amplitude. | Improve skin contact (cleaning, light abrasion) or use active electrodes. [57] [58] |
| Stray Capacitance | Signal loss at high frequencies, distorted waveform. | Shorten cables, use guarded leads, secure wiring to minimize movement. [57] |
| Stray Inductance | High-frequency artifacts, "fake" inductive loops in data. | Twist current-carrying leads together, minimize loop areas in the wiring. [57] |
| Unstable Contact | Signal drift, sudden DC shifts, movement artifacts. | Re-seat electrode, check for proper mounting, use adhesive overlays or stable headgear. [56] [58] |
Workflow: Managing Impedance for High-Quality EEG
Pathway: Impact of Impedance on Data & Analysis
The Scientist's Toolkit
| Item | Function in Impedance Management |
|---|---|
| Abrading Skin Prep Gel | Gently removes dead skin cells (stratum corneum) to significantly lower initial skin-electrode impedance. |
| Electrolyte Gel (for wet electrodes) | Acts as a conductive bridge between skin and electrode, creating a stable, low-impedance interface. |
| Active Dry Electrodes | Incorporate a local amplifier to buffer the signal, overcoming challenges of high skin-electrode impedance without gel. [58] |
| 3D-Printed Hydrogel Electrodes | Provide a gel-like conductive interface in a solid, adhesive form, offering stable impedance for long-term monitoring. [56] |
| Faraday Cage | An enclosed, grounded space that shields the EEG setup from external electromagnetic noise, which becomes more prevalent with high impedance. [57] |
| Impedance Checker | A dedicated device or software feature to measure and verify contact impedance at each electrode prior to and during recording. |
Electroencephalography (EEG) is a fundamental tool in neuroscience research and clinical diagnostics, with electrode technology playing a crucial role in signal acquisition quality. The two primary electrode types—gel-based (wet) and dry systems—offer distinct advantages and present unique challenges, particularly regarding artifact profiles and signal integrity. Understanding these differences is essential for researchers selecting appropriate methodology for EEG experiments, especially in the context of artifact removal and noise filtering research.
Gel-based EEG systems represent the conventional approach, utilizing electrodes filled with conductive gel or paste that requires skin preparation and significant setup time. Dry EEG systems have emerged as a convenient alternative, making direct contact with the scalp without conductive gel. The evolution of dry electrode technology has been driven by advances in amplifier design and active electrode technology that compensate for higher impedance levels characteristic of dry electrode-skin interfaces [61].
Comparative Analysis: Dry vs. Gel-Based EEG Systems
Table 1: Fundamental Characteristics and Practical Considerations
| Feature | Gel-Based (Wet) EEG Systems | Dry EEG Systems |
|---|---|---|
| Setup Time | Lengthy (requires skin abrasion, gel application) [61] | Rapid (minutes) [61] [62] |
| Required Expertise | Trained EEG technologist [61] | Minimal technical training [61] |
| Patient Comfort | Lower (skin irritation, hair damage, cleanup) [61] [63] | Higher (no residue, minimal preparation) [61] |
| Portability | Limited (often wired systems, gel requirements) [62] | High (wireless options available) [62] |
| Ideal Use Cases | Clinical settings with trained staff, long-term monitoring [61] | Rapid screenings, field research, BCIs, settings without EEG specialists [61] |
| Hygiene | Reusable electrodes require cleaning between patients [61] | Disposable options available; more hygienic patient transfer [61] |
Table 2: Signal Quality and Performance Metrics
| Parameter | Gel-Based (Wet) EEG Systems | Dry EEG Systems |
|---|---|---|
| Impedance Management | Conductive gel reduces impedance effectively [61] | Higher impedance overcome by active electrodes & advanced amplifiers [61] |
| Signal-to-Noise Ratio | Generally high [63] | Can be lower; requires noise cancellation circuitry [61] [63] |
| Resting-State Alpha Power | Reliably detects increase in eyes-closed condition [64] | Variable by device; PSBD Headband sensitive, Muse Headband limited [64] |
| High-Frequency Oscillations | Strong performance (e.g., beta, gamma) [64] | PSBD Headphones show strong alignment; others variable [64] |
| Event-Related Potentials (P300) | Robust detection [62] | Comparable detection to wet systems; positive correlations (r = 0.54-0.89) [62] |
| Mismatch Negativity (MMN) | Reliable amplitude and theta power [63] | Detectable but may underestimate amplitude and theta power [63] |
| Faulty Electrodes | Fewer (approximately 2.0 per session) [62] | More frequent (approximately 5.26 per session) [62] |
EEG System Selection Workflow
Artifact Profiles and Troubleshooting
FAQ: Common Experimental Issues
Q: What are the most common physiological artifacts affecting EEG signals, and how do they differ between systems?
A: Both systems are susceptible to similar physiological artifacts, though dry electrodes may be more vulnerable to certain types due to higher impedance [61]:
- Eye Blinks & Movements: High-amplitude frontal deflections from corneal-retinal potential [18]. More pronounced in dry systems with frontal placement.
- Muscle Artifact (EMG): High-frequency, low-amplitude activity from frontalis/temporalis muscles [18]. Dry systems may show increased susceptibility.
- Cardiac Artifact (ECG): Periodic waveforms time-locked to heartbeat [18]. Potentially more prominent in dry systems.
- Electrode Artifacts: Caused by loose electrodes or movement [18]. Dry systems have higher faulty electrode rates [62].
Q: How does the artifact profile differ between dry and gel-based systems during movement?
A: Gel-based systems generally maintain better contact during minor movements due to the conductive bridge provided by gel. Dry electrodes are more susceptible to motion artifacts because they lack this stabilizing conductive medium, making them less ideal for experiments involving significant participant movement [62].
Q: Which system provides more reliable data for resting-state connectivity studies?
A: For higher frequencies (alpha and beta bands), dry EEG reliably estimates resting-state connectivity measures like Minimum Spanning Tree (MST) diameter. However, in very low frequencies (0.5-4 Hz), gel-based systems may provide more reliable connectivity metrics [63].
Q: What are the key limitations of consumer-grade dry EEG systems?
A: Research indicates significant variability among dry EEG devices. For instance, while the PSBD Headband shows sensitivity in low-frequency ranges and replicates modulations in delta, theta, and alpha power, the Muse Headband demonstrates several limitations in signal quality. The potential benefits of consumer dry electrodes in terms of ease of use should be carefully weighed against the capacity of each specific system [64].
Troubleshooting Guides
Problem: Excessive noise in dry EEG recordings
- Cause: High electrode-skin impedance and insufficient shielding [61]
- Solution: Ensure proper electrode-scalp contact; use systems with active electrodes and dynamic common mode noise rejection circuitry (CMRR >130dB) [61]
- Prevention: Select dry systems with built-in pre-amplification and excellent shielding mechanisms
Problem: Unstable signals during long recordings with gel-based EEG
- Cause: Gel drying over time, particularly in warm environments [61]
- Solution: For recordings exceeding 1-2 hours, use collodion-fixed electrodes with periodic gel replenishment [61]
- Prevention: Ensure proper gel volume during initial application; use hygroscopic gels for long-term studies
Problem: Artifact contamination in critical datasets
- Cause: Multiple sources (physiological, environmental, electrode) [18]
- Solution: Implement advanced artifact removal algorithms (CLEnet, ART, GEDAI) that leverage deep learning or leadfield filtering approaches [4] [3] [29]
Artifact Mitigation Strategy
Advanced Artifact Removal Methodologies
Table 3: Contemporary Artifact Removal Algorithms
| Algorithm | Underlying Technology | Key Advantages | Performance Notes |
|---|---|---|---|
| CLEnet [4] | Dual-scale CNN + LSTM with improved EMA-1D | Extracts morphological & temporal features; handles multi-channel EEG & unknown artifacts | SNR improvement: 2.45%; RRMSEt decrease: 6.94% [4] |
| ART (Artifact Removal Transformer) [3] | Transformer architecture | Captures millisecond-scale EEG dynamics; removes multiple artifact types simultaneously | Sets new benchmark; superior to other DL methods in BCI applications [3] |
| GEDAI [29] | Leadfield filtering with SENSAI | Theory-informed; uses forward model of brain signal generation; fully automated | Classification accuracy to AUC 0.80; 91% subject identification accuracy [29] |
| DWT-CNN-BiGRU [31] | Discrete Wavelet Transform + CNN + Bidirectional GRU | Effective for spatial & temporal feature extraction; excellent for classification tasks | 94% accuracy in alcoholic vs. control classification [31] |
| 1D-ResCNN [4] | 1D Residual Convolutional Neural Network | Multi-scale feature extraction with three convolutional kernels | Effective for artifact reconstruction but outperformed by newer architectures [4] |
The Scientist's Toolkit
Table 4: Essential Research Reagents and Computational Tools
| Item | Function/Purpose | Example Applications |
|---|---|---|
| Conductive Electrolyte Gel | Reduces skin-electrode impedance; improves signal continuity [61] | Standard wet EEG setups; long-term clinical monitoring [61] |
| Active Dry Electrodes | Integrated pre-amplification near electrode site [61] | Wireless EEG systems; rapid deployments; high-impedance environments [61] |
| Multi-Pin Dry Electrodes | Penetrate hair layer; improve contact with scalp [64] | Dense hair coverage areas; consumer EEG devices [64] |
| ICA Software Packages | Blind source separation for artifact identification [4] | Component-based artifact removal; preprocessing for research EEG [4] |
| EEGdenoiseNet Dataset | Benchmark dataset for algorithm training/validation [4] | Developing and comparing artifact removal methods [4] |
| Leadfield Model | Theoretical blueprint of brain signal generation [29] | GEDAI algorithm; theory-informed artifact removal [29] |
The choice between dry and gel-based EEG systems involves careful consideration of research objectives, experimental conditions, and technical resources. Gel-based systems remain the gold standard for clinical applications requiring maximum signal fidelity, particularly for low-frequency analysis and MMN studies. Dry electrode systems offer compelling advantages for rapid screening, field research, and scenarios where technical expertise is limited.
For researchers focusing on artifact removal methodologies, both systems present unique challenges that can be addressed with contemporary algorithms like CLEnet, ART, and GEDAI. The future of EEG artifact management lies in specialized deep learning architectures that can handle multi-channel data and diverse artifact types while preserving the neural signals of interest.
When designing experiments, researchers should:
- Validate dry system performance for their specific frequency bands of interest
- Implement appropriate artifact mitigation strategies during both data acquisition and processing
- Consider the trade-offs between convenience and signal quality when selecting electrode technology
- Stay informed of emerging algorithms and validation studies for continuously improving artifact removal capabilities
Optimizing Method Selection Based on Artifact Type and Research Context
FAQs and Troubleshooting Guides
This technical support resource addresses common challenges in EEG artifact removal, providing evidence-based guidance to help researchers select the optimal denoising strategy for their specific context.
FAQ 1: How do I choose the best artifact removal method for my specific type of EEG artifact?
Different artifacts have distinct properties, and method performance varies accordingly. The table below summarizes the suitability of advanced algorithms for common artifact types based on recent peer-reviewed studies.
Table 1: Artifact-Specific Performance of Advanced Removal Methods
| Artifact Type | Recommended Methods | Key Research Findings and Performance Metrics |
|---|---|---|
| Muscle Artifact (EMG) | NovelCNN [4]CLEnet [4] | NovelCNN: Specifically designed for EMG removal [4].CLEnet: Excels at removing mixed artifacts (EMG+EOG), achieving an SNR of 11.498 dB and a CC of 0.925 [4]. |
| Ocular Artifact (EOG) | EEGDNet (Transformer) [4]ART (Transformer) [3] | EEGDNet: Demonstrates outstanding performance in removing EOG artifacts [4].ART: An Artifact Removal Transformer that holistically addresses multiple artifact types in multichannel EEG, setting a new benchmark [3]. |
| Cardiac Artifact (ECG) | CLEnet [4] | Outperformed other models, showing a 5.13% increase in SNR and an 8.08% decrease in temporal domain error (RRMSEt) compared to other deep learning models [4]. |
| Mixed/Unknown Artifacts | CLEnet [4]GEDAI [29] | CLEnet: Specifically tested on a dataset with "unknown artifacts," showing a 2.45% SNR improvement and a 6.94% decrease in RRMSEt [4].GEDAI: Effective for complex mixtures of EOG, EMG, and channel noise, even with 100% temporal contamination. It significantly improved single-trial ERP classification accuracy to an AUC of 0.80 [29]. |
| General Purpose / Broad Spectrum | Nested GAN [65] | An end-to-end method using a nested Generative Adversarial Network. It achieved a high percentage reduction of artifacts in both time (ηtemporal=71.6%) and frequency domains (ηspectral=76.9%) [65]. |
FAQ 2: What is the fundamental difference between traditional blind source separation and newer deep learning or model-based approaches?
The core difference lies in how "true brain signal" is defined and identified.
- Traditional Methods (e.g., ICA, PCA): These are often "blind" and rely on statistical properties of the recorded data. For example, ICA separates signals into statistically independent components, which then require manual or semi-automated inspection to classify as brain signal or artifact [4] [29].
- Newer Model-Based & Deep Learning Approaches:
- Model-Based (e.g., GEDAI): This method is "theoretically informed." It uses a pre-computed forward model (leadfield) of how brain electrical activity projects to the scalp. It then filters out any signal that does not match this spatial blueprint of genuine brain activity [29].
- Deep Learning (e.g., CLEnet, ART): These are data-driven and learn the features of both clean EEG and artifacts from large training datasets. CLEnet, for instance, integrates CNNs and LSTM to learn both the morphological and temporal features of clean EEG, separating it from artifacts in an end-to-end supervised manner [4] [3].
FAQ 3: My research involves multi-channel EEG data for a Brain-Computer Interface (BCI) application. Which methods are most suitable?
For BCI and other applications requiring multi-channel integrity, select methods that preserve spatial relationships across channels.
- Recommended Methods: ART (Artifact Removal Transformer) [3], CLEnet [4], and GEDAI [29] are explicitly designed and validated for multi-channel EEG data.
- Key Advantage: These methods process all channels collectively, allowing them to leverage spatial information for more effective artifact removal and reconstruction of the full scalp topography, which is crucial for source analysis and BCI [4] [3] [29].
Troubleshooting Guide: My EEG data is still noisy after applying a standard artifact removal algorithm. What should I check?
Follow this systematic troubleshooting workflow to isolate the issue.
Systematic Troubleshooting for Persistent EEG Noise
Troubleshooting Guide: I am setting up a new, large-scale EEG study. How can I proactively minimize artifact-related issues?
- Run Pilot Sessions: Before full-scale data collection, conduct pilot sessions to verify that all equipment is working, stimuli are presented correctly, and that your artifact removal pipeline performs as expected on your specific setup [66].
- Establish a Data Quality Protocol: Implement and document a rigorous data collection protocol. This includes standardized electrode application to ensure low impedances, training for all research staff, and a system for regular data quality monitoring [67].
- Plan for Artifact Rejection vs. Correction: Decide upfront on your strategy for handling artifacts. Will you reject contaminated trials or correct them using regression, interpolation, or advanced methods like ICA? This decision should be based on your research question and the nature of your data [66].
- Use a Multi-Modal Approach: If possible, augment your EEG recording with other sensors, such as an eye tracker to monitor blinks or EOG/EMG electrodes to record common artifacts. This provides reference signals that can significantly improve the identification and removal of these artifacts [66].
The Scientist's Toolkit
Table 2: Key Computational Tools and Reagents for EEG Artifact Removal Research
| Tool / Algorithm Name | Type | Primary Function | Key Features / Explanation |
|---|---|---|---|
| CLEnet [4] | Deep Learning Model | Removes various artifacts from multi-channel EEG. | Integrates dual-scale CNN and LSTM with an attention mechanism (EMA-1D) to extract both morphological and temporal features of clean EEG. |
| ART (Artifact Removal Transformer) [3] | Deep Learning Model | End-to-end denoising of multichannel EEG. | Uses transformer architecture to capture millisecond-scale EEG dynamics. Effective against multiple artifact types simultaneously. |
| GEDAI [29] | Model-Based Algorithm | Unsupervised denoising of multichannel EEG. | Uses a leadfield model (forward model) as a theoretical blueprint to separate brain signals from non-brain artifacts. Fully automated via the SENSAI index. |
| Nested GAN [65] | Deep Learning Model | End-to-end artifact removal. | Employs an inner GAN in the time-frequency domain and an outer GAN in the time domain to reconstruct clean EEG. |
| DWT-CNN-BiGRU [68] | Hybrid Processing Pipeline | EEG denoising and classification. | Combines Discrete Wavelet Transform for denoising, CNN for spatial features, and a Bidirectional GRU for temporal dependencies. |
| NeuroTools [69] | Python Software Library | Analysis and visualization of neuroscientific data. | Provides modules for data management, analysis, and visualization. Can be used to build custom analysis pipelines for EEG data. |
Parameter Tuning and Performance Optimization for Algorithmic Approaches
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: How do I choose the right artifact removal algorithm for my specific tES-EEG setup? The optimal algorithm is highly dependent on your stimulation type. For tDCS artifacts, a Complex CNN performs best. For the more complex artifacts from tACS or tRNS, a multi-modular State Space Model (SSM) like the M4 network delivers superior results. Always validate new algorithms on semi-synthetic datasets with known ground truth first. [15]
Q2: My single-channel EEG system is contaminated with eye-blink artifacts. Which method is most effective? For single-channel EOG artifact removal, an automated method combining Fixed Frequency Empirical Wavelet Transform (FF-EWT) with a Generalized Moreau Envelope Total Variation (GMETV) filter has shown excellent performance. This approach effectively identifies contaminated components using kurtosis, dispersion entropy, and power spectral density metrics before filtering. [47]
Q3: What is a major limitation of current deep learning models for EEG artifact removal? Many existing networks are designed for specific artifact types and perform poorly on unknown artifacts. They also often process only single-channel inputs, overlooking inter-channel correlations in multi-channel EEG data, which limits their real-world applicability. [70]
Q4: Which open-source tool is best for researchers without extensive programming experience? For beginners, Brainstorm offers an intuitive graphical interface that requires no programming knowledge and is available as a free standalone application. EEGLAB also provides a user-friendly GUI within MATLAB for common analyses like ICA and filtering. [71]
Q5: How can I optimize a neural network for removing multiple types of artifacts from multi-channel EEG? Implement a dual-branch architecture like CLEnet that integrates dual-scale CNNs for morphological feature extraction and LSTM networks for temporal feature extraction. Incorporating a 1D Efficient Multi-Scale Attention (EMA-1D) mechanism can further enhance performance across diverse artifact types. [70]
Performance Benchmarking and Algorithm Selection
Table 1: Performance Comparison of Deep Learning Models for Specific Artifact Types
| Artifact Type | Optimal Algorithm | Key Performance Metrics | Alternative Algorithms |
|---|---|---|---|
| tDCS | Complex CNN | Best RRMSE in temporal/spectral domains [15] | State Space Models (SSMs) |
| tACS / tRNS | Multi-modular SSM (M4 Network) | Best RRMSE & Correlation Coefficient [15] | Convolutional Neural Networks |
| EOG (Single-Channel) | FF-EWT + GMETV Filter | Lower RRMSE, Higher CC and SAR on real EEG [47] | EMD, SSA, VMD, DWT, ICA |
| Multiple/Unknown | CLEnet (CNN + LSTM + EMA-1D) | SNR: 11.498dB, CC: 0.925 (mixed artifacts) [70] | 1D-ResCNN, NovelCNN, DuoCL |
Table 2: Quantitative Performance of CLEnet vs. Other Models on Mixed Artifacts (EMG + EOG)
| Model | SNR (dB) | Correlation Coefficient (CC) | RRMSE (Temporal) | RRMSE (Spectral) |
|---|---|---|---|---|
| CLEnet | 11.498 | 0.925 | 0.300 | 0.319 |
| DuoCL | 10.912 | 0.901 | 0.321 | 0.332 |
| NovelCNN | 10.654 | 0.894 | 0.329 | 0.341 |
| 1D-ResCNN | 10.423 | 0.883 | 0.335 | 0.348 |
Detailed Experimental Protocols
Protocol 1: Creating a Semi-Synthetic Dataset for Controlled Model Evaluation
This methodology enables rigorous, controlled benchmarking of artifact removal algorithms when pure ground truth data is unavailable. [15] [70]
- Acquire Clean EEG Data: Obtain artifact-free EEG recordings from public repositories or collect them during restful, closed-eye conditions.
- Record or Simulate Artifacts:
- Real Artifacts: Record standalone EOG (from eye blinks), EMG (from jaw clenching), or ECG signals.
- Synthetic Artifacts: Generate tES artifacts (tDCS, tACS, tRNS) or other noise waveforms that match the characteristics of the interference. [15]
- Combine Signals: Artificially mix the clean EEG and artifact signals at controlled Signal-to-Noise Ratios (SNRs) to create a contaminated dataset. The mathematical relationship is:
Contaminated_EEG = Clean_EEG + γ * Artifact, whereγis a scaling factor to achieve the target SNR. - Validate Dataset: Ensure the semi-synthetic data realistically mimics the properties of real contaminated recordings.
Protocol 2: Benchmarking Deep Learning Models for Artifact Removal
This protocol provides a standardized framework for comparing the performance of different deep learning models, such as CLEnet, DuoCL, and 1D-ResCNN. [70]
- Data Preparation: Split the semi-synthetic dataset (from Protocol 1) into training, validation, and test sets (e.g., 70%/15%/15%).
- Model Training:
- Train each model on the training set using the contaminated EEG as input and the clean EEG as the target.
- Use Mean Squared Error (MSE) as the loss function.
- Employ the validation set for early stopping to prevent overfitting.
- Model Evaluation: Use the trained models to reconstruct clean EEG from the contaminated test set.
- Performance Quantification: Calculate standard metrics to compare the reconstructed signal against the known ground truth:
- Signal-to-Noise Ratio (SNR) in decibels (dB).
- Correlation Coefficient (CC) between the reconstructed and clean EEG.
- Relative Root Mean Square Error in both temporal (RRMSEt) and spectral (RRMSEf) domains.
Workflow and Algorithmic Relationships
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Software Tools for EEG Artifact Removal Research
| Tool Name | Platform/Language | Primary Function in Research | Key Advantage |
|---|---|---|---|
| MNE-Python | Python | Comprehensive EEG/MEG processing; signal filtering, ICA, source analysis | Integrates with scientific Python stack; extensible with specialized plugins [71] [72] |
| EEGLAB | MATLAB | Interactive toolbox for ICA, artifact rejection, time-frequency analysis | Large user base & plugin ecosystem; user-friendly GUI [73] [71] |
| NeuroKit2 | Python | User-friendly biosignal processing (EDA, ECG, RSP, EMG, EOG) | Accessible to non-experts; facilitates rapid prototyping [74] |
| FieldTrip | MATLAB | Advanced analysis of MEG, EEG, iEEG; source reconstruction, statistics | High flexibility for building custom analysis pipelines [71] |
| Brainstorm | Standalone (MATLAB/Java) | Intuitive GUI for MEG/EEG source localization and connectivity analysis | No programming required; does not need a MATLAB license [71] |
Handling Motion Artifacts in Ambulatory and Naturalistic Recordings
The expansion of electroencephalography (EEG) into ambulatory and naturalistic recordings presents a significant challenge: the pervasive presence of motion artifacts. Unlike controlled laboratory settings, real-world environments introduce complex movement-related noise that can compromise data integrity. These artifacts originate from multiple sources, including electrode-skin interface instability, cable movement, and modulation of power line interference due to variable impedance [75]. Their amplitude can be orders of magnitude greater than neural signals, and their non-stationary, unpredictable nature makes them particularly difficult to remove with conventional filtering techniques [75]. Effectively managing these artifacts is therefore a critical prerequisite for reliable EEG analysis in dynamic contexts such as sports science, neurorehabilitation, and cognitive studies in ecological settings.
FAQ: Addressing Common Researcher Questions
Q1: Why are motion artifacts particularly problematic in wearable EEG systems compared to traditional lab setups?
Motion artifacts are more problematic in wearable systems due to three primary factors: the uncontrolled environment, the presence of natural body movements, and the use of dry or semi-dry electrodes for rapid setup [5]. These factors lead to reduced electrode stability and increased susceptibility to artifacts from cable sway and changes in electrode-skin impedance [75]. Furthermore, wearable systems often have a lower channel count (typically below 16 channels), which limits the effectiveness of standard artifact rejection techniques like Independent Component Analysis (ICA) that rely on high spatial resolution [5].
Q2: What are the most reliable methods for detecting motion artifacts in real-time?
The most reliable methods for real-time detection often involve a combination of approaches:
- Artifact Subspace Reconstruction (ASR): An adaptive method that detects and reconstructs data segments contaminated by high-amplitude, non-stationary artifacts, making it suitable for real-time application [11] [5].
- Wavelet Transform Techniques: These are effective for identifying transient, spike-like artifacts caused by cable movement or electrode pops [5].
- Auxiliary Sensors: Data from inertial measurement units (IMUs) or accelerometers can provide a direct correlate of body movement, helping to identify motion-locked artifacts [5]. While promising, the systematic use of these sensors is still underutilized.
Q3: Can we completely remove motion artifacts without affecting the underlying neural signal?
Complete removal without any impact on the neural signal is currently not possible. The fundamental challenge is the overlap in spectral and temporal characteristics between some artifacts and brain signals of interest [75]. The goal of artifact removal is therefore to minimize the artifact's contribution while preserving as much neural data as possible. The choice of algorithm involves a trade-off; more aggressive correction may remove more artifact but risks distorting or removing neural activity, while a conservative approach may leave residual contamination [11].
Q4: How do I choose between simple artifact rejection and advanced correction methods?
The choice depends on your research question, data characteristics, and analysis goals.
- Artifact Rejection: This involves discarding contaminated data epochs. It is the safest method for preserving signal integrity and is recommended when artifacts are infrequent and localized, and when you have a sufficiently large dataset to maintain statistical power after rejection.
- Artifact Correction: Methods like ICA, ASR, or regression-based techniques are necessary when artifacts are pervasive or periodic, when studying continuous processes (e.g., sleep, locomotion), or when the data collection protocol makes re-testing or obtaining more data impractical [11] [20].
Troubleshooting Guides: Solving Common Experimental Problems
Problem 1: Slow Baseline Drifts Correlated with Movement
- Symptoms: Low-frequency, high-amplitude waves that coincide with rhythmic activities like walking or running, often visible in individual channels [75].
- Primary Cause: Relative movement between the electrode and the skin, causing alterations in the ion distribution at the electrode-skin interface [75].
- Solutions:
- Hardware Prevention: Ensure a secure and stable fit of the EEG cap. Use adequate electrolyte gel (for wet systems) and consider headbands or straps to minimize cap movement. Textile-based electrodes may reduce sensitivity to these artifacts on hairless regions [75].
- Post-Processing: High-pass filtering can attenuate very slow drifts. However, for drifts correlated with movement frequency, more advanced techniques like Artifact Subspace Reconstruction (ASR) or blind source separation may be required [11] [75].
Problem 2: High-Frequency, Spike-like Artifacts
- Symptoms: Sudden, large-amplitude transients that are not time-locked to movement and have an irregular shape [75].
- Primary Cause: Cable movement due to triboelectric effects (friction generating charge) or sudden changes in electrode-skin impedance (electrode pops) [1] [75].
- Solutions:
- Hardware Prevention: Secure all cables to the participant's clothing using tape or clips to minimize swing and movement. Use lightweight, flexible cables and check for sufficient electrode gel to maintain stable impedance.
- Post-Processing: Wavelet-based methods are particularly effective at identifying and removing these transient artifacts [5]. Automated algorithms can detect and remove sections of data contaminated by electrode pops.
Problem 3: Widespread, Unstructured Noise During Movement
- Symptoms: High-amplitude, non-stationary noise across multiple channels during gross motor activity, making brain signals indiscernible.
- Primary Cause: Complex body movements leading to a combination of cable movement, unstable electrode-skin contact, and muscle activation (EMG) [1].
- Solutions:
- Hardware Prevention: This is difficult to prevent entirely during dynamic tasks. Using active electrodes can help reject some environmental interference but are less effective for motion artifacts themselves [75].
- Post-Processing: This is a major challenge. Multi-stage pipelines that combine several methods are most effective. For example, the EEG-cleanse pipeline is designed specifically for cleaning EEG during full-body movement, combining motion-adaptive preprocessing with a hybrid labeling strategy [76]. Deep learning approaches are also emerging as a promising solution for this complex artifact type [15] [5].
Quantitative Data: Algorithm Performance Comparison
The table below summarizes the performance of various artifact removal algorithms as validated in recent research, providing a guide for selection based on quantitative metrics.
Table 1: Performance Comparison of Motion Artifact Handling Algorithms
| Algorithm Category | Typical Application Context | Key Performance Metrics | Reported Efficacy | Key Advantages and Limitations |
|---|---|---|---|---|
| Artifact Subspace Reconstruction (ASR) | Ocular, movement, and instrumental artifacts; real-time application [11] [5] | Signal-to-Artifact Ratio (SAR), Relative Root Mean Square Error (RRMSE) [5] | Widely applied; shows robust performance for non-stationary artifacts [11] [5] | Advantage: Adaptive, works well with low-channel setups. Limitation: May require careful parameter tuning [11]. |
| Independent Component Analysis (ICA) | Ocular and persistent muscular artifacts [11] [20] | Accuracy (~71%), Selectivity (~63%) when clean signal is reference [5] | High accuracy for separating physiological artifacts like blinks [20] [5] | Advantage: Excellent for separating sources. Limitation: Requires multiple channels; performance drops with high artifact burden or low channel count [11] [5]. |
| Wavelet Transform | Ocular and muscular artifacts, transient "spike" artifacts [5] | Correlation Coefficient (CC), Mean Absolute Error (MAE) [5] | Effective for transient and spike-like artifacts from cable movement [5] | Advantage: Good at localizing transients in time and frequency. Limitation: May not handle slow, baseline drifts as effectively. |
| Deep Learning (e.g., CNN, SSM) | Muscular and motion artifacts; complex scenarios like tES-artifact removal [15] [5] | Root Relative Mean Squared Error (RRMSE) in temporal/spectral domains, Correlation Coefficient (CC) [15] | RRMSE and CC values indicate superior performance for complex artifacts (e.g., tACS, tRNS) vs. traditional methods [15] | Advantage: Can model complex, non-linear artifact patterns. Limitation: Requires large, labeled datasets for training; "black box" nature [15]. |
| Fixed Frequency EWT + GMETV | Single-channel EOG artifact removal [47] | RRMSE, Correlation Coefficient (CC), Signal-to-Artifact Ratio (SAR) [47] | Substantial improvement in SAR and lower RRMSE/MAE on real and synthetic data [47] | Advantage: Specifically designed for single-channel setups. Limitation: Performance is optimized for specific artifact types (e.g., EOG). |
Experimental Protocols: Detailed Methodologies
Protocol 1: Ocular Artifact Removal Using Independent Component Analysis (ICA)
ICA is a blind source separation technique that decomposes multi-channel EEG data into statistically independent components, allowing for the identification and removal of those representing artifacts [11].
- Data Preparation: Load the raw, continuous EEG data. Apply a high-pass filter (e.g., 1 Hz cutoff) to remove slow drifts that can impede ICA performance.
- ICA Decomposition: Use an algorithm (e.g., Infomax or Extended Infomax) to decompose the filtered data into independent components. Each component has a fixed scalp topography and a time course of activation.
- Component Classification: Visually inspect components to identify those corresponding to ocular artifacts. Key indicators include:
- Topography: Strong frontal focus, typical of eye blinks and movements [20].
- Time Course: Large, low-frequency deflections time-locked with blink events.
- Spectrum: Dominance of low-frequency power (delta/theta bands) [11]. (Optional) Use automated classifiers like ICLabel or SASICA to assist in identification.
- Artifact Removal: Select the components identified as artifacts and remove them from the data.
- Signal Reconstruction: Project the remaining components back to the sensor space, resulting in an EEG dataset with the ocular artifacts subtracted.
Protocol 2: Automated Pipeline for Full-Body Movement (EEG-Cleanse)
The EEG-cleanse pipeline is a modular, automated method designed for cleaning EEG data collected during full-body movement without specialized hardware [76].
- Data Input and Channel Location: Import the raw EEG data and assign standard channel locations.
- Band-Pass Filtering: Apply a band-pass filter (e.g., 1-45 Hz) to remove extraneous low- and high-frequency noise.
- Bad Channel Detection and Removal: Automatically identify channels with excessive noise or flat-line signals based on statistical criteria (e.g., abnormal kurtosis, variance) and remove them.
- Artifact Subspace Reconstruction (ASR): Use the ASR algorithm to identify and remove large-amplitude, non-stationary artifacts by reconstructing the contaminated data segments based on clean calibration data or the statistics of the overall recording [76].
- Spherical Interpolation: Reconstruct the bad channels removed in Step 3 using spherical interpolation from the clean, surrounding channels.
- Re-referencing: Re-reference the data to a common average reference to improve the signal quality.
Protocol 3: Single-Channel EOG Removal using FF-EWT and GMETV
This protocol is designed for portable, single-channel EEG systems where multi-channel techniques like ICA are not feasible [47].
- Decomposition: Apply Fixed Frequency Empirical Wavelet Transform (FF-EWT) to the contaminated single-channel EEG signal. This decomposes the signal into six Intrinsic Mode Functions (IMFs) with compact frequency support [47].
- Artifact Component Identification: Automatically identify the IMFs contaminated with EOG artifacts using a feature-based threshold. The metrics used are kurtosis (KS), dispersion entropy (DisEn), and power spectral density (PSD) [47].
- Filtering: Process the artifact-related IMFs identified in the previous step with a finely tuned Generalized Moreau Envelope Total Variation (GMETV) filter. This filter selectively suppresses the artifact content within these components [47].
- Signal Reconstruction: Reconstruct the clean EEG signal by summing the processed artifact components with the unchanged, clean IMFs.
Signaling Pathways and Workflows
The following diagram illustrates the primary sources of motion artifacts in a traditional biopotential acquisition chain, as identified through experimental observation and electrical modeling [75].
Diagram 1: Motion artifact sources and their effects in the EEG signal.
Diagram 2: Multi-Stage EEG-Cleanse Pipeline Workflow
This diagram outlines the workflow of the EEG-cleanse pipeline, a modular and automated approach for cleaning EEG data recorded during full-body movement [76].
Diagram 2: The automated EEG-cleanse pipeline for full-body movement.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools for Motion Artifact Management in Research
| Tool / Solution | Category | Primary Function in Artifact Handling |
|---|---|---|
| Dry / Semi-Dry Electrodes | Hardware | Enables rapid setup for mobile EEG; trades off some signal stability for convenience and user comfort [5]. |
| Active Electrode Systems | Hardware | Amplifies signal at the source (electrode), reducing interference picked up by cables; effective against power line noise but less so against motion artifacts [75]. |
| Inertial Measurement Units (IMUs) | Auxiliary Sensor | Provides objective, synchronized movement data (acceleration, rotation) to identify motion-locked artifacts for informed rejection or correction [5]. |
| Artifact Subspace Reconstruction (ASR) | Software Algorithm | An adaptive, automated method for identifying and removing large-amplitude, non-stationary artifacts in multi-channel data, suitable for real-time use [11] [76] [5]. |
| Fixed Frequency EWT + GMETV | Software Algorithm | A specialized method for robust artifact removal in single-channel EEG recordings, where multi-channel techniques are not applicable [47]. |
| Deep Learning Models (CNNs, SSMs) | Software Algorithm | Models complex, non-linear artifact patterns (e.g., from muscle or motion); shows high performance but requires significant training data [15] [5]. |
Evaluating Performance: Validation Metrics and Comparative Analysis of Denoising Methods
In electroencephalography (EEG) research, particularly in the fields of artifact removal and noise filtering, the performance of novel algorithms and processing techniques must be quantitatively validated using robust, standardized metrics. These metrics allow researchers to objectively compare the effectiveness of different methods, ensure the integrity of neural signals, and draw reliable conclusions about brain activity. Among the most critical metrics are the Signal-to-Noise Ratio (SNR), the Correlation Coefficient (CC), and the Root Mean Square Deviation (RMSD), also referred to as Root Mean Square Error (RMSE). Their collective application provides a comprehensive view of a method's performance across temporal, spectral, and similarity domains. This technical support guide provides researchers with a foundational understanding of these metrics, detailed protocols for their calculation, and troubleshooting advice for common experimental challenges.
Metric Definitions and Theoretical Foundations
Signal-to-Noise Ratio (SNR)
Definition: SNR quantifies the level of a desired neural signal relative to the level of background noise. It is a crucial indicator of signal quality and the effectiveness of artifact removal pipelines.
Relevance: In EEG analysis, a high SNR indicates that the brain signal of interest (e.g., an Event-Related Potential) is clear and distinguishable from contaminating noise, which can originate from ocular, muscular, environmental, or instrumental sources [21]. It is especially important for validating EEG systems designed for use in mobile or real-world conditions [77].
Correlation Coefficient (CC)
Definition: The Correlation Coefficient (often Pearson's r) measures the strength and direction of the linear relationship between two signals. In the context of artifact removal, it is typically used to compare a processed, "cleaned" EEG signal against a ground-truth, clean reference.
Relevance: A CC value close to 1 indicates that the denoising process has successfully preserved the original neurophysiological signal's morphology while removing artifacts. It is a key metric for assessing signal fidelity and ensuring that the cleaning process is not distorting the underlying brain activity [15] [78].
Root Mean Square Deviation (RMSD/RMSE)
Definition: RMSD measures the average magnitude of the differences between a processed signal and a reference signal. It provides a standard measure of error.
Relevance: A lower RMSD indicates that the cleaned signal is closer to the true, uncontaminated EEG. A common variant used in EEG denoising is the Root Relative Mean Squared Error (RRMSE), which normalizes the error, making it easier to compare results across different datasets or conditions [15].
Table 1: Overview of Standardized Evaluation Metrics for EEG
| Metric | Acronym | Ideal Value | Primary Interpretation | Key Advantage |
|---|---|---|---|---|
| Signal-to-Noise Ratio | SNR | Higher is better | Strength of neural signal relative to noise | Direct indicator of signal quality for analysis |
| Correlation Coefficient | CC | Closer to 1 is better | Fidelity of cleaned signal to original | Ensures neurophysiological content is preserved |
| Root Mean Square Deviation | RMSD | Lower is better | Average magnitude of error | Standard, intuitive measure of accuracy |
Experimental Protocols and Calculation Methodologies
This section outlines standard experimental workflows for applying these metrics, particularly in studies involving artifact removal.
Benchmarking with Semi-Synthetic Datasets
A rigorous and controlled approach for evaluating artifact removal techniques involves the use of semi-synthetic datasets [15].
Protocol:
- Acquire Clean EEG: Record a baseline of clean EEG data from subjects in a resting state or during a task unrelated to the artifacts being studied.
- Generate or Record Artifacts: Synthetically generate noise waveforms that mimic specific artifacts (e.g., tES stimulation pulses, ocular blinks, EMG bursts) or record these artifacts separately.
- Create Synthetic Dataset: Add the known artifacts to the clean EEG data at controlled amplitudes. This creates a dataset where the ground-truth, clean EEG is known, enabling precise calculation of all metrics.
- Apply Denoising Algorithm: Process the contaminated signal with the artifact removal technique under evaluation (e.g., a deep learning model, ICA, or wavelet-based filter).
- Calculate Metrics: Compute SNR, CC, and RMSD by comparing the algorithm's output against the known ground-truth signal.
Figure 1: Experimental workflow for benchmarking artifact removal techniques using a semi-synthetic dataset with a known ground truth.
Formulas and Calculation Steps
Table 2: Calculation Formulas for Key Metrics
| Metric | Key Formula(s) | Parameters |
|---|---|---|
| Signal-to-Noise Ratio (SNR) | ( \text{SNR}{\text{dB}} = 10 \log{10}\left(\frac{P{\text{signal}}}{P{\text{noise}}}\right) )Where ( P ) denotes the average power of the signal and noise components. | - ( P{\text{signal}} ): Power of the neural signal of interest.- ( P{\text{noise}} ): Power of the artifact or noise. |
| Correlation Coefficient (CC) | ( CC = \frac{ \sum{i=1}^{n} (xi - \bar{x}) (yi - \bar{y}) }{ \sqrt{ \sum{i=1}^{n} (xi - \bar{x})^2 } \sqrt{ \sum{i=1}^{n} (y_i - \bar{y})^2 } } ) | - ( xi ): Sample from the ground-truth signal.- ( yi ): Corresponding sample from the cleaned signal.- ( \bar{x}, \bar{y} ): Means of the respective signals. |
| Root Mean Square Deviation (RMSD) | ( \text{RMSD} = \sqrt{ \frac{1}{n} \sum{i=1}^{n} (yi - xi)^2 } )( \text{RRMSE} = \sqrt{ \frac{ \frac{1}{n} \sum{i=1}^{n} (yi - xi)^2 }{ \frac{1}{n} \sum{i=1}^{n} (xi)^2 } } ) | - ( xi ): Ground-truth signal sample.- ( yi ): Cleaned signal sample.- ( n ): Total number of samples. |
The Scientist's Toolkit: Research Reagents & Materials
Table 3: Essential Resources for EEG Artifact Removal Research
| Item / Technique | Category | Function in Evaluation |
|---|---|---|
| Semi-Synthetic Datasets | Data | Provides a controlled benchmark with known ground truth for rigorous metric calculation [15]. |
| Public EEG Datasets (e.g., Bonn, PhysioNet) | Data | Offers standardized, real-world data for comparative algorithm testing and validation [79] [80]. |
| Independent Component Analysis (ICA) | Algorithm | A blind source separation technique used to isolate and remove artifact components from EEG signals [81] [5]. |
| Wavelet Transform | Algorithm | Provides time-frequency analysis ideal for isolating and removing non-stationary artifacts [81] [80]. |
| Deep Learning Models (CNN, LSTM, Hybrid) | Algorithm | Advanced models for end-to-end artifact removal that can adapt to complex noise patterns [15] [79]. |
| Auditory Oddball Paradigm | Experimental Protocol | A classic task for generating Event-Related Potentials (e.g., P300) used to benchmark system performance and SNR [77]. |
Frequently Asked Questions (FAQs) & Troubleshooting
Q1: After applying my artifact removal algorithm, the SNR has improved, but the Correlation Coefficient has dropped significantly. What does this mean?
A: This is a classic sign of over-filtering or signal distortion. Your algorithm is likely removing too much of the neural signal along with the noise, thereby altering the original waveform's shape. The high SNR suggests the remaining signal is clean, but the low CC indicates it is no longer a faithful representation of the original brain activity.
- Troubleshooting Steps:
- Soften parameters: Review your algorithm's thresholding or rejection parameters and make them less aggressive.
- Inspect components: If using a method like ICA, re-examine the components you are rejecting to ensure they do not contain relevant neural information.
- Visual inspection: Always visually compare the input (contaminated), ground truth (if available), and output (cleaned) signals to identify where distortion is occurring.
Q2: My RMSD value is low, but my colleagues report that the cleaned signal still appears noisy upon visual inspection. Why is there a discrepancy?
A: A low RMSD indicates that the average error is small, but it may not capture specific, localized artifacts.
- Troubleshooting Steps:
- Check for localized noise: RMSD averages error across the entire epoch. Short-duration, high-amplitude artifacts (e.g., a muscle twitch) might not significantly impact the overall average but can be critically disruptive. Use additional metrics like peak-to-peak amplitude or visual inspection to catch these.
- Use complementary metrics: Rely on a suite of metrics, not just one. Calculate the Signal-to-Noise Ratio (SNR) in addition to RMSD to get a better picture of noise levels [77] [21].
- Analyze in segments: Calculate RMSD for shorter, overlapping windows of the signal to identify epochs where the error is concentrated.
Q3: How can I reliably calculate SNR when a true "clean" reference signal is not available (a common scenario with real EEG data)?
A: This is a fundamental challenge. In the absence of a ground truth, proxy measures must be used.
- Troubleshooting Steps:
- Pre-stimulus baseline: For evoked potentials, the EEG segment immediately before the stimulus is often used as a noise estimate. The SNR can then be calculated as the ratio of the post-stimulus signal power to this baseline noise power.
- Template matching: If you have multiple trials, you can create an average template (assuming the signal is time-locked and the noise is random). The noise in each trial can be estimated as the deviation from this template.
- Statistical methods: Techniques like the Huang-Hilbert Transform can be used to calculate the instantaneous frequency and amplitude, from which SNR-like measures can be derived in the time-frequency domain [80].
Q4: For a comprehensive evaluation of my artifact removal method, which combination of metrics is most informative?
A: A multi-faceted evaluation is essential. A strong benchmark should include:
- A temporal domain metric: RMSD or RRMSE to quantify point-by-point accuracy [15].
- A similarity metric: Correlation Coefficient (CC) to ensure waveform morphology is preserved [15] [78].
- A spectral or quality metric: SNR to assess the clarity of the final signal for downstream analysis [77].
- Application-specific performance: Ultimately, test how the cleaning affects your final goal, such as the classification accuracy in a Brain-Computer Interface (BCI) task [81] [79]. An effective method should improve all these metrics simultaneously.
Benchmarking Traditional vs. Machine Learning Approaches
Frequently Asked Questions (FAQs)
Q1: When should I choose a traditional method over a deep learning model for EEG denoising? Traditional methods like Independent Component Analysis (ICA) or wavelet thresholding are preferable when you have limited computational resources, need model interpretability, or are working with small datasets [82] [5]. They are also established choices for specific, well-defined artifacts like ocular movements when reference signals are available [4] [82]. However, for complex, non-stationary artifacts like muscle noise (EMG) that have spectral overlap with neural signals, deep learning models generally achieve superior performance [83] [82].
Q2: My deep learning model removes noise effectively but also seems to distort the underlying neural signal. How can I address this? This is a common trade-off between noise suppression and signal fidelity [83] [82]. To mitigate it, you can:
- Modify the Loss Function: Use a composite loss function that includes a temporal correlation metric to explicitly enforce the preservation of original signal dynamics [83] [84].
- Try a Different Architecture: Studies indicate that WGAN-GP often achieves a better balance between artifact removal and detail preservation compared to standard GANs, leading to lower error rates [83]. Alternatively, hybrid models like CLEnet, which combine CNNs and LSTMs, are specifically designed to extract and preserve morphological and temporal features of the genuine EEG [4].
Q3: For a real-time BCI application, which denoising approach is most suitable? Computational efficiency is critical for real-time systems. While complex models like transformers or hybrid CNN-LSTMs offer high performance, they have significant computational demands [82]. For real-time scenarios, the following are more appropriate:
- Adaptive Filtering: Techniques like adaptive noise cancellation are computationally efficient and effective for known, predictable noise sources like power line interference [85] [5].
- Lightweight Deep Models: Simpler CNN architectures or shallow autoencoders provide a good balance between denoising power and speed, making them suitable for low-latency applications [82] [86].
Q4: How do I handle artifacts in data from wearable EEG systems with a low number of channels? Low-channel-count configurations limit the effectiveness of source separation methods like ICA [5] [87]. Recommended approaches include:
- Channel-Aware Deep Learning: Use models specifically designed for multi-channel input that can learn inter-channel correlations, such as CLEnet [4].
- Single-Channel Focused Models: Deploy algorithms based on CNNs with attention mechanisms (e.g., CNN-CBAM) that are trained to detect and localize artifacts within a single channel of data, which is common in wearable sleep research [87].
Troubleshooting Guides
Issue 1: Poor Generalization of Deep Learning Model to New Data
Problem: Your trained denoising model performs well on your test set but fails on new EEG recordings from a different dataset or subject.
Solutions:
- Data Diversity: Ensure your training dataset includes a wide variety of artifact types, subjects, and recording conditions. Using semi-synthetic datasets, where clean EEG is artificially contaminated with noise, can help create a robust benchmark with known ground truth [15] [4].
- Leverage Domain Adaptation: Consider using a Mixture-of-Experts (MoE) framework. This involves training several specialized "expert" models on different signal-to-noise ratio (SNR) ranges or artifact subtypes, and a "gating" network that learns to select the best expert for a given input signal [84].
- Architecture Choice: Opt for models proven to generalize well across various artifacts. For instance, State Space Models (SSMs) have shown excellent performance in removing complex tACS and tRNS artifacts, which are highly dynamic [15].
Issue 2: Ineffective Removal of Specific High-Amplitude Artifacts
Problem: The pipeline fails to adequately remove a specific artifact, such as EMG from jaw clenching or EOG from blinks.
Solutions:
- Targeted Model Selection: No single model is best for all artifacts. Benchmark your data against several architectures. Research shows Complex CNNs are highly effective for tDCS artifacts, while SSM-based models excel on tACS and tRNS noise [15]. For EMG artifacts, a novel MoE framework that partitions EMG into quantifiable subtypes has been shown to improve performance in high-noise settings [84].
- Pre-processing with Traditional Methods: Consider a hybrid pipeline. Use a traditional method like wavelet transform for initial, coarse removal of high-amplitude artifacts, then apply a deep learning model for finer denoising of the residual signal [82] [5].
Issue 3: High Computational Cost and Slow Processing Speed
Problem: The denoising process is too slow for your application or requires excessive computational resources.
Solutions:
- Model Simplification: Forgo large, monolithic models. Explore efficient architectures like CLEnet, which uses dual-scale CNNs and an efficient attention mechanism to achieve high performance with a lower computational footprint [4].
- Dimensionality Reduction: Apply feature selection before classification. Mutual Information (MI)-based feature selection can dramatically reduce input dimensionality (e.g., by 75%), which significantly decreases computational load and can even improve model robustness and accuracy [86].
- Hardware Acceleration: Implement model inference using GPU acceleration or dedicated neuromorphic hardware, which is particularly efficient for running spiking neural networks (SNNs) [85] [86].
Performance Benchmarking Table
The table below summarizes the quantitative performance of various denoising approaches as reported in recent literature, providing a guide for method selection.
| Method Category | Specific Model/Technique | Best For Artifact Type | Key Performance Metrics | Reported Advantages |
|---|---|---|---|---|
| Deep Learning (Hybrid) | CLEnet (CNN + LSTM + EMA-1D) [4] | Mixed (EMG+EOG), Unknown | SNR: 11.50 dB, CC: 0.93, RRMSEt: 0.30 | Superior in multi-channel & unknown artifact removal |
| Deep Learning (Generative) | WGAN-GP [83] | Non-linear, Time-varying | SNR: 14.47 dB, PSNR: 19.28 dB | High training stability, good noise suppression |
| Deep Learning (Hybrid) | M4 (State Space Models) [15] | tACS, tRNS | Best RRMSE & CC for tACS/tRNS | Excels at complex, oscillatory artifacts |
| Deep Learning (Hybrid) | Complex CNN [15] | tDCS | Best RRMSE & CC for tDCS | Optimal for direct current stimulation artifacts |
| Statistical Framework | Mixture-of-Experts (MoE) [84] | EMG (High Noise) | Competitive SOTA performance | Superior lower-bound performance in high-noise settings |
| Traditional | Wavelet Transform + Thresholding [83] [82] | Ocular, Motion | Varies with parameters | Handles non-stationary noise, interpretable |
| Traditional | ICA / Adaptive Filtering [82] [85] | Ocular, Powerline | Varies with data & reference | Established, computationally efficient for specific artifacts |
Experimental Protocol for Method Benchmarking
To rigorously benchmark traditional versus machine learning approaches for EEG denoising, follow this structured experimental protocol.
1. Data Preparation and Preprocessing
- Dataset: Use a public benchmark dataset like EEGdenoiseNet [4] [84] or create a semi-synthetic dataset by adding known artifacts to clean EEG recordings [15] [4]. This provides a controlled environment with a ground truth for evaluation.
- Preprocessing: Resample all data to a consistent sampling rate (e.g., 250 Hz). Apply a band-pass filter (e.g., 0.5-30 Hz) and a notch filter (50/60 Hz) to remove baseline drift and power line interference [86]. Normalize the signals.
2. Model Implementation & Training
- Select Models: Choose a representative set of models from different categories (e.g., ICA, Wavelet, CNN, LSTM, GAN, SSM, MoE).
- Training Setup: For deep learning models, use mean squared error (MSE) or a composite loss function as the objective [82]. Optimize using the ADAM or RMSProp optimizer. Standardize the training data for all models to ensure a fair comparison.
3. Quantitative Evaluation
- Core Metrics: Calculate the following metrics by comparing the denoised output to the ground truth clean EEG:
- Advanced Metrics: Consider Peak Signal-to-Noise Ratio (PSNR) and Dynamic Time Warping (DTW) to assess signal fidelity and structural preservation [83].
The following workflow diagram illustrates the key stages of this benchmarking protocol:
Research Reagent Solutions
This table outlines key computational "reagents" – datasets, models, and metrics – essential for conducting EEG denoising research.
| Resource Type | Name / Example | Function / Purpose |
|---|---|---|
| Benchmark Dataset | EEGdenoiseNet [4] [84] | Provides clean EEG, EMG, and EOG signals to create semi-synthetic data with known ground truth for controlled model evaluation. |
| Deep Learning Model | CLEnet [4] | A hybrid CNN-LSTM model with an attention mechanism for removing various artifact types from multi-channel EEG. |
| Deep Learning Model | M4 (State Space Model) [15] | A multi-modular network designed to excel at removing complex, oscillatory artifacts like those from tACS and tRNS. |
| Statistical Framework | Mixture-of-Experts (MoE) [84] | A framework that uses multiple specialized sub-models to handle different noise levels or types, improving performance in high-noise settings. |
| Evaluation Metric | RRMSE (Relative Root Mean Squared Error) [15] | Quantifies reconstruction error in both temporal (RRMSEt) and spectral (RRMSEf) domains. |
| Evaluation Metric | Correlation Coefficient (CC) [15] [83] | Measures how well the temporal dynamics of the clean neural signal are preserved after denoising. |
Performance Analysis Across Different Artifact Types and Recording Conditions
Frequently Asked Questions (FAQs)
Q1: What are the most common types of artifacts that affect EEG recordings? EEG signals are susceptible to various physiological and non-physiological artifacts. Key physiological artifacts include:
- Electrooculographic (EOG): Caused by eye movements and blinks.
- Electromyographic (EMG): Generated by muscle tension, especially from the head, jaw, and neck.
- Electrocardiographic (ECG): The heartbeat's electrical signal can sometimes be picked up. Non-physiological artifacts include spatial electromagnetic noise and poor electrode contact. These artifacts are challenging because their frequency spectra often overlap with the genuine neural signals of interest [4] [29].
Q2: My EEG data is contaminated with unknown artifacts. Which removal method is most robust? For data with unknown or mixed artifacts, recent deep learning models show significant promise. The CLEnet model, which integrates dual-scale CNNs and LSTM with an attention mechanism, was specifically tested on a dataset containing "unknown artifacts" and showed superior performance. It achieved a 2.45% improvement in Signal-to-Noise Ratio (SNR) and a 2.65% improvement in the average correlation coefficient (CC) compared to other models, making it highly robust for complex, real-world scenarios [4].
Q3: How do traditional artifact removal methods compare to modern deep learning approaches? Traditional and deep learning methods have distinct strengths and limitations, as summarized in the table below.
| Method Category | Examples | Key Advantages | Key Limitations |
|---|---|---|---|
| Traditional Methods | Regression, Filtering, Blind Source Separation (BSS) like ICA [4] [29] | Well-established; BSS methods can be effective without deep learning infrastructure. | Often require reference signals or manual inspection; less effective with overlapping frequencies [4] [29]. |
| Modern Deep Learning | Nested GAN [65], CLEnet [4], DWT-CNN-BiGRU [31] [88] | End-to-end automated removal; superior at handling complex and unknown artifacts; achieves high performance metrics [65] [31] [4]. | Requires large datasets for training; computationally intensive to develop [4]. |
Q4: Are there artifact removal solutions suitable for real-time processing in BCI systems? Yes, performance is a critical factor for Brain-Computer Interface (BCI) systems. The nested Generative Adversarial Network (GAN) method was proposed explicitly for an effective end-to-end EEG artifact removal method essential for BCI. Furthermore, the GEDAI algorithm is noted for being remarkably fast, with processing times on par with real-time capable algorithms, making it an excellent candidate for real-time applications [65] [29].
Troubleshooting Guides
Issue 1: Poor Classification Accuracy Due to Muscular (EMG) Artifacts
Problem: Your deep learning model for classifying EEG signals (e.g., alcoholic vs. control subjects) is underperforming due to strong EMG contamination.
Solution: Implement a Discrete Wavelet Transform (DWT) based preprocessing pipeline.
- Recommended Technique: DWT combined with a CNN-BiGRU model [31] [88].
- Experimental Protocol:
- Data Preparation: Use a publicly available dataset (e.g., from Kaggle) with 64 electrodes and a 256 Hz sampling rate [31].
- Preprocessing: Apply DWT to the raw EEG signals for denoising. DWT is effective because it localizes artifacts in both time and frequency domains [31].
- Standard Scaling: Normalize the data to improve model convergence [31].
- Model Architecture & Training:
- Feature Extraction: Use Convolutional Neural Network (CNN) layers to extract spatial features from the denoised signals [31] [88].
- Temporal Dependencies: Use Bidirectional Gated Recurrent Unit (BiGRU) layers to capture long-range temporal dependencies in the EEG data [31] [88].
- Classification: Employ a final dense layer with a softmax activation for binary classification.
- Validation: This approach has been shown to achieve up to 94% accuracy, 0.94 precision, 0.95 recall, and a 0.94 F1-score on classification tasks, significantly outperforming models using DFT or DCT preprocessing [31] [88].
Issue 2: Handling Mixed and Unknown Artifacts in Multi-Channel Data
Problem: You are working with multi-channel EEG data contaminated by a mixture of unknown artifact types, and standard methods are failing.
Solution: Employ the CLEnet deep learning architecture, designed for this specific challenge.
- Recommended Technique: The CLEnet model, which combines Dual-scale CNN, LSTM, and an Efficient Multi-Scale Attention mechanism (EMA-1D) [4].
- Experimental Protocol:
- Data: Use a 32-channel EEG dataset containing unknown artifacts. CLEnet was benchmarked on a custom dataset for this purpose [4].
- Model Architecture & Workflow: The process involves three key stages, visualized in the workflow below.
- Training: Train the model in a supervised manner using Mean Squared Error (MSE) as the loss function to reconstruct clean EEG from artifact-contaminated input [4].
- Performance: On a multi-channel artifact removal task, CLEnet achieved the best performance, with an SNR of 11.498 dB and a CC of 0.925 for mixed artifacts, outperforming other mainstream models like 1D-ResCNN and DuoCL [4].
Issue 3: Need for a Fast, Theoretically-Informed Method Without Manual Intervention
Problem: You need a fully automated, fast denoising method that does not rely on manual component selection or "clean" data segments for training.
Solution: Implement the GEDAI (Generalized Eigenvalue De-Artifacting Instrument) algorithm.
- Recommended Technique: GEDAI, which uses leadfield filtering and the SENSAI index [29].
- Experimental Protocol:
- Theoretical Blueprint: GEDAI starts with a pre-computed "leadfield" model, which is a forward model of how brain-sourced electrical activity projects onto the scalp [29].
- Data Comparison: It takes a chunk of your recorded, noisy EEG data and uses Generalized Eigenvalue Decomposition (GEVD) to compare it against the clean leadfield blueprint [29].
- Automated Thresholding: The SENSAI (Signal & Noise Subspace Alignment Index) automatically finds the optimal threshold to separate brain signals from noise without manual input [29].
- Signal Reconstruction: The EEG is reconstructed using only the components identified as genuine brain activity [29].
- Performance: GEDAI has been tested on heavily contaminated data and outperforms PCA and ICA methods, especially in low Signal-to-Noise Ratio (SNR) conditions. It is also fast, with processing times up to 15 times faster than ICA-based methods, making it suitable for real-time applications [29].
The following tables consolidate key quantitative findings from recent studies to facilitate easy comparison of techniques.
Table 1: Performance of Deep Learning Models on Specific Tasks
| Model | Artifact Type | Key Metric 1 | Key Metric 2 | Key Metric 3 | Key Metric 4 |
|---|---|---|---|---|---|
| Nested GAN [65] | General / Mixed | MSE = 0.098 | PCC = 0.892 | η_temporal = 71.6% | η_spectral = 76.9% |
| CLEnet [4] | Mixed (EMG+EOG) | SNR = 11.498 dB | CC = 0.925 | RRMSE_t = 0.300 | RRMSE_f = 0.319 |
| DWT-CNN-BiGRU [31] [88] | N/A (Classification) | Accuracy = 94% | F1-Score = 0.94 | Precision = 0.94 | Recall = 0.95 |
Table 2: Performance Comparison Against Benchmark Models
| Model | Comparison Context | SNR Improvement | CC Improvement | RRMSE_t Reduction | RRMSE_f Reduction |
|---|---|---|---|---|---|
| CLEnet [4] | vs. Benchmarks (Multi-channel) | +2.45% | +2.65% | -6.94% | -3.30% |
| CLEnet [4] | vs. DuoCL (ECG artifacts) | +5.13% | +0.75% | -8.08% | -5.76% |
The Scientist's Toolkit: Research Reagent Solutions
This table details essential computational tools and datasets used in the featured experiments.
| Item Name | Type | Function / Explanation | Example Use Case |
|---|---|---|---|
| EEGdenoiseNet [4] | Benchmark Dataset | A semi-synthetic dataset providing clean EEG and artifact (EMG, EOG) signals, allowing for controlled performance evaluation. | Training and benchmarking new deep learning models for artifact removal [4]. |
| GEDAI EEGLAB Plugin [29] | Software Tool | An open-source plugin for the popular EEGLAB toolbox that implements the unsupervised GEDAI denoising algorithm. | Rapid, automated cleaning of EEG datasets without the need for model training [29]. |
| CLEnet Model [4] | Deep Learning Architecture | A ready-to-train model code integrating CNN, LSTM, and attention mechanisms for robust artifact removal from multi-channel EEG. | Addressing complex artifact scenarios with unknown noise sources in research data [4]. |
| DWT-CNN-BiGRU Pipeline [31] [88] | Processing Pipeline | A combined method using Discrete Wavelet Transform for denoising and a hybrid deep learning model for subsequent analysis (e.g., classification). | Improving the accuracy of EEG-based classification tasks (e.g., alcoholism detection) by enhancing signal quality [31] [88]. |
Validation Frameworks for Semi-Synthetic and Real EEG Datasets
Frequently Asked Questions
Q1: What are the main advantages and disadvantages of using semi-synthetic versus real EEG datasets for validation?
Semi-synthetic datasets are created by adding well-defined artifacts (like EOG or EMG) to clean EEG recordings [4]. This approach provides a ground truth, allowing for precise, quantitative evaluation of artifact removal algorithms using metrics like Signal-to-Noise Ratio (SNR) and Correlation Coefficient (CC) [4]. However, a key limitation is that they may not fully capture the complexity of real-world, unknown artifacts [4]. Real datasets contain authentic, complex artifacts but lack a perfect ground truth, making quantitative assessment more challenging and often requiring expert visual inspection for validation [89].
Q2: My deep learning model performs well on a semi-synthetic test set but fails on real-world data. What could be the cause?
This is a common problem indicating a generalization gap. The likely cause is that the semi-synthetic data used for training did not adequately represent the full diversity and complexity of artifacts found in real recordings. Your model may have overfitted to the specific, known artifacts in your synthetic set (like the particular EOG patterns you added) and cannot handle "unknown" artifacts present in the real data [4]. To address this, ensure your semi-synthetic training data includes a wide variety of artifact types and strengths. It is also crucial to incorporate a final validation step using a real, high-quality dataset where artifacts have been meticulously identified by experts [89].
Q3: What are the key quantitative metrics for validating artifact removal performance on semi-synthetic data?
When a ground truth is available, the following metrics are essential for a comprehensive evaluation [4]:
- Signal-to-Noise Ratio (SNR): Measures the power of the desired signal relative to the noise. Higher values indicate better artifact removal.
- Average Correlation Coefficient (CC): Quantifies the similarity between the cleaned signal and the original clean EEG. A value closer to 1 is better.
- Relative Root Mean Square Error (in Temporal and Frequency domains): Measures the error between the cleaned and original signal. Lower values are better.
The table below summarizes these core metrics:
Table 1: Key Quantitative Metrics for Validating Artifact Removal on Semi-Synthetic EEG Data
| Metric | Description | Interpretation |
|---|---|---|
| Signal-to-Noise Ratio (SNR) | Ratio of signal power to noise power | Higher value = Better performance |
| Correlation Coefficient (CC) | Linear correlation between cleaned and pure signal | Closer to 1 = Better performance |
| Relative Root Mean Square Error (RRMSE) | Error in temporal (t) or frequency (f) domain | Lower value = Better performance |
Q4: How can I validate an artifact removal method when I only have a real dataset with no ground truth?
In the absence of a ground truth, a multi-faceted approach is necessary:
- Visual Inspection: Always perform a thorough visual comparison of the data before and after processing across all channels [89]. This helps identify obvious failures or distortions of the neural signal of interest.
- Downstream Task Performance: A highly effective validation method is to test whether the cleaning process improves performance on a downstream task, such as motor imagery classification accuracy or emotion recognition accuracy [90] [91]. If cleaning the data leads to better task performance, it is a strong indicator of effectiveness.
- Expert Annotation: Use datasets where artifacts have been manually marked by human experts as a benchmark [89]. Compare your algorithm's output to these expert annotations.
- Quality Metrics: Calculate signal quality metrics from the cleaned data, such as the presence of expected physiological patterns (e.g., event-related desynchronization in motor imagery tasks) [90].
Q5: What is a robust experimental protocol for benchmarking a new artifact removal algorithm?
A robust benchmarking protocol should evaluate both reconstruction fidelity and practical utility. The following workflow is recommended:
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools and Datasets for EEG Artifact Removal Research
| Tool / Resource | Type | Primary Function in Research |
|---|---|---|
| EEGdenoiseNet [4] | Benchmark Dataset | Provides semi-synthetic data with ground truth for standardized evaluation of artifact removal algorithms. |
| HBN-EEG Dataset [7] | Real EEG Dataset | A large-scale, publicly available dataset suitable for testing generalization on real, complex data. |
| Independent Component Analysis (ICA) [92] [89] | Algorithm | A classic blind source separation technique used to isolate and remove artifacts like eye blinks and heartbeats. |
| CLEnet [4] | Deep Learning Model | An end-to-end network combining CNN and LSTM designed to remove various artifact types from multi-channel EEG. |
| Wavelet-Packet Decomposition [90] | Signal Processing Method | Used for data augmentation and channel selection by analyzing spectral-energy complexity. |
| FieldTrip Toolbox [89] | Software Toolbox | Provides a comprehensive set of functions for EEG/MEG analysis, including visual and automatic artifact rejection. |
Detailed Experimental Protocols
Protocol 1: Creating and Using a Semi-Synthetic Dataset
This protocol is based on methodologies used to benchmark modern deep learning models like CLEnet and others [4].
- Source Clean EEG: Obtain clean EEG segments from a trusted source, such as resting-state recordings from a public database or clean segments extracted from your own data using advanced cleaning methods.
- Source Artifacts: Record or obtain pure artifact signals. This includes:
- EOG: From frontal electrodes during eye blinks and movements.
- EMG: From temporal electrodes during jaw clenching or swallowing.
- ECG: From electrodes placed to capture the heartbeat.
- Linear Mixing: Artificially contaminate the clean EEG segments by adding the artifact signals at varying signal-to-noise ratios. A common formula is:
Contaminated_EEG = Clean_EEG + α * Artifact, whereαis a scaling factor that controls the contamination level [4]. - Dataset Splitting: Ensure the clean EEG data used for creating the training, validation, and test splits are from separate recording sessions or subjects to prevent data leakage and ensure a fair evaluation of model generalization.
Protocol 2: Validating with a Downstream Task (e.g., Motor Imagery Classification)
This protocol validates the practical utility of artifact removal by testing if it improves performance on a meaningful BCI task [90].
- Dataset: Use a standard public MI dataset, such as BCI Competition IV 2a [90].
- Preprocessing: Apply your artifact removal method to the raw MI data.
- Feature Extraction & Classification: Use a standardized pipeline (e.g., Common Spatial Patterns with an LDA classifier or a lightweight multi-branch network [90]) to decode the MI tasks from both the raw and cleaned data.
- Comparison: The key outcome is the change in classification accuracy. A significant improvement in accuracy on the cleaned data demonstrates the practical value of your artifact removal method. For example, a recent study showed that their augmentation and channel selection method led to accuracies over 86% on MI data [90].
Protocol 3: Component-Based Artifact Removal with ICA
This protocol outlines the use of ICA, a widely adopted method for removing well-defined physiological artifacts [92] [89].
- Preprocessing: The data is high-pass filtered (e.g., at 1 Hz) to remove slow drifts, which can improve the subsequent ICA decomposition.
- Decomposition: ICA is run on the preprocessed data. Mathematically, this calculates an "unmixing matrix" (W) that separates the recorded data (X) into statistically independent components (S), such that S = WX [92].
- Component Identification: The resulting components are inspected. Artifactual components are identified based on their:
- Topography: Eye blinks have strong frontal distributions; muscle noise is localized to temporal areas.
- Time Course: Blinks are large, infrequent spikes; cardiac artifacts are periodic.
- Frequency Spectrum: Muscle artifacts have high-frequency broadband activity.
- Reconstruction: The identified artifact components are removed, and the remaining components are projected back to the sensor space using the mixing matrix, resulting in cleaned EEG data [92] [89].
Computational Efficiency and Practical Implementation Considerations
This technical support resource provides practical guidance for researchers implementing electroencephalography (EEG) artifact removal techniques, focusing on computational efficiency and troubleshooting for experimental settings.
Troubleshooting Guides and FAQs
Q1: My deep learning denoiser performs well on test data but poorly on my new experimental recordings. What should I do? This indicates a generalization problem, common when training data doesn't match real-world conditions. Implement data augmentation by adding synthetic artifacts to your clean data [82]. Consider using models like the Artifact Removal Transformer (ART), which was trained on pseudo clean-noisy data pairs generated via ICA to improve generalization across datasets [3]. Alternatively, try GEDAI, which uses a theoretical leadfield model of the brain rather than relying solely on data-driven patterns [29].
Q2: How can I reduce processing time for denoising large-scale EEG datasets? For large datasets, choose algorithms with lower computational complexity. GEDAI processes data significantly faster than ICA-based methods—up to 15 times faster in some cases [29]. When using deep learning models, select architectures based on your computational constraints: shallow CNNs or simple autoencoders offer greater efficiency for real-time applications, while transformer-based models provide higher accuracy at greater computational cost [82].
Q3: What should I do when my denoising method removes neural signals along with artifacts? This over-cleaning problem often occurs when artifact and neural signals overlap. Methods that incorporate brain signal characteristics can help. Try GEDAI with its SENSAI component, which automatically finds the optimal threshold to maximize similarity between denoised data and theoretical brain activity patterns while minimizing similarity in discarded components [29]. Alternatively, combine spatial and temporal methods—research shows Fingerprint + ARCI + SPHARA combinations better preserve neural signals while removing artifacts [93].
Q4: Which denoising approach works best for dry EEG systems with more movement artifacts? Dry EEG is more susceptible to movement artifacts than gel-based systems [93]. Use a combination approach: start with ICA-based methods (Fingerprint + ARCI) to remove physiological artifacts, then apply spatial filtering (SPHARA) for additional noise reduction [93]. The improved SPHARA version, which includes zeroing of artifactual jumps in single channels before application, shows particularly good results for dry EEG [93].
Q5: How do I choose between traditional and deep learning methods for my BCI application? Consider your accuracy requirements and computational resources. Deep learning models like nested GANs and transformers generally achieve higher performance metrics but require more resources [65] [3] [82]. For real-time BCI applications with limited resources, faster methods like GEDAI or optimized traditional methods may be preferable [29]. Always validate chosen methods on data resembling your actual application.
Performance and Computational Efficiency Comparison
Table 1: Quantitative Performance of EEG Denoising Methods
| Method | Key Metrics | Computational Efficiency | Best Use Cases |
|---|---|---|---|
| Fingerprint + ARCI + improved SPHARA [93] | SD: 6.15 μV (from 9.76 μV); RMSD: 6.90 μV; SNR: 5.56 dB [93] | Moderate (multiple processing stages) | Dry EEG, movement artifacts |
| Nested GAN [65] | MSE: 0.098; PCC: 0.892; RRMSE: 0.065; ηtemporal: 71.6%; ηspectral: 76.9% [65] | High computational demand | Offline processing, high accuracy requirements |
| GEDAI [29] | Classification AUC: 0.80; Subject identification: 91% accuracy [29] | Fast (15x faster than ICA); real-time capable [29] | Real-time BCI, large datasets |
| ART (Transformer) [3] | Superior MSE and SNR vs. other DL models [3] | High computational demand | Multichannel EEG, multiple artifact types |
| ICA-based Methods [93] [82] | Varies by implementation | Moderate to high demand; slower than GEDAI [29] | Physiological artifact removal |
Table 2: Computational Characteristics of Deep Learning Architectures
| Model Type | Denoising Performance | Computational Efficiency | Implementation Considerations |
|---|---|---|---|
| Transformer (ART) [3] | High; captures transient dynamics well [3] | Lower; resource-intensive [82] | Best for offline analysis with sufficient resources |
| GAN-based [65] | High; good artifact suppression [65] | Lower; complex adversarial training [82] | Requires careful balancing of generator/discriminator |
| CNN & Autoencoder [82] | Moderate to good | Higher; efficient for real-time [82] | Good balance for many BCI applications |
| Hybrid Methods [93] [82] | Very good (complementary strengths) | Varies by combination | Can optimize efficiency by combining fast and accurate methods |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Computational Resources for EEG Denoising Research
| Item | Function/Purpose | Implementation Notes |
|---|---|---|
| Leadfield Model [29] | Theoretical blueprint of how brain activity projects to scalp electrodes; enables physically-informed denoising | Pre-computed; provides reference for distinguishing brain signals from artifacts |
| SENSAI Component [29] | Automatically finds optimal threshold for removing noisy components | Balances artifact removal with neural signal preservation |
| Independent Component Analysis (ICA) [93] [3] | Blind source separation for isolating artifact components | Used alone or to generate training data for deep learning models |
| Spatial Harmonic Analysis (SPHARA) [93] | Spatial filtering for noise reduction and dimensionality reduction | Particularly effective combined with temporal methods |
| Dry EEG Cap (64-channel) [93] | Rapid application for ecological experiments | More prone to movement artifacts than gel-based systems |
| eego Amplifier [93] | High-quality signal acquisition at 1,024 Hz sampling rate | Provides clean data foundation for subsequent denoising |
Experimental Workflow and Method Selection
Technical Support Center: Troubleshooting Guides and FAQs for EEG Research
This technical support center is designed for researchers, scientists, and drug development professionals utilizing electroencephalography (EEG) in pharmacodynamic research and clinical trials. Effective artifact removal and noise filtering are critical for obtaining clean neural data to accurately assess drug effects on brain function. The following guides and FAQs address common experimental challenges and present state-of-the-art solutions validated in recent research.
Troubleshooting Guide: Common EEG Data Quality Issues
Issue 1: Persistent High Impedance or Signal Abnormalities in Specific Electrodes
Problem: During setup, a reference (REF) electrode impedance remains high (indicated as grey in the recording software) despite repeated reapplication, potentially affecting all channels [14].
Systematic Diagnosis Protocol:
- Check Electrode/Cap Connections: Verify all plugs are secure. Re-clean and re-apply the problematic electrode, and consider swapping it with a known good electrode to rule out a hardware fault [14].
- Test Recording Hardware: Restart the recording software and amplifier. If possible, connect the participant to a different EEG system in another room. If the issue persists, this rules out problems with the original software, computer, or amplifier [14].
- Inspect the Headbox: Swap the headbox with another unit. If the issue remains, the headbox is likely not the cause [14].
- Isolate Participant-Specific Factors:
- Remove all metal accessories from the participant [14].
- Try alternative ground (GND) placements, such as the participant's hand, collarbone, or sternum. In documented cases, applying GND to the experimenter's hand or not connecting it resolved the issue, pointing to participant-specific "oversaturation" [14].
- Check for excessive electroconductive gel causing "bridging" between electrodes [14].
Recommended Action:
- For studies where EEG is a secondary variable, proceed with recording if a stable signal is achieved, as it may settle. For primary EEG outcomes, exhaust all troubleshooting steps; consider rescheduling if a clean signal is unattainable [14].
Issue 2: Ineffective Artifact Removal Blurring Pharmacodynamic Signals
Problem: Standard pre-processing, such as Independent Component Analysis (ICA) with component subtraction, inadvertently removes neural signals alongside artifacts. This can artificially inflate effect sizes (e.g., of event-related potentials) and bias source localization, leading to incorrect conclusions about drug effects [2].
Solution with Advanced Protocol: Implement a targeted artifact reduction method.
- Principle: Instead of fully subtracting artifactual components, cleaning is targeted only to the specific periods (for eye movements) or frequencies (for muscle artifacts) where the artifact manifests [2].
- Tool: Use the RELAX pipeline (freely available as an EEGLAB plugin) [2].
- Outcome: This method has been shown to better preserve neural signals, mitigate effect size inflation, and reduce source localization biases in tasks like the Go/No-Go and N400, thereby enhancing the validity of pharmacodynamic findings [2].
Frequently Asked Questions (FAQs)
Q1: What are the most effective modern methods for removing unknown or multiple artifact types from multi-channel EEG data?
A1: Deep learning models that integrate multiple architectural features show superior performance in handling complex, unknown artifacts in multi-channel data.
- CLEnet: This model integrates dual-scale Convolutional Neural Networks (CNN) and Long Short-Term Memory (LSTM) networks with an attention mechanism. It is specifically designed to extract both morphological and temporal features from EEG signals, enabling effective separation of neural data from various artifacts without requiring a reference signal. On a dataset with unknown artifacts, CLEnet improved the Signal-to-Noise Ratio (SNR) by 2.45% and the average Correlation Coefficient (CC) by 2.65% compared to other leading models [4].
- ART (Artifact Removal Transformer): A transformer-based, end-to-end model that effectively captures millisecond-scale EEG dynamics. It is trained on pseudo clean-noisy data pairs and can remove multiple artifact sources simultaneously, significantly improving subsequent brain-computer interface (BCI) performance [3].
Q2: How can we validate that our denoising protocol preserves genuine brain signals relevant to drug action?
A2: Beyond standard metrics, performance should be validated using downstream neurobehavioral prediction tasks.
- ERP Classification: After denoising with an advanced algorithm like GEDAI, the accuracy of classifying which image a participant saw based on a single-trial Event-Related Potential (ERP) soared to an Area Under the Curve (AUC) of 0.80, significantly outperforming other methods (0.72) [29]. This demonstrates enhanced sensitivity to neural correlates of cognition.
- Brain Fingerprinting: In a task to identify an individual from a group based on their unique resting-state EEG pattern, data cleaned with GEDAI achieved a 91% success rate, compared to 76% for the next-best algorithm [29]. This confirms the preservation of individual neural signatures.
Q3: Are there denoising solutions that leverage the physical properties of brain signal generation?
A3: Yes, the Generalized Eigenvalue De-Artifacting Instrument (GEDAI) uses a theoretically informed "leadfield filtering" approach.
- Principle: GEDAI uses a pre-computed "forward model" (leadfield) that describes how electrical activity generated inside the brain should project onto scalp electrodes. It compares recorded EEG to this blueprint of genuine brain activity and isolates components that do not match this spatial signature [29].
- Advantage: This unsupervised, fully automated method is fast and robust, especially in challenging conditions with heavy artifact contamination. It is available as an open-source EEGLAB plugin [29].
Comparison of State-of-the-Art EEG Denoising Algorithms
Table 1: Summary of Advanced EEG Denoising Methods for Research Application. SNR = Signal-to-Noise Ratio, CC = Correlation Coefficient, RRMSE = Relative Root Mean Square Error.
| Method Name | Core Principle | Best For Artifact Types | Key Performance Metrics | Availability |
|---|---|---|---|---|
| RELAX [2] | Targeted period/frequency cleaning of ICA components | Ocular, Muscle | Reduces effect size inflation & source bias | EEGLAB Plugin |
| EEGDfus [94] | Conditional Diffusion Model | Ocular (EOG) | CC: 0.983-0.992 for EOG removal | Code on GitHub |
| MSTP-Net [95] | Multi-Scale Temporal Propagation Network | General Artifacts | SNR: 12.76 dB, CC: 0.9221, RRMSE reduced by 21.7% (temporal) | Pre-trained model on GitHub |
| CLEnet [4] | Dual-scale CNN + LSTM + Attention | Unknown, Mixed, Multi-channel | SNR: +2.45%, CC: +2.65%, RRMSEt: -6.94% | Code on GitHub |
| GEDAI [29] | Leadfield Filtering & GEVD | Mixed (EOG, EMG, Noise) | 91% subject identification accuracy, fast processing | EEGLAB Plugin |
| ART [3] | Transformer Architecture | Multiple sources simultaneously | Superior MSE and SNR, improves BCI performance | Code on request |
Experimental Protocols for Key Denoising Methods
Protocol 1: Implementing the RELAX Pipeline for Targeted Artifact Reduction
Application: Ideal for cleaning EEG data from cognitive tasks (e.g., Go/No-Go, N400) in clinical trials to prevent bias in ERP and connectivity analyses [2].
Methodology:
- Data Input: Load preprocessed, continuous EEG data into EEGLAB.
- ICA Decomposition: Run ICA to decompose the data into independent components.
- RELAX Processing: Execute the RELAX plugin. The algorithm will: a. Identify components corresponding to artifacts (e.g., eye movements, muscle activity). b. Apply cleaning selectively, targeting only the time periods (for eye artifacts) or frequency bands (for muscle artifacts) where the artifact is present, rather than subtracting the entire component.
- Data Reconstruction: The cleaned data is reconstructed in the electrode space, now with artifacts reduced and neural signals better preserved [2].
Protocol 2: Denoising Multi-Channel EEG with CLEnet for Unknown Artifacts
Application: Suitable for real-world EEG data collected in non-laboratory settings or when artifacts cannot be easily classified, often encountered in long-term pharmacodynamic monitoring [4].
Methodology:
- Data Preparation: Format multi-channel EEG data into segments. Normalize the data.
- Model Loading: Load the pre-trained CLEnet model [4].
- Feature Extraction: a. Morphological Feature Extraction: The dual-branch CNN with different kernel sizes extracts features at multiple scales. An embedded EMA-1D attention module enhances temporal feature preservation during this stage. b. Temporal Feature Extraction: The extracted features are passed to an LSTM network to capture long-range temporal dependencies characteristic of genuine EEG.
- Signal Reconstruction: A fully connected layer decodes the fused features to reconstruct the artifact-free EEG signal [4].
Research Reagent Solutions: Essential Materials for EEG Denoising Research
Table 2: Key Computational Tools and Datasets for Developing and Validating EEG Denoising Methods.
| Item / Resource | Function / Description | Example Use in Research |
|---|---|---|
| EEGLAB | An open-source MATLAB environment for EEG analysis. | Serves as a platform for running and integrating denoising plugins like RELAX [2] and GEDAI [29]. |
| EEGDenoiseNet | A public benchmark dataset containing clean EEG and artifact signals [94] [95] [4]. | Used for training and fair comparison of deep learning models like EEGDfus and MSTP-Net. |
| Pre-trained Models (e.g., MSTP-Net, CLEnet) | Ready-to-use denoising neural networks available on code repositories. | Allows researchers to apply state-of-the-art denoising without the computational cost of training from scratch [95] [4]. |
| Semi-Synthetic Datasets | Data created by adding real artifacts (EOG, EMG) to clean EEG recordings. | Enables supervised training of deep learning models where the ground-truth clean signal is known [4] [3]. |
Workflow and Algorithm Diagrams
Diagram 1: Systematic EEG Troubleshooting Workflow
Systematic EEG Troubleshooting Workflow
Diagram 2: GEDAI Leadfield Filtering Principle
GEDAI Leadfield Filtering Principle
Diagram 3: Deep Learning Denoising Model Relationships
Deep Learning Denoising Model Relationships
Conclusion
EEG artifact removal remains a dynamic field where traditional methods like ICA and regression are effectively complemented by advanced deep learning approaches. The optimal denoising strategy depends on specific research requirements, artifact types, and recording conditions, with hybrid methods increasingly demonstrating superior performance. Future directions point toward more automated, real-time solutions capable of handling unknown artifacts in mobile recording environments. For drug development professionals, these advancements enable more precise pharmacodynamic modeling and cleaner biomarker extraction, ultimately enhancing the reliability of EEG-based endpoints in clinical trials. As the technology evolves, the integration of robust artifact removal pipelines will continue to be crucial for advancing both neuroscience research and therapeutic development.
References
- 1. EEG Artifacts: Types, Detection, and Removal Techniques ... [https://www.bitbrain.com/blog/eeg-artifacts]
- 2. EEG is better when cleaning effectively targets artifacts [https://pubmed.ncbi.nlm.nih.gov/41092567/]
- 3. Augmenting brain-computer interfaces with ART: An artifact ... [https://www.sciencedirect.com/science/article/pii/S1053811925001259]
- 4. A novel EEG artifact removal algorithm based on an ... [https://www.nature.com/articles/s41598-025-98653-1]
- 5. A Systematic Review of Techniques for Artifact Detection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473706/]
- 6. AnEEG: leveraging deep learning for effective artifact ... [https://www.nature.com/articles/s41598-024-75091-z]
- 7. EEG Challenge (2025) [https://eeg2025.github.io/eeg2025.github.io/]
- 8. Validation of an electroencephalography data assimilation- ... [https://www.nature.com/articles/s44172-025-00525-z]
- 9. EEG Artifacts: Overview, Physiologic ... [https://emedicine.medscape.com/article/1140247-overview]
- 10. Appendix 4. Common Artifacts During EEG Recording - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK390358/]
- 11. Optimizing EEG Signal Integrity: A Comprehensive Guide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11505294/]
- 12. Identifying the sources of the pulse artefact in EEG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3601330/]
- 13. How To Reduce Noise In EEG Recordings [11 Solutions] [https://mentalab.com/reduce-noise-in-eeg-recordings/]
- 14. EEG Trouble Shooting | Center for ... [https://www.centerforsleepandcognition.com/eeg-trouble-shooting]
- 15. A deep learning approach to artifact removal in ... [https://www.sciencedirect.com/science/article/pii/S030645222500990X]
- 16. A deep learning approach to artifact removal in ... [https://pubmed.ncbi.nlm.nih.gov/41101623/]
- 17. that goes Electrode in the night pop [https://www.thoracic.org/professionals/clinical-resources/sleep/sleep-fragments/electrode-pop.php]
- 18. Artifacts [https://www.learningeeg.com/artifacts]
- 19. TMSi — an Artinis company — Common non-physiological (external)... [https://www.tmsi.artinis.com/blog/common-non-physiological-external-eeg-artifacts]
- 20. Getting to know EEG artifacts and how to handle them in ... [https://pressrelease.brainproducts.com/eeg-artifacts-handling-in-analyzer/]
- 21. Signal to Noise in EEG - NMSBA [https://www.nmsba.com/neuromarketing-companies/neuromarketing-2021/signal-to-noise-in-eeg]
- 22. Removal of Artifacts from EEG Signals: A Review - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6427454/]
- 23. Expert Pharmacodynamic (PD) Modeling Solutions [https://xyzagen.com/solutions/quantitative-model-informed-drug-development/pharmacodynamic-modeling/]
- 24. Pharmacokinetics, Pharmacodynamics, and... | Altasciences [https://www.altasciences.com/resource-center/blog/pharmacokinetics-pharmacodynamics-and-toxicokinetics-demystified]
- 25. Basic Concepts in Population Modeling , Simulation, and Model -Based... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3917320/]
- 26. A Mechanistic Physiologically Based Pharmacokinetic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473830/]
- 27. EEG Signal Processing for Dummies [https://www.neuroelectrics.com/blog/eeg-signal-processing-for-dummies]
- 28. Blind Signal Separation - an overview [https://www.sciencedirect.com/topics/engineering/blind-signal-separation]
- 29. Noise-cancelling headphones for the Brain? How a New ... [https://cibm.ch/noise-cancelling-headphones-for-the-brain-how-a-new-algorithm-eliminates-noise-in-eeg-data/]
- 30. Electroencephalography Signal Processing [https://pmc.ncbi.nlm.nih.gov/articles/PMC10385593/]
- 31. Improving EEG classification of alcoholic and control ... [https://www.frontiersin.org/journals/neuroinformatics/articles/10.3389/fninf.2025.1618050/full]
- 32. Independent Component Analysis (ICA) – demystified [https://pressrelease.brainproducts.com/independent-component-analysis-demystified/]
- 33. Independent Component Analysis for artifact removal [https://eeglab.org/tutorials/06_RejectArtifacts/RunICA.html]
- 34. Independent Component Analysis of EEG data [https://eeglab.org/tutorials/ConceptsGuide/ICA_background.html]
- 35. Optimizing EEG ICA decomposition with data cleaning in ... [https://www.nature.com/articles/s41598-024-64919-3]
- 36. Estimating sensor-space EEG connectivity [https://www.sciencedirect.com/science/article/pii/S138824572500464X]
- 37. Based Wavelet for Filters Elimination in... Artifact [https://pubmed.ncbi.nlm.nih.gov/35994164/]
- 38. Spiking neural networks for EEG signal analysis using ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1652274/full]
- 39. Denoising of EEG signals using Discrete Wavelet ... [https://biomedpharmajournal.org/vol8no1/denoising-of-eeg-signals-using-discrete-wavelet-transform-based-scalar-quantization/]
- 40. Signal EEG and Analysis by Artifact Removal Wavelet Transform [https://link.springer.com/chapter/10.1007/978-3-319-11776-8_44]
- 41. Cleaning Up EEG Signals for Better Diagnosis - Simple Science [https://scisimple.com/en/articles/2025-10-08-cleaning-up-eeg-signals-for-better-diagnosis--akedyn1]
- 42. A Hybrid CNN-LSTM Approach for Muscle Artifact Removal ... [https://www.mdpi.com/2076-3417/15/9/4953]
- 43. A Bioinspired Multimodal CNN-LSTM Network for EEG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12655944/]
- 44. A One-Dimensional CNN-LSTM Model for Epileptic Seizure ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7772824/]
- 45. EEGMamba: Bidirectional State Space Model with Mixture ... [https://openreview.net/forum?id=13PclvlVBa]
- 46. Classification of EEG Signal Using Deep Learning ... [https://link.springer.com/article/10.1007/s42979-025-03743-6]
- 47. Single-channel EOG artifact removal using fixed frequency ... [https://www.nature.com/articles/s41598-025-10276-8]
- 48. A hybrid EMG–EEG interface for robust intention detection ... [https://www.nature.com/articles/s41598-025-24831-w]
- 49. Hybrid EEG Feature Learning Method for Cross-Session ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384435/]
- 50. A hybrid approach for EEG motor imagery classification ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482525010261]
- 51. NeuXus open-source tool for real-time artifact reduction in ... [https://www.sciencedirect.com/science/article/pii/S1053811923005049]
- 52. Review of challenges associated with the EEG artifact ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809421003384]
- 53. EEG artifact removal method - MNE Forum - Discourse [https://mne.discourse.group/t/eeg-artifact-removal-method/3669]
- 54. Self-Supervised EEG Denoising via Dual-Branch ... [https://www.sciencedirect.com/science/article/abs/pii/S0950705125017423]
- 55. Assessing the impact of artifact correction and artifact rejection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11888300/]
- 56. The future of brain activity monitoring may look like a strand ... [https://www.psu.edu/news/research/story/future-brain-activity-monitoring-may-look-strand-hair]
- 57. Precautions for good impedance measurements (EIS ... [https://www.biologic.net/documents/eis-precautions-electrochemistry-battery-application-note-5/]
- 58. Why Brain Wearables Are Making Traditional EEG Labs ... [https://globalrph.com/2025/10/why-brain-wearables-are-making-traditional-eeg-labs-obsolete-in-2025/]
- 59. Recent Advances in Portable Dry Electrode EEG [https://www.mdpi.com/1424-8220/25/16/5215]
- 60. Individual stability of single-channel EEG measures over ... [https://www.nature.com/articles/s41598-025-13614-y]
- 61. Comparison of Dry Electrode EEG System with ... - Zeto Inc. [https://zeto-inc.com/blog/comparison-of-dry-electrode-eeg-system-with-conventional-eeg-system/]
- 62. Systematic comparison between a wireless EEG system ... [https://www.sciencedirect.com/science/article/abs/pii/S1053811918307961]
- 63. Comparison of dry and wet electroencephalography for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11576458/]
- 64. Resting-state EEG recorded with gel-based vs. consumer ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2024.1326139/full]
- 65. End-to-End EEG Artifact Removal Method via Nested ... [https://pubmed.ncbi.nlm.nih.gov/41183389/]
- 66. 5 Basics of EEG 101: Data Collection, Processing & Analysis [https://imotions.com/blog/learning/best-practice/5-basics-eeg-data-processing/?srsltid=AfmBOordn-JXQnVpz54Iprmz4af2FojEjCYIfjVZsSmjfKPsvpojohhW]
- 67. Managing EEG studies: How to prepare and what to do once ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11276027/]
- 68. Improving EEG classification of alcoholic and control ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12401955/]
- 69. NeuralEnsemble/NeuroTools [https://github.com/NeuralEnsemble/NeuroTools]
- 70. A novel EEG artifact removal algorithm based on an advanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12134218/]
- 71. Open-Source EEG Analysis Tools: Making Brain Research ... [https://www.brainaccess.ai/open-source-eeg-analysis-tools-making-brain-research-more-accessible/]
- 72. Overview of the MNE tools suite [https://mne.tools/stable/install/mne_tools_suite.html]
- 73. 5. Preprocess data [https://eeglab.org/tutorials/05_Preprocess/]
- 74. neurokit2 [https://pypi.org/project/neurokit2/]
- 75. Motion Artifacts in Dynamic EEG Recordings [https://www.mdpi.com/1424-8220/24/19/6363]
- 76. EEG-cleanse: an automated pipeline for cleaning ... [https://www.sciencedirect.com/science/article/pii/S2215016125005461]
- 77. Proposing Metrics for Benchmarking Novel EEG Technologies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4861738/]
- 78. Evaluation Metric - an overview [https://www.sciencedirect.com/topics/computer-science/evaluation-metric]
- 79. Enhanced EEG signal classification in brain computer ... [https://www.nature.com/articles/s41598-025-07427-2]
- 80. Classification of ictal and interictal EEG using RMS ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809418300843]
- 81. Automatic Artifact Removal from Electroencephalogram ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4562337/]
- 82. A comprehensive review of deep learning models for ... [https://link.springer.com/article/10.1007/s42452-025-07808-2]
- 83. Frontiers | Adversarial denoising of EEG signals: a comparative ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1583342/full]
- 84. A Statistical Mixture-of-Experts Framework for EMG Artifact ... [https://arxiv.org/abs/2509.19385]
- 85. Mastering EEG for Enhanced BCI Performance [https://www.numberanalytics.com/blog/mastering-eeg-for-bci-performance]
- 86. seizure classification with temporal spiking neural networks and... EEG [https://jeas.springeropen.com/articles/10.1186/s44147-025-00796-5]
- 87. Artifact detection and localization in single-channel mobile ... [https://arxiv.org/html/2504.08469v2]
- 88. Improving EEG classification of alcoholic and control ... [https://pubmed.ncbi.nlm.nih.gov/40904893/]
- 89. Introduction on dealing with artifacts [https://www.fieldtriptoolbox.org/tutorial/preproc/artifacts/]
- 90. Motor imagery EEG classification via wavelet-packet ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1689647/full]
- 91. A machine learning-based EEG signal analysis framework ... [https://pubmed.ncbi.nlm.nih.gov/41215981/]
- 92. Removing Artifacts From EEG Data Using Independent ... - TMSi [https://www.tmsi.artinis.com/blog/removing-eeg-artifacts-with-independent-component-analysis-ica]
- 93. Frontiers | Combination of spatial and temporal de- noising and artifact ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1576954/full]
- 94. EEGDfus: A Conditional Diffusion Model for Fine-Grained ... [https://pubmed.ncbi.nlm.nih.gov/40030273/]
- 95. EEG noise reduction based on the Multi-Scale Temporal ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809425013151]