Adaptive Decoding Algorithms for Non-Stationary Neural Signals: From Theory to Clinical Translation
This article provides a comprehensive examination of adaptive decoding algorithms, which are critical for interpreting the non-stationary neural signals that underlie complex brain functions and neurological disorders.
Adaptive Decoding Algorithms for Non-Stationary Neural Signals: From Theory to Clinical Translation
Abstract
This article provides a comprehensive examination of adaptive decoding algorithms, which are critical for interpreting the non-stationary neural signals that underlie complex brain functions and neurological disorders. Aimed at researchers, scientists, and drug development professionals, it explores the foundational challenges of neural signal variability and the limitations of traditional analysis methods. The scope spans from core methodological innovations, including Bayesian adaptive designs and transformer-based architectures, to practical optimization strategies for enhancing robustness and computational efficiency. The content further delves into rigorous validation frameworks and comparative analyses, highlighting how these advanced algorithms are poised to revolutionize neurotherapeutic decision-making, precision brain imaging, and the development of personalized neural prostheses.
The Challenge of Neural Non-Stationarity: Foundations and Clinical Imperatives
Troubleshooting Guide: Frequently Asked Questions
What is a non-stationary neural signal, and why is it a problem for my analysis? A non-stationary neural signal is one whose statistical properties—such as mean firing rate, variance, and relationship to movement parameters—change over time [1] [2]. This is a problem because many standard neural decoding algorithms (e.g., linear regression or Kalman filters) are built on the assumption that these statistical properties are stationary. When this assumption is violated, the model's performance degrades over time, leading to inaccurate decoding of movement intentions or other neural states [2]. For instance, a neuron's mean firing rate might steadily increase while the animal's behavior remains consistent, breaking the fixed model relationship [2].
How can I visually identify non-stationarity in my recorded neural data? You can identify potential non-stationarity by plotting the average firing rates of individual neurons across many trials over the course of an experiment. If a substantial subpopulation (around 50% in some studies) shows significant trends or variations in their averaged firing rates while behavioral outputs are consistent, this is a key indicator of non-stationarity [2]. The figure below illustrates this concept.
My decoding model performance drops during long recording sessions. Is non-stationarity the cause? Yes, this is a classic symptom. Subjects may change their level of attention or engagement, and neural representations themselves can drift over time, making a model trained on initial data less accurate for later data [2]. The solution is to move from static to adaptive decoding models that update their parameters as new neural and behavioral observations come in [2].
The Fourier Transform of my signal is difficult to interpret. Is this related to non-stationarity? Yes. The standard Fourier Transform assumes signal properties are stable over time. For non-stationary signals, it conflates time and frequency information, obscuring when specific frequency components occur [3] [4]. This makes it poor for identifying transient events or tracking how neural oscillations change over time. You should use time-frequency analysis techniques like spectrograms or wavelet transforms instead [5] [6].
What are the main sources of variation in non-stationary neural signals? Trial-by-trial variation in neural signals can be broken down into two main components, as shown in the table below.
| Component of Variation | Description | Impact on Behavior |
|---|---|---|
| Shared Variation | Correlated fluctuations across a population of neurons, often from common input. This component is expressed as neuron-neuron latency correlations [7]. | Propagates through the sensory-motor circuit to drive trial-by-trial variation in behavioral latency and performance. It is challenging to eliminate by simple averaging [7]. |
| Independent Variation | Fluctuations local to individual neurons. Surprisingly, this arises more from the underlying probability of spiking (synaptic inputs) than from the stochasticity of spiking itself [7]. | Can be reduced by averaging across a large population of neurons [7]. |
Can you provide a practical example of an adaptive algorithm for handling non-stationarity? A common approach is the Adaptive Kalman Filter. While a standard Kalman filter uses fixed parameters, an adaptive version updates its parameters (the state transition and observation matrices) over time as new training data (neural activity and measured kinematics) becomes available [2]. This allows the model to "track" the dynamic relationship between neural firing and behavior. A recursive update method can make this process computationally efficient for real-time use [2]. The following diagram outlines a general adaptive decoding workflow.
Experimental Protocols & Methodologies
Protocol 1: Quantifying Neural and Behavioral Latency Relationships
This protocol is used to investigate how trial-by-trial variations in neural response latency relate to behavioral latency [7].
- Task: Train a subject (e.g., a non-human primate) to perform a step-ramp visual pursuit task. A target appears and begins moving; the subject must initiate smooth eye movement to track it [7].
- Recording: Implant a multielectrode array in the relevant brain area (e.g., motor cortex, area MT). Record single-unit activity and behavioral kinematics (e.g., hand or eye position) simultaneously over hundreds of trials [2] [7].
- Latency Estimation:
- Behavioral Latency: For each trial, use an objective algorithm to determine the time point at which smooth pursuit eye movement begins [7].
- Neural Latency: For each recorded neuron, create a spike density function for each trial. Use an objective method to estimate the response latency on a trial-by-trial basis [7].
- Analysis:
- Bin all trials for a given neuron into quintiles based on behavioral latency.
- Calculate the mean neural latency for each quintile.
- Perform regression analysis to determine the sensitivity (slope) of neural latency to behavioral latency. A slope of 1 would indicate a perfect one-to-one relationship [7].
Protocol 2: Cycle-by-Cycle Analysis of Neural Oscillations
This methodology moves beyond Fourier analysis to characterize the non-sinusoidal and non-stationary properties of neural oscillations, such as those in EEG or LFP recordings [4].
- Signal Preprocessing: Filter the raw neural signal into a frequency band of interest (e.g., theta, alpha).
- Cycle Detection: Identify individual oscillatory cycles in the filtered signal by finding consecutive peaks.
- Feature Extraction: For each cycle, compute the following time-domain features:
- Peak-Trough Symmetry: Calculate as P / (P + T), where P is the time from a zero-crossing to the next peak, and T is the time from a zero-crossing to the next trough. A value of 0.5 indicates a perfect sinusoid [4].
- Rise-Decay Symmetry: Calculate as R / (R + D), where R is the rise time from a zero-crossing to a peak and D is the decay time from a zero-crossing to a trough [4].
- Interpretation: Plot the distributions of these symmetry measures over many cycles. The mean and variance of these distributions reveal how much and how consistently the oscillations deviate from a pure sinusoid, providing a measure of aperiodicity and non-stationarity [4].
The Scientist's Toolkit: Key Research Reagents & Materials
| Item | Function in Research |
|---|---|
| Silicon Microelectrode Arrays (e.g., 100-electrode arrays) | Chronic implantation allows for long-term recording from a population of neurons in areas like motor cortex, critical for tracking non-stationarity over time [2]. |
| Multi-channel Neural Signal Acquisition System (e.g., Cerebus system) | Systems that can filter, amplify, and digitally record raw waveforms from all electrodes simultaneously at high sampling rates (e.g., 30 kHz) [2]. |
| Offline Spike Sorter (e.g., Plexon Offline Sorter) | Software used to isolate the activity of single units (individual neurons) from the recorded waveforms based on spike shape and other features [2]. |
| Robotic Arm or Kinematic Tracking System (e.g., KINARM) | Precisely measures and records the subject's behavioral output, such as joint angles or hand position, which is essential for modeling the neural-behavioral relationship [2]. |
| Time-Frequency Analysis Software | Software (e.g., custom MATLAB or Python scripts) to compute spectrograms, wavelet transforms, and other time-frequency distributions for analyzing non-stationary signal components [5]. |
Limitations of Traditional Machine Learning and Fixed Analysis Windows
Frequently Asked Questions (FAQs)
FAQ 1: What are the primary data-related limitations of traditional machine learning for neural signal analysis? Traditional machine learning (ML) models face significant challenges with neural data due to its non-stationary, non-linear, and non-Gaussian nature. These models are highly dependent on data quality and struggle with distributional shifts that occur across different recording sessions or between different subjects. This violation of the standard assumption that data samples are independently and identically distributed (i.i.d.) severely limits model generalizability [8] [9].
FAQ 2: How do fixed analysis windows hinder accurate neural decoding? Fixed analysis windows are often ineffective for neural signals because they cannot adapt to the dynamic nature of brain activity. This is particularly problematic in applications like Steady-State Visual Evoked Potential (SSVEP) decoding, where using short, fixed windows to increase the information transfer rate can cause a significant drop in decoding accuracy. These static windows fail to capture the evolving temporal patterns of neural responses [10].
FAQ 3: Why is model interpretability a problem in clinical neurotechnology? Complex models like deep neural networks often function as "black boxes," making it difficult to understand how they arrive at a specific prediction. This lack of transparency is a major barrier to clinical adoption, as doctors and researchers require explainability to trust and effectively use a model's output for diagnostic or therapeutic decisions [9] [11].
FAQ 4: What is the "cross-subject and cross-session" generalization problem? This refers to the challenge of a decoding model, trained on data from one set of subjects or one recording session, failing to perform accurately on data from new subjects or subsequent sessions. This is caused by the inherent variability and randomness of brain electrical activity between individuals and over time [8].
Troubleshooting Guides
Issue 1: Poor Model Performance on New Subjects or Sessions
Problem: Your trained model, which performed well on its original training data, shows significantly degraded accuracy when applied to data from a new subject or a new recording session from the same subject.
Solution: Implement Domain Adaptation (DA) techniques. DA helps to minimize the distributional differences between your source domain (original training data) and target domain (new subject/session data) [8].
- Step 1: Identify the DA Approach. Choose a method based on your data and goals:
- Feature-based DA: Transform the feature spaces of the source and target domains to make them more similar [8].
- Model-based DA: Fine-tune a pre-trained model from the source domain using a small amount of labeled data from the target subject/session [8].
- Instance-based DA: Adjust the weights of source domain samples, emphasizing those most relevant to the target domain [8].
- Step 2: Consider Advanced Architectures. For complex scenarios, use deep learning frameworks combined with DA. Transformer models with adaptive attention mechanisms have shown success in learning robust, subject-agnostic features from neural data like EEG [10] [11].
- Step 3: Validate Rigorously. Always test the adapted model on a completely held-out dataset from the target domain to ensure true generalizability and avoid overfitting [12].
Issue 2: Inability to Capture Dynamic Neural Patterns
Problem: Your model fails to decode short-term fluctuations in neural states, which is critical for applications like adaptive deep brain stimulation (aDBS).
Solution: Move beyond static features and fixed windows by implementing models that capture spatiotemporal dynamics.
- Step 1: Employ Adaptive Time-Series Models. Replace static analyzers with models designed for sequential data. LSTMs and Transformers can learn from the temporal context of the signal [11].
- Step 2: Leverage Temporal-Spatial Feature Extraction. Use models that can simultaneously analyze information across both time and the spatial arrangement of electrodes (channels). Transformer architectures are particularly effective at this, using multi-head self-attention to model long-range temporal dependencies and spatial attention for inter-channel interactions [11].
- Step 3: Integrate with a Closed-Loop System. Frame the decoding output to directly control stimulation parameters in real-time. This creates an intelligent adaptive DBS (iDBS) system that responds to the patient's current brain state [12].
Experimental Protocols & Data
The following table summarizes the performance of various decoding algorithms reported in the literature, highlighting the challenge of cross-subject generalization.
| Model/Algorithm Type | Key Characteristic | Reported Performance Limitation / Advantage |
|---|---|---|
| Filter Bank CCA (FBCCA) [10] | Unsupervised | Performance declines significantly in short time windows. |
| Task-Related Component Analysis (TRCA) [10] | Supervised | Exhibits weak performance under cross-subject conditions. |
| Traditional CNN/LSTM [11] | Deep Learning | Struggles with spatial connections (CNNs) or long-range temporal dependencies (LSTMs). |
| SSVEPTransformer [10] | Transformer-based | Demonstrated better performance in short time windows and cross-subject conditions compared to traditional models. |
| Adaptive Transformer [11] | Transformer with Adaptive Attention | Achieved 98.24% accuracy on EEG tasks, effectively modeling temporal-spatial relationships. |
Detailed Protocol: Domain Adaptation for Cross-Subject EEG Decoding
Aim: To improve the generalization performance of an EEG-based classification model when applied to a new, unseen subject.
Methodology:
- Data Preparation:
- Source Domain (
Ds): Use a publicly available EEG dataset (e.g., TUH EEG Corpus, CHB-MIT) with data from multiple subjects{xi, yi}i=1Ns[11]. - Target Domain (
Dt): Select one subject as the hypothetical new user. Hold out this subject's data{xj, yj}j=1Ntduring initial training [8]. - Preprocessing: Apply standard preprocessing: band-pass filtering, downsampling, and artifact removal (e.g., for eye blinks and muscle noise) [8].
- Source Domain (
- Feature Extraction:
- Extract features from multiple domains: time, frequency, and time-frequency domains (e.g., using wavelet transforms) [8].
- Alternatively, for an end-to-end deep learning approach, use the raw or minimally processed data and allow the model to learn its own features.
- Model Training with DA:
- Baseline: Train a standard classifier (e.g., SVM, Random Forest) on the source domain data only. Test its performance on the held-out target subject. This establishes a performance baseline.
- Intervention - Feature-Based DA: Implement a feature-based DA algorithm, such as aligning the source and target feature distributions in a shared latent space using a method like Least Squares Transformation [10].
- Train a new classifier on the transformed source features and evaluate it on the target subject's data.
- Evaluation:
Experimental Workflow Visualization
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Research |
|---|---|
| Public EEG Datasets (e.g., TUH EEG Corpus, CHB-MIT) [11] | Provide standardized, annotated neural data for training and benchmarking decoding algorithms, ensuring reproducibility and comparison across studies. |
| Domain Adaptation Algorithms (e.g., Least Squares Transformation, RPA) [8] [10] | Techniques designed to minimize distributional differences between data from different subjects or sessions, directly addressing the generalization problem. |
| Transformer Architectures [10] [11] | Advanced neural network models that use self-attention mechanisms to effectively capture long-range temporal and spatial dependencies in non-stationary neural signals. |
| Open-Source ML Frameworks (e.g., TensorFlow, PyTorch) [13] | Provide the foundational tools and libraries for building, training, and testing custom deep learning models for neural decoding. |
| Hyperparameter Optimization Tools (e.g., Bayesian Optimization) [12] | Automate the search for the best model parameters, which is crucial for achieving robust performance and saving researcher time. |
Adaptive Algorithm Decision Pathway
Clinical and Research Consequences of Ignoring Temporal Variability
Foundations: Understanding Temporal Variability in Neural Signals
What is temporal variability and why is it a problem in neural decoding?
Temporal variability refers to the inconsistency in the timing of neural responses across multiple trials of the same task. In experimental settings, this means that the brain's response to an identical stimulus or the execution of an identical cognitive process (like memory recall or motor imagery) does not occur at precisely the same millisecond every time.
This is a critical problem because many standard decoding algorithms rely on time-locked analysis. These methods assume that task-relevant neural signals are consistently aligned to an external event marker. They perform decoding by analyzing data point-by-point across trials, an approach that fails when the neural dynamics shift in time. When timing is inconsistent, the averaged or analyzed signal appears blurred and degraded, much like a photograph of a moving subject taken with a slow shutter speed. This dramatically reduces the signal-to-noise ratio and compromises the accuracy of decoding mental contents [14].
How does ignoring temporal variability impact my research outcomes?
Ignoring temporal variability can lead to systematic errors and false conclusions in your research. The primary consequences are:
- Reduced Decoding Accuracy: The core performance metric for most decoding studies will be significantly lower. Models trained on misaligned data fail to learn the true neural signature of the cognitive process, leading to poor generalization on new test data.
- Invalidated Conclusions: Findings may incorrectly suggest that a certain brain area does not encode specific information, when in reality, the variability of the response masked the signal.
- Inefficient Use of Data: The statistical power of your experiments is weakened. You may need to collect significantly more data to achieve a desired effect size, increasing time and resource costs.
- Limited Clinical Translation: Algorithms developed under the assumption of fixed timing will perform poorly in real-world clinical applications, such as Brain-Computer Interfaces (BCIs), where users' neural responses are inherently non-stationary [15].
In which specific experimental paradigms is temporal variability most critical?
Temporal variability is particularly detrimental in paradigms involving covert, self-paced cognitive processes. The table below summarizes high-risk paradigms.
Table: Experimental Paradigms Highly Susceptible to Temporal Variability
| Paradigm | Reason for High Variability | Primary Consequence |
|---|---|---|
| Memory Recall | Self-paced retrieval of information; latency varies with memory strength and search effort. | Inaccurate decoding of recalled content [14]. |
| Mental Imagery | No external pacing for the onset and dynamics of the imagined scene or action. | Poor performance in imagery-based BCIs [15]. |
| Decision Making | The cognitive process of deliberation has variable duration. | Misalignment of neural correlates of evidence accumulation and choice. |
| Free-Keying Motor Tasks | Movement initiation is self-paced, unlike cue-triggered movements. | Blurred motor cortical signals and reduced classification of movement type. |
Troubleshooting Guide: Identifying Temporal Variability in Your Data
How can I diagnose if temporal variability is affecting my dataset?
Before implementing complex solutions, confirm that temporal variability is the root of your problem. Follow this diagnostic workflow:
Diagnostic Steps:
- Visual Inspection: Plot single-trial neural responses (e.g., raw signals, band power). Look for jitter in the latency of characteristic deflections or power changes relative to the event marker.
- Cross-Trial Consistency: Compute the inter-trial phase coherence (ITPC) or similar metrics. Low coherence at the time of the expected response suggests high temporal jitter.
- Time-Locked Decoding: Train a decoding model (e.g., an LDA or SVM) at each time point independently and plot the resultant accuracy over time.
- Interpret Result:
- If decoding accuracy forms a sharp, narrow peak that falls off quickly, your signal is highly time-locked but variable. This is a classic signature of temporal variability.
- If accuracy is consistently low and flat across time, the issue may be a fundamentally low signal-to-noise ratio or an incorrect neural feature, not just misalignment [14].
My decoding accuracy is low. Is it poor signal quality or temporal variability?
Distinguishing between these causes is essential for effective troubleshooting. The table below contrasts key indicators.
Table: Differentiating Low SNR from Temporal Variability
| Indicator | Suggests Temporal Variability | Suggests Poor Signal Quality (Low SNR) |
|---|---|---|
| Single-Trial Plots | Clear, strong responses that are misaligned (jittered) across trials. | Noisy, weak, or non-existent responses in most trials. |
| Time-Locked Decoding | Accuracy shows a prominent but narrow peak in time. | Accuracy is low and flat across the entire time window. |
| Grand Average Signal | The event-related potential/field (ERP/ERF) appears small and smeared. | The ERP/ERF is small but not necessarily smeared; noise dominates. |
| Solution | Implement alignment or adaptive algorithms like ADA. | Improve preprocessing, artifact removal, or feature extraction. |
Experimental Protocols & Solutions
What is the Adaptive Decoding Algorithm (ADA) and how do I implement it?
The Adaptive Decoding Algorithm (ADA) is a non-parametric method designed to handle temporal variability directly. Instead of assuming fixed timing, ADA performs a two-level prediction that explicitly accounts for trial-specific latency [14].
Core Protocol: Implementing ADA
Step-by-Step Methodology:
- Input Data: Your training set consists of multi-trial neural data (e.g., MEG/EEG timeseries) and corresponding task labels (e.g., Class A vs. Class B).
- Window Estimation (Training): For each training trial, ADA identifies the temporal window that is most informative for the task. This can be done using an internal cross-validation loop or a measure of discriminability between classes.
- Decoder Construction: A model (e.g., a classifier) is trained using the features extracted from the selected informative windows of all training trials. This model learns the neural patterns associated with the task, independent of their exact timing.
- Testing Phase: For a new, unlabeled test trial:
- A sliding window analysis is performed.
- The algorithm identifies the window within this trial that most likely contains the task-relevant signal.
- The pre-trained decoder is applied specifically to this window to generate the final prediction (e.g., "Class A") [14].
Are there advanced signal processing techniques to manage variable, high-frequency signals?
For signals with complex, high-frequency bursts, such as neuronal spikes, traditional time-frequency decomposition methods may be insufficient. The Hyperlet Transform (HLT) is a super-resolution technique designed for this challenge.
Key Advantages of HLT:
- High Resolution: Provides highly localized representations of short signal bursts.
- Computational Efficiency: Dramatically speeds up super-resolution operations compared to other methods.
- Improved Pattern Recognition: By unmixing complex signals, HLT enhances downstream tasks like neuronal spike detection and sorting, leading to cleaner neural features for decoding [16].
How can deep learning architectures be designed to handle temporal variability?
Deep learning models can inherently learn to be invariant to certain transformations, including temporal shifts. A highly effective architecture is the Hierarchical Attention-Enhanced Convolutional-Recurrent Network.
Experimental Protocol for Motor Imagery Classification (as demonstrated in [15]):
- Spectral Feature Extraction: The architecture first uses Convolutional Neural Network (CNN) layers to extract spatial features from the input EEG signals, treating electrode locations as spatial dimensions.
- Temporal Dynamics Modeling: The spatial features are then fed into Long Short-Term Memory (LSTM) layers. LSTMs are specialized for sequence modeling and can capture the temporal evolution of the neural signal, learning which temporal patterns are diagnostic.
- Attention Mechanism: An attention layer is applied to the LSTM outputs. This layer learns to adaptively weight different time points based on their importance for the classification task. This is the key to handling variability—the model learns to "focus" on the relevant neural activity regardless of its precise timing.
- Classification: The weighted features are finally passed to a fully connected layer for classification (e.g., left-hand vs. right-hand motor imagery). This approach achieved a state-of-the-art accuracy of 97.25% on a four-class motor imagery task, demonstrating the power of explicitly modeling temporal structure and importance [15].
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Computational Tools for Non-Stationary Neural Signal Research
| Tool / Algorithm | Type | Primary Function | Key Reference |
|---|---|---|---|
| Adaptive Decoding Algorithm (ADA) | Decoding Algorithm | Handles temporal jitter via trial-specific window selection. | [14] |
| Hyperlet Transform (HLT) | Signal Processing Tool | Provides super-resolution time-frequency decomposition for short bursts. | [16] |
| Hierarchical Attention Model | Deep Learning Architecture | Uses CNNs, LSTMs, and attention to weight informative time points. | [15] |
| Common Spatial Patterns (CSP) | Feature Extraction | Extracts spatially discriminative patterns; requires alignment or adaptation. | [15] |
| Filter Bank CSP (FBCSP) | Feature Extraction | Extends CSP to multiple frequency bands, improving feature robustness. | [15] |
Frequently Asked Questions (FAQs)
Can't I just use more data to average out temporal variability?
While increasing trial count can improve the signal-to-noise ratio of a grand average, it does not solve the core problem of blurring. Averaging misaligned trials will still result in a temporally smeared and potentially attenuated representation of the true neural response. This limits the resolution at which you can study neural dynamics and is ineffective for single-trial decoding, which is essential for BCIs and real-time applications.
Is temporal variability only a concern for EEG/MEG, or also for fMRI?
Temporal variability is a concern for all neuroimaging modalities, but its impact is scaled by the temporal resolution of the technology. It is most critical for high-temporal-resolution techniques like EEG and MEG, where shifts of tens of milliseconds are meaningful. In fMRI, with its resolution of seconds, neural events happening hundreds of milliseconds apart are collapsed into a single volume. However, variability in the Hemodynamic Response Function (HRF) across brain regions and individuals is a well-studied problem that also requires careful modeling.
Are there clinical trial implications for ignoring neural temporal variability?
Yes. Ignoring temporal variability in clinical neuroscience research can lead to failed trials. For example:
- Inaccurate Endpoints: If a drug is intended to improve cognitive function (e.g., memory recall speed), but the neural biomarker for recall is misaligned and blurred, you may fail to detect a true treatment effect.
- BCI Rehabilitation: Clinical trials for BCIs used in stroke rehabilitation will see poor patient outcomes and high dropout rates if the decoding algorithm is not robust to the natural trial-to-trial variability in the patient's neural signals. Improving trial efficiency and participant retention through better technology is a major focus in the clinical trial industry [17] [18].
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: When is it necessary to record EEG and fMRI simultaneously, rather than in separate sessions?
Simultaneous recording is necessary when your research question requires that both datasets capture identical brain activity from the very same trial. This is crucial for analysis methods that rely on a direct trial-by-trial relationship between the electrophysiological (EEG) and hemodynamic (fMRI) signals, such as EEG-informed fMRI analysis [19]. If your hypothesis involves investigating the direct coupling between these signals in a resting state or during decision-making tasks, simultaneous recording is essential [19]. However, if your study design can tolerate the variance introduced by separate sessions (e.g., different sensory stimulation, habituation effects), and your analysis does not depend on a perfect one-to-one trial correspondence, then separate sessions may be preferable, as they often provide higher signal quality for each modality [19].
Q2: What are the most effective methods for handling physiological artifacts in EEG data?
The optimal method depends on the artifact type [20]:
- Eye blinks and movements: Use Independent Component Analysis (ICA) or regression-based subtraction. These artifacts are most prominent in frontal channels [20].
- Muscle artifacts (e.g., from jaw clenching): For transient artifacts, rejection of contaminated epochs is common. For persistent, localized muscle noise, ICA can be effective. Filtering can also attenuate the impact, though it may not remove it completely [20].
- Pulse (heartbeat) artifacts: If an electro-cardiogram (ECG) is co-registered, use specialized algorithms to identify and remove the heartbeat. Without ECG, ICA or using an average reference can reduce its influence [20].
- Sweating/Skin potentials: High-pass filtering can reduce slow drifts. The best strategy is prevention by ensuring a fresh and dry recording environment [20].
Q3: My decoding algorithm performs poorly across different subjects or sessions. What strategies can improve generalization?
This is a classic challenge due to the non-stationarity of neural signals. Domain Adaptation (DA) techniques are designed to address this by minimizing distributional differences [8]. You can consider:
- Feature-based DA: Transforming features from different subjects/sessions into a common space where their probability distributions are similar [8].
- Model-based DA: Fine-tuning a decoder pre-trained on a source subject's (or group's) labeled data using a small amount of data from your target subject. This is highly efficient when data collection is difficult [8].
- Instance-based DA: Re-weighting or selecting samples from your source dataset that are most similar to the target data, thereby reducing the influence of less relevant samples [8].
Q4: How can we validate the concordance between EEG source localization and fMRI findings?
A robust method involves a two-pronged approach on the same cortical surface [21]:
- Independent Comparison (MEM-concordance): Compare EEG sources localized with a method like Maximum Entropy on the Mean (MEM) with significant fMRI clusters. Quantify concordance using metrics like minimal geodesic distances between local extrema and overlap measurements of their spatial extents [21].
- fMRI-Relevance Index (α): Statistically test whether the fMRI cluster can serve as a relevant prior for the EEG inverse solution. A significantly positive α index suggests that sources located within that fMRI cluster can explain the scalp EEG data, providing evidence for concordance [21].
Troubleshooting Common Experimental Issues
Table 1: Troubleshooting Common Data Quality Issues
| Problem | Possible Causes | Solutions & Checks |
|---|---|---|
| Poor EEG signal quality during simultaneous EEG-fMRI | Gradient and ballistocardiogram (BCG) artifacts [19]; Loose electrode contact [20]. | Use artifact removal algorithms designed for MRI environments [19]; Ensure cap fit and check impedances (target 5-10 kΩ) [22]. |
| Low signal-to-noise ratio in fMRI-informed EEG source imaging | Overly restrictive fMRI priors; Mismatch between hemodynamic and electrical sources. | Use multiple Temporally Coherent Networks (TCNs) from fMRI as flexible covariance priors in a Parametric Empirical Bayesian (PEB) framework (e.g., the NESOI approach) [23]. |
| Decoding performance drops with unknown timing of neural events | Temporal variability in cognitive processes (e.g., memory recall); Assumption of fixed latency in analysis. | Employ algorithms that account for trial-specific timing, like the Adaptive Decoding Algorithm (ADA), which identifies the most informative temporal window for each trial [14]. |
| Discrepancies observed between EEG and fMRI activation maps | Different physiological origins and sensitivities of the signals; fMRI may reflect metabolic load while EEG reflects synchronized pyramidal activity [19]. | This may be a valid finding. Ensure your task reliably produces signals in both modalities. Consider that the underlying neural generator may be a distributed network, parts of which are visible to only one modality [21] [19]. |
Experimental Protocols for Multimodal Integration
Protocol 1: Integrating fMRI-Derived Networks for EEG Source Imaging (NESOI)
This protocol uses fMRI to provide spatial priors for estimating EEG source dynamics [23].
- fMRI Data Processing: Use Independent Component Analysis (ICA) on the fMRI data to extract multiple Temporally Coherent Networks (TCNs). These can include both task-related and resting-state networks [23].
- Lead Field Calculation: Construct a lead field matrix based on the individual's head model and electrode positions [23].
- EEG Source Reconstruction with PEB: Set up a Parametric Empirical Bayesian (PEB) model for EEG source imaging. Use the TCNs from Step 1 as the covariance components (priors) in the source space of the model [23].
- Model Estimation: Let the PEB framework estimate the hyperparameters that control the contribution of each TCN prior, based on the scalp EEG data. This provides a solution that integrates the high temporal resolution of EEG with the high spatial resolution of fMRI TCNs [23].
Workflow for fMRI-Informed EEG Source Imaging
Protocol 2: Assessing EEG-fMRI Concordance in Epileptic Spike Analysis
This protocol provides a quantitative method to compare generators of interictal spikes identified by EEG and fMRI [21].
- Data Acquisition & Preprocessing: Acquire EEG and fMRI data simultaneously. Preprocess both datasets and coregister them to the same anatomical space (e.g., the cortical surface) [21].
- fMRI Analysis: Perform an event-related fMRI analysis using the onset of spikes (marked on the EEG) as events. Identify clusters of significant BOLD response (activation/deactivation) and interpolate them onto the cortical surface [21].
- EEG Source Localization: Estimate distributed EEG sources of the averaged spikes on the cortical surface using an method like Maximum Entropy on the Mean (MEM), independent of the fMRI results [21].
- Quantify MEM-Concordance: For each fMRI cluster, calculate:
- The minimal geodesic distance between its local extremum and that of the EEG source.
- The spatial overlap between the extent of the fMRI cluster and the EEG source [21].
- Calculate fMRI-Relevance Index (α): For each fMRI cluster, estimate the α index to test if constraining the EEG source to that cluster significantly explains the scalp EEG data. A positive α supports concordance [21].
The Scientist's Toolkit
Table 2: Key Research Reagents & Computational Tools
| Item / Resource | Function / Application | Key Details |
|---|---|---|
| Parametric Empirical Bayesian (PEB) Framework | A flexible framework for EEG source imaging that allows incorporation of various priors, including those from fMRI [23]. | Enables the use of fMRI-derived Temporally Coherent Networks (TCNs) as covariance priors to guide the EEG inverse solution [23]. |
| Independent Component Analysis (ICA) | A data-driven method for separating mixed signals into statistically independent components [23] [20]. | Used to extract TCNs from fMRI data [23] and to isolate and remove artifacts (e.g., eye blinks, muscle noise) from EEG data [20]. |
| Domain Adaptation (DA) Algorithms | Enhances the generalization of neural decoders across subjects or sessions by minimizing distributional differences in the data [8]. | Categorized into instance-based, feature-based, and model-based approaches, with growing use in combination with deep learning [8]. |
| Adaptive Decoding Algorithm (ADA) | Decodes neural signals when the timing of cognitive events is variable and unknown across trials [14]. | A nonparametric method that, for each trial, estimates the temporal window most likely to contain task-relevant signals before decoding [14]. |
| Multimodal Dataset (e.g., from [24]) | Provides a benchmark for developing and testing new analytical methods, particularly for understanding the relationship between different neural signals. | Includes single neurons, local field potentials (LFP), intracranial EEG (iEEG), and fMRI from the same participants during a continuous naturalistic task (movie watching) [24]. |
The Role of Adaptive Decoding in Brain-Computer Interfaces (BCIs) and Neuroprosthetics
Frequently Asked Questions (FAQs)
1. What is adaptive decoding and why is it necessary in BCIs? Adaptive decoding refers to algorithms that can update their parameters over time to compensate for changes in neural signals, known as non-stationarity. These changes can be caused by factors like neuronal plasticity, learning, electrode instability, or tissue response around the implant. Without adaptation, a decoder's performance will degrade, making long-term, reliable BCI operation impossible [25].
2. What are the main technical approaches to adaptive decoding? Research has explored several methodological approaches, which can be broadly categorized [8]:
- Model-based Adaptation: Tuning the parameters of a pre-defined model (e.g., a Kalman Filter) using recent data.
- Feature-based Adaptation: Transforming neural features to align distributions from different sessions or subjects.
- Instance-based Adaptation: Intelligently weighting or selecting training samples to improve model generalization.
3. My BCI performance drops significantly across days. How can adaptive decoding help? Cross-session and cross-subject performance drops are a primary challenge that adaptive decoding aims to solve. Techniques like domain adaptation (DA) can rapidly transfer knowledge from previous, large datasets (source domain) to new sessions or subjects (target domain) with minimal new data. For instance, you can pre-train a model on source data and then fine-tune it with a small amount of target subject data, significantly reducing calibration time and maintaining accuracy [8].
4. Are there adaptive methods that don't require knowing the user's intended movement? Yes. Self-training methods like Bayesian regression updates can use the decoder's own output as a substitute for the true intended movement to periodically update the neuronal tuning model. This allows the decoder to adapt without external training signals or assumptions about the user's goals [25].
5. What is the role of deep learning in modern adaptive decoders? Deep learning models, such as Recurrent Neural Networks (RNNs) and Transformers, can automatically learn complex spatiotemporal features from neural data. Their architectures are naturally suited for sequence decoding (e.g., of sentences) and can be combined with domain adaptation techniques to create powerful, end-to-end adaptive decoders that generalize well across sessions [8] [26].
Troubleshooting Guide: Common Experimental Issues
| Problem Area | Specific Issue | Potential Causes | Recommended Solutions |
|---|---|---|---|
| Decoder Performance | High error rate on new days or with new subjects. | Distribution shift (non-stationarity) in neural data; poor generalizability of static decoder [8]. | Implement a Domain Adaptation (DA) strategy. Fine-tune a pre-trained model with a small amount of new subject/session data [8]. |
| Gradual performance decay within a single long session. | Within-session neuronal tuning changes or recording instability [25]. | Use a recursive self-training algorithm (e.g., Bayesian regression update) that updates decoder parameters every few minutes using recent decoder outputs [25]. | |
| Signal Acquisition & Quality | Poor decoding accuracy despite a previously good model. | Electrode impedance changes; poor contact; neuronal recording instability; environmental noise [25]. | Verify signal quality and electrode connections. For invasive systems, check spike waveform stability. For non-invasive systems, ensure proper impedance (<2000 kOhms is a common target) [27]. |
| Real-Time Operation | Unstable or jittery control of a neuroprosthetic. | High latency or inaccurate decoding at each time step. | Employ a state-space model like an Unscented Kalman Filter (UKF). It uses a tuning model and a kinematics model to smooth predictions and is compatible with adaptive updates [25]. |
Experimental Protocols & Performance Data
Protocol 1: Bayesian Regression Self-Training for Motor Decoding
This methodology is designed for continuous adaptation in closed-loop motor BCI experiments [25].
- Initial Decoder Setup: Begin with an Unscented Kalman Filter (UKF) decoder. Its parameters are initially fit using calibration data (e.g., neural activity recorded during attempted or actual arm movements).
- Closed-Loop Operation: The user controls a cursor or prosthetic device using the BCI.
- Batch-Mode Update:
- Every 2 minutes, collect a batch of recent neural firing rates and the corresponding decoder outputs (e.g., predicted kinematic states).
- Use Bayesian linear regression to compute new tuning parameters. The previous parameters serve as the prior, and the new data provides the likelihood to form the posterior.
- A transition model is applied to the parameters to account for potential drift.
- This process can be formulated to allow for the temporary omission or addition of neurons from the population.
Quantitative Performance: In offline reconstructions with non-human primates, this self-training update significantly improved the accuracy of hand trajectory reconstructions compared to a static decoder. In real-time closed-loop experiments spanning 29 days, the adaptive updates were crucial for maintaining control accuracy without requiring knowledge of the user's intended movements [25].
Protocol 2: Domain-Adaptive Speech Decoding for a Speech Neuroprosthesis
This protocol outlines the process for decoding attempted speech from a person with paralysis [26].
- Training Data Collection: The user attempts to speak 260-480 sentences, cued by a computer monitor. Intracortical neural activity is recorded from relevant speech motor areas (e.g., ventral premotor cortex).
- Model Architecture & Training: A Recurrent Neural Network (RNN) is trained to predict sequences of phonemes from neural data. To handle non-stationarity:
- Use unique input layers for each day to account for across-day changes.
- Implement rolling feature adaptation to account for within-day changes.
- Language Model Integration: The RNN's output (phoneme probabilities) is combined with a statistical language model to infer the most probable word sequence.
- Real-Time Evaluation: The user attempts to speak new, held-out sentences. Decoded words appear on the screen in real-time.
Quantitative Performance: This adaptive approach enabled a speech BCI to achieve a 23.8% word error rate on a 125,000-word vocabulary at a speed of 62 words per minute, demonstrating the feasibility of large-vocabulary decoding [26].
Table 1: Performance Comparison of Adaptive Decoding Algorithms
| Adaptive Method / Study | Application | Key Metric | Reported Performance |
|---|---|---|---|
| Bayesian Self-Training [25] | Motor Control (Non-human primate) | Control Accuracy Maintenance | Maintained accuracy over 29 days in closed-loop experiments. |
| RNN with Daily Adaptation [26] | Speech Decoding (Human Clinical Trial) | Word Error Rate (125k vocabulary) | 23.8% |
| Decoding Speed | 62 words per minute | ||
| Domain Adaptation (DA) Survey [8] | Cross-Subject/Session EEG | Generalization Accuracy | Enabled effective knowledge transfer, reducing need for extensive per-subject calibration. |
The Scientist's Toolkit: Research Reagents & Materials
Table 2: Essential Components for Adaptive BCI Research
| Item | Function in Research | Example / Note |
|---|---|---|
| Intracortical Microelectrode Arrays | Records action potentials (spikes) from populations of neurons. High-density arrays are crucial for decoding complex intentions. | e.g., 96-micro-wire arrays implanted in motor and somatosensory cortex [25] [26]. |
| Unscented Kalman Filter (UKF) | A state-space decoder for predicting continuous kinematic variables (e.g., cursor position) from neural activity. Serves as a base decoder for some adaptive methods [25]. | Preferred over standard Kalman filters for better handling of non-linear dynamics [25]. |
| Recurrent Neural Network (RNN) | A deep learning model ideal for decoding temporal sequences, such as phonemes in speech or movement trajectories. | Can be combined with custom input layers and rolling adaptation to combat non-stationarity [26]. |
| Bayesian Linear Regression | The core statistical engine for probabilistic parameter updates in self-training paradigms. | Combines prior knowledge (old parameters) with new evidence (recent data) in a principled way [25]. |
| Domain Adaptation (DA) Framework | A set of computational techniques to minimize distribution differences between training (source) and deployment (target) data domains. | Categorized into instance-based, feature-based, and model-based approaches [8]. |
Methodological Workflows
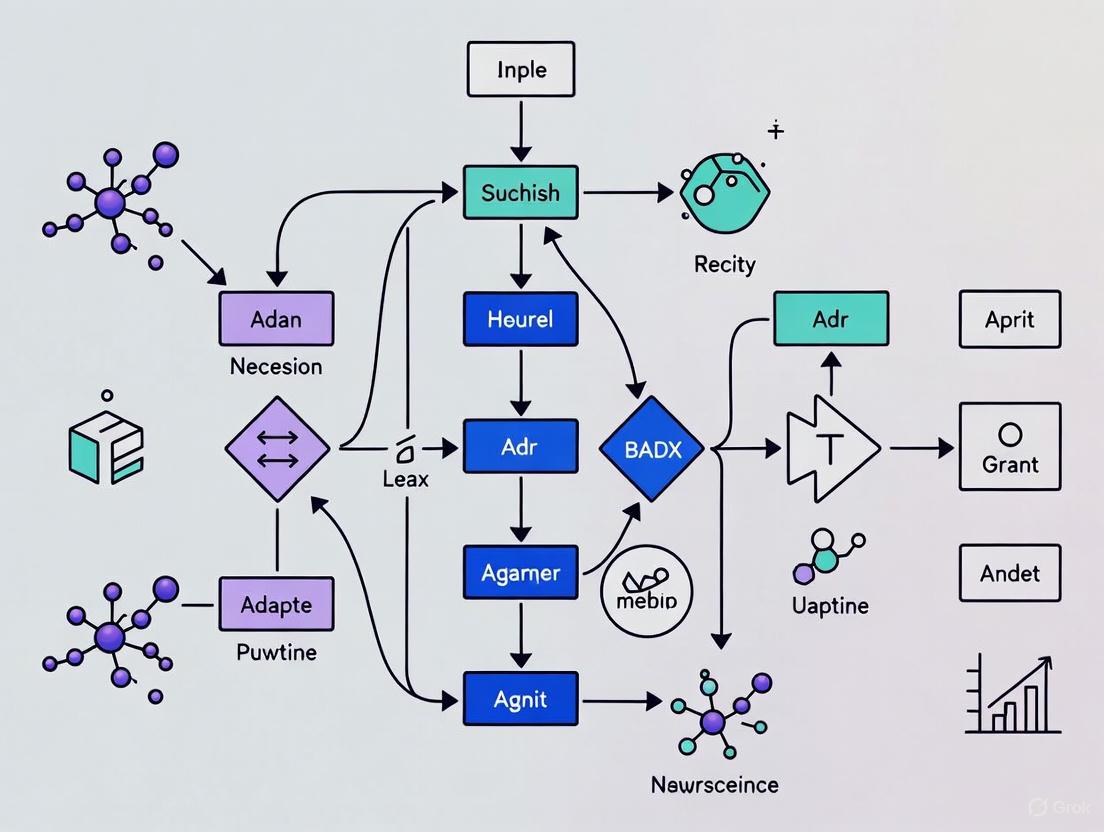
The following diagram illustrates the logical workflow of a self-training adaptive decoder, a key method for handling non-stationary signals.
This diagram outlines a high-level workflow for implementing a domain-adaptive decoding strategy, particularly useful for cross-session or cross-subject applications.
Core Algorithms and Architectures: Methodological Innovations and Applications
Bayesian Adaptive Methods for Dynamic Probability Updating
Frequently Asked Questions (FAQs)
Q1: What are the core advantages of using Bayesian methods over traditional frequentist approaches for decoding non-stationary neural signals? Bayesian methods provide a dynamic framework that integrates prior knowledge and continuously updates probabilistic beliefs with incoming data. This is crucial for non-stationary neural signals, as it allows the model to adapt to changes in signal properties over time or across sessions [28] [29] [30]. Unlike frequentist methods that often provide a single-point estimate, Bayesian approaches output probability distributions, offering a measure of uncertainty for each prediction. This is vital for assessing the reliability of decoded neural commands in brain-computer interfaces (BCIs) and for making informed decisions, especially in safety-critical applications like clinical neurotechnology [29] [30].
Q2: How can I quantify and improve the uncertainty estimates from my Bayesian neural decoder? Uncertainty in Bayesian models arises from two main sources: aleatoric (data noise) and epistemic (model uncertainty). You can improve these estimates by:
- Using Bayesian Neural Networks (BNNs): BNNs place probability distributions over weights, naturally capturing model uncertainty. Techniques like Monte Carlo Dropout or Markov Chain Monte Carlo (MCMC) sampling during inference can approximate these posteriors and provide uncertainty estimates [30].
- Proper Prior Selection: The choice of prior distribution significantly impacts uncertainty calibration. Use informative priors based on historical data or expert knowledge to constrain the model, which is particularly useful when data is limited [28] [31].
- Validation: Calibrate your model's uncertainty by checking if the predicted confidence intervals match the empirical frequency of outcomes on held-out validation data.
Q3: My decoding performance drops significantly between experimental sessions. What adaptive strategies can I use? This is a classic problem of cross-session domain shift. Several domain adaptation (DA) strategies can help:
- Feature-Based DA: Transform the features from both old (source) and new (target) sessions into a shared space where their distributions are aligned. This minimizes distributional differences caused by changes in electrode impedance or neural signal properties [8].
- Model-Based DA: Fine-tune a decoder trained on source session data using a small amount of new target session data. This allows the model to adapt its parameters quickly to the new signal characteristics without requiring a full re-training from scratch [8].
- Instance-Based DA: Re-weight the importance of data points from the source session, prioritizing those that are most similar to the new target session data during model training [8].
Q4: What are the best practices for preprocessing neural data to enhance Bayesian decoding under low signal-to-noise ratio (SNR) conditions? Effective artifact removal is essential for improving SNR before decoding.
- Adaptive Spatial Filtering: For intracortical signals, use advanced methods like the weighted Common Average Reference (CAR) filter with a Kalman filter to adaptively estimate and remove common noise across channels. This has been shown to improve force decoding accuracy from local field potentials (LFPs) by 33% compared to standard CAR filters [32].
- Artifact Correction vs. Rejection: For EEG, studies show that correcting artifacts using Independent Component Analysis (ICA) is often sufficient and preferable to outright rejecting contaminated trials. While the combination of correction and rejection does not significantly boost decoding accuracy in most cases, correction is still recommended to minimize artifact-related confounds that could artificially inflate performance metrics [33].
Troubleshooting Guides
Issue 1: Poor Model Adaptation to Rapidly Changing Neural States
Symptoms: The decoder's performance lags when the subject's cognitive state, behavior, or neural patterns change quickly. The model seems to be "stuck" on previous statistics.
Diagnosis and Solutions:
| Potential Cause | Diagnostic Checks | Recommended Solution |
|---|---|---|
| Insufficiently Informative Prior | Check if the prior distribution is too diffuse ("uninformative"), causing the model to learn slowly from new data. | Use a more informative prior based on data from the initial calibration or previous sessions. Implement a "forgetting" mechanism by using a fading-memory likelihood that places more weight on recent observations [28]. |
| Fixed, Inadequate Model Structure | The model lacks the capacity to capture the new neural dynamics. | Employ a Bayesian Adaptive Regression framework. Define a model that can dynamically switch between different regimes or states. The hidden state (e.g., the intended movement direction) can be inferred using recursive Bayesian filters like the Kalman filter or particle filters, which are designed for tracking dynamic states [14] [32]. |
Experimental Workflow for Dynamic Tracking: The following diagram outlines a recursive Bayesian filtering approach for tracking a continuously updating neural state.
Issue 2: Decoder Performance Degradation Over Long Time Scales (e.g., Days/Weeks)
Symptoms: The decoder trained on day 1 performs poorly when applied on day 7, even with initial recalibration. This is often due to non-stationarities in the neural code.
Diagnosis and Solutions:
| Potential Cause | Diagnostic Checks | Recommended Solution |
|---|---|---|
| Covariate Shift | Compare the feature distributions (e.g., mean power in specific frequency bands) between the initial training session and the new degraded session. | Apply Domain Adaptation (DA) techniques. As highlighted in the FAQs, use feature-based DA to find a domain-invariant feature space. This allows a decoder trained on source domain data (Day 1) to generalize to a target domain (Day 7) without extensive re-labeling [8]. |
| Inadequate Handling of Neural Sparsity and Variability | The sampling or model is not adapting to the changing sparsity and information content of the neural signal. | Implement an adaptive sampling rate allocation strategy. Inspired by methods in compressed sensing, you can segment the neural feature space and allocate more "decoding resources" (e.g., model complexity) to blocks of data with higher information content (sparsity), thereby improving the efficiency and robustness of the overall decoding framework [34]. |
Protocol for Feature-Based Domain Adaptation:
- Data Preparation: Collect labeled neural data from your initial session (source domain,
D_s) and a small amount of (potentially unlabeled) data from the new session (target domain,D_t). - Feature Extraction: Extract features (e.g., power spectral density, spike rates) from both
D_sandD_t. - Domain Alignment: Train a feature transformation network or apply an algorithm (e.g., Correlation Alignment CORAL, Maximum Mean Discrepancy MMD minimization) to project features from both domains into a new, domain-invariant feature space. The goal is to make
P(features_s) ≈ P(features_t)[8]. - Decoder Training & Application: Train your Bayesian decoder on the transformed source domain features and their labels. This decoder should then be applied directly to the transformed target domain features for inference.
Issue 3: High Computational Cost of Bayesian Inference in Real-Time Applications
Symptoms: The decoding algorithm cannot run in real-time due to the computational burden of sampling from posterior distributions.
Diagnosis and Solutions:
| Potential Cause | Diagnostic Checks | Recommended Solution |
|---|---|---|
| Intractable Posterior | Using exact inference for complex models, leading to slow performance. | Replace exact inference with approximate methods. Use Variational Inference (VI) to approximate the true posterior with a simpler, tractable distribution. This is often faster than MCMC sampling and more suitable for real-time BCIs [30]. |
| Overly Complex Model | The model has too many parameters or layers for the available hardware. | Use a Bayesian Neural Network (BNN) with simplified architecture. Alternatively, employ techniques like adaptive layer parallelism. While originally proposed for LLMs, the core idea is relevant: for simpler decoding decisions, use intermediate network layers to generate predictions, bypassing the full computational graph and speeding up inference without sacrificing output consistency [35]. |
Protocol: Evaluating Bayesian Adaptive Filtering for Kinematic Decoding
This protocol details how to implement and test a Bayesian adaptive filter for decoding continuous movement parameters (e.g., hand velocity) from neural signals.
1. Hypothesis: A Bayesian adaptive filter (e.g., Kalman filter) will provide more accurate and robust decoding of hand kinematics from motor cortical signals compared to a standard Wiener filter, especially in the presence of non-stationary neural tuning.
2. Materials and Reagents:
- Neural Data: Intracortical recordings from a multi-electrode array (e.g., in primary motor cortex). Include both Single-Unit Activity (SUA) and Local Field Potentials (LFP) [32].
- Behavioral Data: Simultaneously recorded hand kinematics (position, velocity, force) at a high temporal resolution.
- Preprocessing Tools: Software for spike sorting, LFP filtering, and artifact removal (e.g., using the adaptive weighted CAR method) [32].
3. Detailed Methodology:
Preprocessing:
- Apply the weighted Common Average Reference (CAR) method with a Kalman filter to remove common noise and artifacts from the intracortical channels [32].
- Extract neural features: For SUA, use binned spike counts. For LFP, extract signal power in specific frequency bands (e.g., beta: 13-30 Hz, gamma: 70-200 Hz) over sliding time windows.
- Smooth and standardize all neural features and kinematic data.
Model Definition (Kalman Filter):
- State Transition Model:
x_t = A * x_{t-1} + w_t, wherex_tis the kinematic state (e.g., 2D velocity) andw_tis process noise. - Observation Model:
y_t = C * x_t + q_t, wherey_tis the vector of neural features andq_tis observation noise. - The matrices
A(state transition) andC(observation) are learned from a training dataset via maximum likelihood estimation.
- State Transition Model:
Bayesian Recursive Inference:
- Prediction Step: Predict the next state:
p(x_t | y_1:t-1) = N(x_t | A * μ_{t-1}, A * Σ_{t-1} * A^T + W). - Update Step: Update the belief with new neural data
y_t:p(x_t | y_1:t) = N(x_t | μ_t, Σ_t), where the meanμ_tand covarianceΣ_tare updated using the standard Kalman gain equations. This update is the application of Bayes' rule.
- Prediction Step: Predict the next state:
Validation:
- Use a held-out test dataset not used for model fitting.
- Quantify decoding performance using the Coefficient of Determination (R²) between the decoded and actual kinematics [32].
The table below summarizes key results from selected studies on adaptive methods in neural decoding and related fields.
Table 1: Performance of Adaptive Methods in Signal Decoding and Clinical Trials
| Application Area | Method | Key Performance Metric | Result | Source |
|---|---|---|---|---|
| Force Decoding (Rat Motor Cortex) | Weighted CAR + Kalman Filter (Artifact Removal) | Decoding Accuracy (R² value) | 33% improvement in R² compared to standard CAR filters [32]. | [32] |
| EEG Decoding (Various ERP Paradigms) | ICA Artifact Correction + Artifact Rejection | Impact on SVM/LDA Decoding Performance | No significant performance improvement in vast majority of cases. Recommendation: Use artifact correction to avoid confounds, but rejection may be unnecessary [33]. | [33] |
| Clinical Trial Design | Bayesian Adaptive Design | Efficiency (Sample Size, Duration) | Can reduce number of patients exposed to inferior treatments; enables seamless Phase II/III trials, accelerating development [28] [29]. | [28] [29] |
| LLM Decoding (Computer Science) | AdaDecode (Adaptive Layer Parallelism) | Decoding Throughput (Speedup) | Up to 1.73x speedup while guaranteeing output parity with standard decoding [35]. | [35] |
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Components for Bayesian Adaptive Decoding Research
| Item | Function in Research | Specific Example / Note |
|---|---|---|
| Multi-Electrode Array | Records rich, high-resolution information about kinematic and kinetic states from multiple neurons simultaneously. Essential for obtaining the high-dimensional input for decoders [32]. | Utah array, Neuropixels probe. |
| Bayesian Neural Network (BNN) | A type of neural network that provides uncertainty estimates for its predictions. Crucial for assessing the reliability of decoded commands in safety-critical BCI applications [30]. | Can be implemented using libraries like PyTorch or TensorFlow with probability distributions over weights. |
| Domain Adaptation Algorithm | Enhances decoder generalizability across subjects or sessions by minimizing distributional differences in neural data. Addresses the problem of non-stationarity and inter-subject variability [8]. | Methods include feature-based (e.g., MMD minimization) and model-based (fine-tuning) approaches. |
| Markov Chain Monte Carlo (MCMC) Sampler | A computational method for approximating complex posterior distributions in Bayesian inference. Used when exact inference is intractable [30]. | Software: Stan, PyMC. Can be computationally intensive for real-time use. |
| Variational Inference (VI) Engine | An alternative, often faster, method for approximate Bayesian inference. Optimizes a simpler distribution to closely match the true posterior. More suitable for real-time BCI than MCMC in many cases [30]. | Often implemented automatically in probabilistic programming libraries. |
| Adaptive Spatial Filter | Removes common noise and artifacts from neural signals in a data-driven way, improving the signal-to-noise ratio before decoding [32]. | Weighted CAR filter with Kalman adaptation is an example for intracortical data [32]. |
| Kalman Filter / Particle Filter | The core algorithmic engine for recursive Bayesian state estimation. Used for dynamically tracking continuously varying neural states or kinematic parameters [14] [32]. | Kalman filter is optimal for linear Gaussian models; particle filters handle more complex, non-linear models. |
Transformer-Based Models with Adaptive Attention Mechanisms
Frequently Asked Questions
Q1: Why does my transformer model fail to decode non-stationary neural signals effectively?
Standard transformer architectures assume temporal consistency in input signals, which is often violated in neural data. The non-stationarity of neural signals—caused by factors like neuronal property variance, recording degradation, and attention fluctuations—severely limits traditional self-attention mechanisms that lack explicit frequency-domain modeling capabilities [36] [2] [37]. To address this, implement adaptive frequency-domain attention mechanisms that can dynamically emphasize fault-related frequency components while preserving long-range temporal dependencies [36].
Q2: What causes performance degradation when applying transformers to motor cortex decoding tasks?
Performance degradation typically stems from the fundamental mismatch between the transformer's stationary assumptions and the inherent non-stationarity of neural motor signals. As observed in primate studies, neural firing patterns in motor cortex significantly vary over time—some neurons show increasing mean firing rates while kinematic parameters remain consistent [2]. This temporal variability creates a moving target for fixed-parameter models. Consider implementing adaptive Kalman filters or recurrent neural network decoders that update their parameters as new observations become available [2] [37].
Q3: How can I improve my model's robustness to recording degradation in chronic intracortical recordings?
Chronic recording degradation manifests as decreased mean firing rates and reduced numbers of isolated units over time due to factors like glial scarring [37]. Implement dual-path extraction with gated residual enhancement (GRE-DB modules) that maintain performance under signal degradation conditions [36]. Additionally, employ retraining schemes where decoders are periodically updated with new session data rather than relying solely on initial training, as this approach maintains performance when neural preferred directions change over time [37].
Q4: What computational optimizations are available for attention mechanisms in long neural signal sequences?
For handling long sequential neural data, consider optimized attention implementations like PyTorch's scaleddotproduct_attention (SDPA) with FlashAttention-2 backend, memory-efficient attention, or CuDNN attention [38]. These fused kernel operations significantly reduce memory usage and computational overhead while maintaining accuracy. When deploying in production, leverage training optimizations like sliding window attention that carry over to inference, fundamentally shaping the model's capabilities [39] [38].
Troubleshooting Guides
Performance Degradation Under Noisy Conditions
Symptoms: Model accuracy decreases significantly under low signal-to-noise ratio (SNR) conditions; failure to detect periodic vibration patterns in bearing fault diagnosis applications [36].
| Problem Cause | Diagnostic Steps | Solution |
|---|---|---|
| Inadequate noise suppression | Analyze model performance across multiple SNR levels; examine attention weight distribution | Integrate ultra-wide convolutional kernels at initial stage to suppress high-frequency noise [36] |
| Limited frequency-domain modeling | Compare time-domain vs frequency-domain feature importance | Implement adaptive frequency-domain attention mechanism to highlight informative diagnostic features [36] |
| Insufficient multiscale feature extraction | Visualize activations at different network depths | Add multiscale dilated convolutions to extract hierarchical temporal features [36] |
Protocol: To diagnose noise-related issues, systematically evaluate your model using the Paderborn University and Case Western Reserve University bearing fault datasets at SNR levels from -4dB to 10dB. Compare your model's performance against the ALMFormer architecture, which integrates large-kernel convolution and multiscale CNN structures [36].
Temporal Misalignment in Neural Decoding
Symptoms: Inconsistent decoding performance across trials; variable latency in detecting movement intention from motor cortex signals [2] [14].
| Problem Cause | Diagnostic Steps | Solution |
|---|---|---|
| Fixed temporal window assumption | Measure trial-to-trial timing variability | Implement Adaptive Decoding Algorithm (ADA) with two-level prediction that estimates optimal temporal windows per trial [14] |
| Non-adaptive model parameters | Track model performance degradation over session time | Develop adaptive Kalman filter or linear regression methods that update parameters with new observations [2] |
| Ignoring neural population dynamics | Analyze changes in preferred directions across neurons | Incorporate population vector models that account for neural property variance [37] |
Protocol: For temporal alignment issues, implement the ADA framework which first estimates, for each trial, the temporal window most likely to reflect task-relevant signals, then decodes test trials based on selection of informative windows. Validate using a model of memory recall based on real perception data [14].
Computational Bottlenecks in Long Sequence Processing
Symptoms: Slow training times; memory overflow with long neural recordings; inability to process full experimental sessions [36] [38].
| Problem Cause | Diagnostic Steps | Solution |
|---|---|---|
| Standard self-attention complexity | Profile computation time by sequence length | Replace with optimized attention kernels (FlashAttention, PyTorch SDPA, TransformerEngine) [38] |
| Inefficient attention computation | Monitor GPU memory usage during training | Implement sliding window attention or dilated attention mechanisms [39] |
| Suboptimal implementation | Compare different attention backends | Use PyTorch SDPA which dynamically selects most efficient backend based on input properties [38] |
Protocol: To address computational limitations, benchmark your attention implementation using a Vision Transformer backbone with sequence lengths matching your neural data. Compare default attention against optimized implementations like FlashAttention-2, which can reduce step time from 370ms to 242ms on NVIDIA H100 GPUs [38].
Experimental Protocols
Adaptive Frequency-Domain Attention Implementation
Objective: Enhance transformer robustness to noise in non-stationary neural signals through frequency-domain adaptation [36].
Workflow for Adaptive Frequency Attention
Procedure:
- Input Preparation: Segment neural signals into overlapping windows with 50% overlap, applying pre-emphasis filtering [2]
- Large-Kernel Convolution: Apply ultra-wide convolutional kernels (kernel size ≥ 64) at the initial stage to suppress high-frequency noise
- Multiscale Feature Extraction: Implement parallel dilated convolutions with dilation rates [1, 2, 4, 8] to capture temporal features at multiple timescales
- Gated Residual Enhancement: Process features through GRE-DB module with dual-path extraction and gated downsampling
- Frequency-Domain Attention: Apply adaptive frequency-domain attention to highlight diagnostically relevant frequency components
- Classification: Use feedforward layers with softmax activation for final prediction
Validation: Evaluate using 10-fold cross-validation on Paderborn University bearing fault dataset, reporting accuracy at SNR levels from -4dB to 10dB [36].
Adaptive Decoding Algorithm for Temporal Variability
Objective: Overcome trial-specific timing uncertainties in cognitive tasks like memory recall or motor imagery [14].
ADA Temporal Window Selection
Procedure:
- Temporal Window Generation: For each trial, generate multiple candidate temporal windows with varying onsets and durations
- Window Scoring: Calculate likelihood scores for each window containing task-relevant signals using nonparametric density estimation
- Optimal Window Selection: Select windows with highest likelihood scores for each trial
- Feature Extraction: Compute time-frequency features (wavelet coefficients, band power) from selected windows
- Model Training: Train decoder using features from optimally selected windows rather than fixed time points
- Cross-Validation: Implement leave-one-trial-out cross-validation to assess generalizability
Validation Metrics: Use controlled simulations with known ground truth timing, plus real MEG data from memory recall experiments [14].
Non-Stationarity Robustness Evaluation
Objective: Quantify model resilience to neural population changes over chronic recording periods [37].
Simulation Parameters:
| Non-Stationarity Type | Simulation Metric | Manipulation Range |
|---|---|---|
| Recording degradation | Mean Firing Rate (MFR) | 10-100% of baseline [37] |
| Recording degradation | Number of Isolated Units (NIU) | 10-100% of baseline [37] |
| Neuronal property variance | Preferred Directions (PDs) | 0-180° rotation [37] |
Procedure:
- Baseline Establishment: Train models on initial neural recordings with original statistics
- Controlled Degradation: Systematically vary MFR, NIU, and PDs using population vector models
- Performance Tracking: Evaluate decoder performance (correlation coefficient, R²) at each degradation level
- Comparison Framework: Test multiple decoders (OLE, Kalman Filter, RNN) under both static and retrained schemes
- Breakpoint Analysis: Identify degradation thresholds where performance drops significantly
The Scientist's Toolkit
Research Reagent Solutions
| Reagent/Tool | Function | Application Note |
|---|---|---|
| ALMFormer Architecture | Integrates adaptive frequency attention with large-kernel convolution | Optimal for bearing fault diagnosis under strong noise; achieves superior recognition accuracy at various SNRs [36] |
| Adaptive Decoding Algorithm (ADA) | Nonparametric method for trial-variable neural responses | Specifically designed for cognitive processes with uncertain timing (imagery, memory recall) [14] |
| Recurrent Neural Network Decoders | Nonlinear sequential modeling of neural dynamics | Outperforms OLE and Kalman filters under small recording degradation; sensitive to serious signal degradation [37] |
| PyTorch SDPA | Optimized attention computation with multiple backends | Reduces training step time by ~35% on H100 GPUs; supports FlashAttention-2, memory-efficient attention [38] |
| Population Vector Model | Simulation of neural population dynamics with controllable non-stationarity | Enables systematic testing of decoder robustness to MFR, NIU, and PD changes [37] |
| Gated Residual Enhancement Dual-Branch | Enhanced feature representation in noisy environments | Uses dual-path extraction, gated downsampling, and residual integration [36] |
The Adaptive Decoding Algorithm (ADA) for Trial-Specific Timing
Core Algorithm Concept and Data Features
The Adaptive Decoding Algorithm (ADA) is designed to overcome a fundamental challenge in neural signal analysis: the Heisenberg uncertainty principle, which makes it impossible to simultaneously determine the exact timing and frequency features of impulse components in non-stationary signals using classical Fourier or standard wavelet analysis [40]. ADA addresses this by integrating a model of shift-invariant pattern recognition, inspired by the human visual system's ability to identify "what" and "when" independently, with an advanced wavelet analysis using Krawtchouk functions as the mother wavelet [40]. This integration allows ADA to precisely identify the localization and frequency characteristics of impulse components in EEG signals, such as blinks (0.5-1 Hz) and muscle artifacts (16 Hz), invariant to time shifts [40].
Key Quantitative Features Processed by ADA: The table below summarizes the primary types of neural signal features that ADA is designed to characterize, along with their typical values and experimental significance.
| Feature Type | Description | Example Values / Range | Experimental Significance |
|---|---|---|---|
| Impulse Components [40] | Transient, localized events in the signal (e.g., blinks, muscle artifacts). | Blinks: 0.5 - 1 Hz; Muscle artifacts: ~16 Hz | Identification and removal of noise; isolation of bursts of brain activity. |
| Rhythmic Duration [41] | Length of sustained rhythmic episodes (e.g., theta, alpha) within a single trial. | Frontal Theta: Increased duration with working memory load [41]. | Tracks temporal dynamics of cognitive processes, superior to average power estimates. |
| Trial-to-Trial Variability [42] | Stability of neural spiking activity across trials, measured by Fano Factor (FF). | FF ~1 (Poisson process); Decreased FF during working memory delay indicates stability [42]. | Distinguishes between persistent activity and intermittent burst-coding models of neural computation. |
| Preictal Features [43] | EEG changes predicting seizure onset, including spectral and complexity measures. | Start: 83 ± 60 min before seizure; Duration: 56 ± 47 min [43]. | Key for personalized seizure prediction and intervention; timing varies between individuals and seizures. |
Detailed Experimental Protocols
Protocol for Single-Trial Rhythm Characterization
This protocol is based on the extended Better OSCillation detection (eBOSC) method, used to characterize the power and duration of rhythmic episodes in single trials [41].
- Step 1: Data Acquisition. Record neural signals (EEG, MEG, or LFP) during the cognitive or behavioral task of interest across multiple trials.
- Step 2: Preprocessing. Apply a bandpass filter to the raw signal to focus on the frequency band of interest (e.g., theta: 4-8 Hz, alpha: 8-13 Hz).
- Step 3: Rhythm Detection.
- Calculate the time-frequency representation of the signal using the continuous wavelet transform or similar methods.
- Establish a power threshold and a duration threshold for defining significant rhythmic episodes.
- For each time point and frequency, compare the signal's power against the power threshold. Cluster adjacent supra-threshold points in the time-frequency plane to define rhythmic episodes.
- Discard episodes that do not exceed the minimum duration threshold.
- Step 4: Data Extraction. For each identified rhythmic episode in a trial, extract its duration (ms) and mean amplitude (power). For trial-specific timing, the precise onset and offset of each episode are recorded.
Protocol for Validating ADA on Working Memory Data
This protocol leverages the analysis of trial-to-trial variability (Fano Factor) to test ADA's performance against different theoretical models [42].
- Step 1: Neural Data Collection. Perform single-neuron recordings (e.g., from macaque PFC) during a working memory task with a delay period.
- Step 2: Spike Train Processing. For each neuron and trial, align spike times to a task event (e.g., stimulus onset). Divide the time axis into bins of size Δ (e.g., 50-500 ms).
- Step 3: Fano Factor Calculation.
- For each time bin
t, count the number of spikesN(t,Δ)in that bin for every trial. - Calculate the mean spike count across trials:
〈N(t,Δ)〉. - Calculate the variance of the spike count across trials:
Var(N(t,Δ)). - Compute the Fano Factor:
FF(t,Δ) = Var(N(t,Δ)) / 〈N(t,Δ)〉.
- For each time bin
- Step 4: Model Comparison. Compare the empirically observed FF during the delay period with the predictions of a doubly stochastic Poisson model (which simulates intermittent bursting) and persistent activity models. A low, stable FF supports the persistent activity model and validates ADA's capability to track stable neural states [42].
System Workflow and Signaling Pathways
The following diagram illustrates the core signal processing and decision pathway of the Adaptive Decoding Algorithm (ADA).
Frequently Asked Questions (FAQs) & Troubleshooting
Q1: My decoding accuracy is low and inconsistent across subjects. What could be the issue? A1: High inter-subject variability is a common challenge [44] [41]. To address this:
- Ensure Personalized Feature Selection: Do not assume preictal period onset or optimal features are identical for all patients. Implement a feature importance analysis (e.g., using machine learning models) to identify the best distinguishing features for each subject or session [43]. Common impactful features include Spectral Entropy and Hjorth mobility [43].
- Check Preprocessing: Apply a bandpass filter (e.g., 0.5-75 Hz) to remove constant trends and high-frequency noise. Use a moving window analysis (e.g., 6s windows with 3s overlap) for feature extraction to balance sensitivity and stability [43].
- Consider Advanced Preprocessing: Integrate a Rational Dilated Wavelet Transform (RDWT) as a preprocessing step. RDWT uses non-integer dilation factors for a more flexible time-frequency tiling, which can mitigate localized noise and enhance rhythm-specific information, particularly in challenging, non-stationary recordings [44].
Q2: How can I determine if a observed neural pattern is a sustained rhythm or a series of transient bursts? A2: This is a key distinction for single-trial characterization [41].
- Use Rhythm Detection Algorithms: Employ a method like the extended Better OSCillation detection (eBOSC). This algorithm defines significant rhythmic episodes based on both a power threshold and a minimum duration threshold, effectively separating sustained rhythms from transient, arrhythmic activity [41].
- Analyze Trial-to-Trial Variability: Calculate the Fano Factor (FF) of spike counts across trials. A significant increase in FF that correlates with firing rate suggests an intermittent bursting regime. In contrast, a low and stable FF indicates sustained, persistent activity across trials [42].
Q3: The neural signals are too noisy for reliable decoding of specific components like muscle artifacts. How can I improve signal quality? A3:
- Leverage ADA's Core Innovation: The algorithm based on Krawtchouk wavelets and shift-invariant visual recognition is specifically designed to bypass the limitations of classical analysis and precisely identify the localization and frequency of impulse components like muscle artifacts (e.g., at 16 Hz) even in the presence of noise [40].
- Validate with a Specialist: If working with clinical data, have a specialist physician manually review the segments where the algorithm detects specific activity (like preictal patterns) to confirm they are not explained by artifacts from sleeping, eating, or other activities [43].
Q4: How do I handle the trade-off between temporal and frequency resolution when analyzing non-stationary signals? A4: Standard techniques like STFT have a fixed resolution trade-off.
- Move Beyond Standard Wavelets: While standard Discrete Wavelet Transform (DWT) offers multi-resolution analysis, its fixed integer dilations can be limiting.
- Implement Rational Dilation: Use a Rational Dilated Wavelet Transform (RDWT), which employs non-integer dilation factors (e.g., 3/2, 5/3). This provides a more flexible and adaptive tiling of the time-frequency plane, better capturing the variable dynamics of non-stationary neural signals [44].
The Scientist's Toolkit: Research Reagent Solutions
The table below lists essential computational "reagents" and their functions for implementing and experimenting with ADA.
| Research Reagent (Algorithm/Metric) | Function / Application |
|---|---|
| Krawtchouk Wavelets [40] | A discrete mother wavelet used to precisely compute time and frequency features of local impulses in EEG signals, invariant to time shifts. |
| Rational Dilated Wavelet Transform (RDWT) [44] | A preprocessing technique using non-integer dilation factors for superior time-frequency localization in non-stationary signals like EEG. |
| Fano Factor (FF) [42] | A key metric (variance/mean of spike counts) for quantifying trial-to-trial variability and dissociating neural coding models. |
| Extended BOSC (eBOSC) [41] | A rhythm detection algorithm to characterize the duration and power of sustained rhythmic (vs. arrhythmic) episodes in single trials. |
| Spectral Entropy [43] | A feature quantifying signal irregularity in the frequency domain; often a top discriminator for preictal state identification. |
| Hjorth Mobility [43] | A feature indicating the mean frequency or standard deviation of the signal, useful for characterizing state changes in EEG. |
| Doubly Stochastic Poisson Model [42] | A statistical spiking model used to simulate and test predictions of intermittent burst-coding hypotheses. |
FAQs and Troubleshooting Guides
This technical support center provides solutions for researchers and scientists applying deep learning architectures, particularly within the context of adaptive decoding algorithms for non-stationary neural signals.
Frequently Asked Questions (FAQs)
Q1: Why are hybrid CNN-LSTM architectures particularly suited for processing non-stationary neural signals like EEG?
Hybrid CNN-LSTM architectures are uniquely suited for non-stationary neural data because they simultaneously capture both spatial and temporal features [45] [46]. CNNs excel at extracting local spatial patterns from data arranged in channels or frequency bands, such as identifying features from specific brain regions [47]. LSTMs subsequently model the temporal dependencies in these features, learning how brain signal patterns evolve over time, which is crucial for dealing with signal non-stationarities [48] [49]. This combined spatial-temporal learning makes the hybrid model more robust to the distribution shifts often encountered across different recording sessions or subjects [8].
Q2: What are the most common challenges when training a hybrid model for neural decoding, and how can they be addressed?
Common challenges and their solutions are summarized in the table below.
Table 1: Common Training Challenges and Solutions for Hybrid Models
| Challenge | Description | Potential Solution |
|---|---|---|
| Vanishing Gradients | Difficulty in training LSTM layers over long sequences due to diminishing weight updates. | Use of Rectified Linear Unit (ReLU) or Leaky ReLU activation functions; Gradient clipping [49]. |
| Overfitting | Model performs well on training data but poorly on new, unseen data from a different subject/session. | Implement Dropout and L2 regularization; Employ Domain Adaptation techniques [8]. |
| Class Imbalance | Critical neural events (e.g., specific cognitive states) are rare in the dataset. | Use dynamic class weighting in the loss function (e.g., weighted cross-entropy) [46]. |
| Hyperparameter Tuning | Manual tuning of parameters (e.g., learning rate, filters) is inefficient and suboptimal. | Leverage metaheuristic optimization algorithms like the Squirrel Search Algorithm (SSA) [46]. |
Q3: How can I improve my model's generalization from a source domain (labeled data) to a target domain (new subject/session)?
Improving generalization across domains is a core focus of adaptive decoding. Key strategies include:
- Feature-Based Domain Adaptation (DA): These methods aim to minimize the distributional differences between the source and target domains by projecting their data into a common feature space. This ensures that the reconstructed features follow a similar probability distribution, making the decoder more robust [8].
- Model-Based DA (Fine-tuning): When a small amount of labeled data is available from the target subject or session, you can fine-tune a pre-trained model from the source domain. This approach significantly reduces time and computational costs while adapting the model to the new data characteristics [8].
- Incorporating Attention Mechanisms: Adding an attention layer allows the model to learn to selectively focus on the most informative features or time points in the input data. This improves performance and interpretability by highlighting critical biomarkers in the neural signal [47].
Troubleshooting Guide
This guide addresses specific error messages and performance issues.
Table 2: Troubleshooting Common Experimental Issues
| Problem | Possible Cause | Solution |
|---|---|---|
| Poor accuracy on minority classes (e.g., rare neural events) | Severe class imbalance in the dataset. | Apply dynamic class weighting in the loss function to penalize misclassifications of minority classes more heavily [46]. |
| High training accuracy, but low validation/test accuracy | Overfitting to the training data, often due to domain shift. | (1) Increase dropout and L2 regularization. (2) Apply DA techniques to align feature distributions. (3) Augment training data [8]. |
| Training is unstable (loss oscillates or becomes NaN) | Learning rate is too high; Exploding gradients. | (1) Reduce the learning rate. (2) Implement gradient clipping. (3) Use adaptive optimizers like Adam [49]. |
| Model fails to generalize to a new subject | Domain shift; Inter-subject variability in neural signals. | Implement a Feature-Based DA method to project data from both subjects into a domain-invariant feature space [8]. |
Experimental Protocols and Data Presentation
Protocol 1: Implementing a Basic CNN-LSTM for Neural Signal Classification
This protocol outlines the steps for building a hybrid model to classify neural signals, such as EEG or ECoG.
Workflow Diagram: Basic CNN-LSTM for Neural Signals
Methodology:
- Signal Acquisition & Preprocessing: Acquire raw neural signals (e.g., EEG, ECoG). Preprocess by downsampling, band-pass filtering to remove noise (e.g., muscle artifacts, eye blinks), and normalizing [8].
- Feature Extraction: Extract features from the preprocessed signals. Common approaches include time-frequency domain features (e.g., using wavelets) or functional brain connectivity metrics [8].
- Model Architecture:
- CNN Stage: The formatted input tensor is passed through 1D convolutional layers to extract local spatial patterns across channels or features.
- Sequence Modeling: The CNN's output is reshaped into a sequence of feature vectors and fed into LSTM layers to learn long-term temporal dependencies.
- Attention (Optional): An attention layer can be added after the LSTM to allow the model to focus on the most relevant time steps [47].
- Classification: The final representations are passed through a fully connected layer with a softmax activation for classification.
- Training: Use an Adam optimizer and categorical cross-entropy loss. Employ early stopping and reduce the learning rate on a plateau to prevent overfitting.
Protocol 2: Enhancing Generalization with Domain Adaptation
This protocol describes how to adapt a model trained on one subject (source) to perform well on another (target) using feature-based domain adaptation.
Workflow Diagram: Domain Adaptation for Neural Decoding
Methodology:
- Data Preparation: You will need a large, labeled dataset from the source domain (e.g., multiple subjects) and a smaller, often unlabeled, dataset from the target domain (a new subject) [8].
- Model Architecture: Build a model with a shared feature extractor (a CNN-LSTM backbone) for both source and target data.
- Domain Adaptation Layer: Incorporate a domain adaptation loss that measures the discrepancy between the feature distributions of the source and target domains (e.g., Maximum Mean Discrepancy). The goal is to minimize this loss [8].
- Training: The total loss is a combination of the task-specific loss (e.g., classification loss on the source data) and the domain adaptation loss. This joint training forces the feature extractor to learn representations that are both discriminative for the task and invariant to the domain shift.
The following table summarizes the performance of various deep learning architectures as reported in recent literature, providing a benchmark for expected outcomes.
Table 3: Performance Comparison of Deep Learning Architectures
| Architecture | Application Domain | Key Performance Metrics | Reference / Dataset |
|---|---|---|---|
| Hybrid CNN-LSTM-Attention | Medical Image Diagnosis | Accuracy: >95% (Peak: 98%) across 10 medical image datasets. | [47] |
| Hybrid CNN-LSTM (IntrusionX) | Network Intrusion Detection | Binary Accuracy: 98%; 5-class Accuracy: 87%; High recall for minority classes. | NSL-KDD [46] |
| Hybrid CNN-LSTM | Student Performance Prediction | Accuracy: 98.93% and 98.82% on two educational datasets. | [49] |
| optSAE + HSAPSO | Drug Target Identification | Accuracy: 95.52%; Computational Complexity: 0.010 s/sample. | DrugBank, Swiss-Prot [50] |
The Scientist's Toolkit: Research Reagent Solutions
This section details key computational "reagents" and their functions for building adaptive neural decoders.
Table 4: Essential Research Reagents for Neural Signal Decoding Experiments
| Research Reagent | Function / Explanation | Relevance to Adaptive Decoding |
|---|---|---|
| Preprocessed Datasets (e.g., NSL-KDD, OULAD) | Standardized, benchmark datasets used for training and, most importantly, for fair comparison against other models in the literature. | Provides a reliable baseline for evaluating new DA algorithms before moving to proprietary neural data [46] [49]. |
| Metaheuristic Optimizers (e.g., SSA, HSAPSO) | Algorithms that efficiently search the high-dimensional space of model hyperparameters (learning rate, number of layers, etc.), leading to better and more reproducible performance. | Replaces inefficient manual tuning, which is crucial for finding optimal configurations for complex hybrid models and DA frameworks [50] [46]. |
| Domain Adaptation (DA) Layers | A software component (e.g., using MMD loss) integrated into the model that explicitly reduces the distributional difference between source and target domain features. | The core technical solution for tackling non-stationarity and inter-subject variability, enabling model generalization [8]. |
| Attention Mechanism | A neural network layer that learns to assign a weight (importance score) to different parts of the input, improving performance and providing interpretability. | Helps the model focus on the most salient neural features or time periods, which can be critical for understanding decoding decisions [47]. |
| Grad-CAM Visualization | A technique that produces a heatmap highlighting the regions of the input that were most influential for the model's prediction. | Acts as a diagnostic tool to verify if the model is learning neurologically plausible patterns from the data [47]. |
Troubleshooting Guide and FAQs
This technical support center provides solutions for common challenges in neural signal decoding research, specifically framed within the context of a thesis on Adaptive decoding algorithms for non-stationary neural signals.
Frequently Asked Questions
Q1: How can I improve my model's performance when the timing of cognitive events like memory recall is variable across trials?
A: Temporal variability in neural responses, especially during covert cognitive processes, is a classic challenge for time-locked analyses. To address this:
- Implement the Adaptive Decoding Algorithm (ADA): This nonparametric method uses a two-level prediction. First, it estimates the temporal window most likely to contain the task-relevant signal for each individual trial. Second, it performs decoding based on these selected informative windows. This approach explicitly accounts for trial-specific timing and has been shown to substantially outperform methods that assume a fixed temporal structure [14].
- Leverage Attention Mechanisms: Integrate dot-product attention mechanisms into your model. This allows the network to dynamically learn and focus on the most diagnostically valuable information in time, effectively weighing the importance of different time points, which is crucial for handling temporal uncertainty [51].
Q2: My motor imagery decoding model works well on the training subject but performs poorly on new subjects. What strategies can I use to handle this inter-subject variability?
A: The non-stationarity of EEG signals across subjects is a major obstacle. Transfer learning and domain adaptation are key strategies.
- Employ Multi-Source Dynamic Transfer Learning: Use frameworks like the Multi-source Dynamic Conditional Domain Adaptation Network (MSDCDA). It incorporates a dynamic residual block to adjust network parameters for each subject's specific feature distribution, effectively mitigating conflicts that arise when combining data from multiple source domains. This is complemented by adversarial learning using Margin Disparity Discrepancy (MDD) to align the conditional distributions between source and target domains, significantly enhancing performance on new, unseen subjects [52].
- Apply Classifier-Based Transfer: In scenarios with limited data from a new subject, apply transfer learning directly in the classifier space. Knowledge transferred from previous sessions or other subjects can improve decoding performance by up to 3%, helping to mitigate the effects of non-stationarity [53].
- Utilize Spatial Filtering: For a more traditional approach, use Common Spatial Patterns (CSP) followed by a Linear Discriminant Analysis (LDA) classifier. The CSP filter bank is highly effective at extracting discriminative spatial features for motor imagery, which can then be adapted for new subjects [54].
Q3: How can I build trust in my deep learning model's epileptic seizure detection for clinical use when its decisions are often a "black box"?
A: For clinical adoption, model interpretability is as crucial as accuracy.
- Integrate Explainable AI (XAI) Frameworks: Implement a system like the XAI-based computer-aided ES detection system (XAI-CAESDs). After detection using a Stacking Ensemble Classifier, apply the SHapley Additive exPlanations (SHAP) method. SHAP quantifies the contribution of each input feature (e.g., from a specific EEG channel or time point) to the final prediction, providing medical experts with understandable and actionable insights into why a seizure was flagged [55].
- Incorporate a Hybrid Attention Mechanism: Design your model with a hybrid attention module that learns the importance of features from different EEG channels. This forces the model to "focus" on clinically relevant channels, and the resulting attention weights can be visualized, offering a degree of interpretability into the model's decision process [51].
Q4: What is a robust experimental protocol for collecting EEG data for a lower-limb motor imagery decoding study?
A: A well-designed protocol is critical for generating high-quality, reproducible data.
- Protocol Design: Structure your session with multiple trials (e.g., 22). Each trial should consist of:
- Resting Phase: 15 seconds of relaxation (no motor imagery).
- Cue and Transition: A 2-second acoustic alert between tasks to mark transitions. The data from these short intervals should be excluded from analysis to avoid artifacts from external stimuli.
- Motor Imagery Phase: 30 seconds of kinesthetic motor imagery (e.g., imagining the leg movement of pedaling). To train both initiation and cessation commands, alternate trials between having the rehabilitation device (e.g., a cycle ergometer) active and inactive [54].
- Participant Preparation: Before the first session, provide users with clear guidelines on performing kinesthetic motor imagery (feeling the movement) rather than visual imagery. Administer a questionnaire like the Movement Imagery Questionnaire-3 to assess the user's imagery ability [54].
Experimental Protocols for Key Application Areas
Protocol for Epilepsy Detection with an Explainable AI Framework
This protocol is based on the XAI-CAESDs system for secure and interpretable epileptic seizure detection [55].
EEG Analysis and Decision Pipeline
- Data Acquisition & Security: Collect EEG data according to standard clinical practice. To ensure patient privacy and data integrity, employ a blockchain-based security system to protect the biomedical EEG data [55].
- Preprocessing: Remove artifacts (e.g., noise from muscle movement) using a Butterworth filter [55].
- Feature Engineering:
- Decomposition: Decompose the cleaned EEG signals using a Dual-Tree Complex Wavelet Transform (DTCWT) to extract both real and imaginary eigenvalue components [55].
- Feature Extraction: From the decomposed signals, extract a comprehensive set of features from multiple domains:
- Frequency Domain (FD) features.
- Time Domain (TD) linear features.
- Non-linear features, such as Fractal Dimension (FD) [55].
- Feature Selection: Select the most discriminative features by evaluating Correlation Coefficients (CC) and Distance Correlation (DC) [55].
- Seizure Detection Module: Feed the selected features into a Stacking Ensemble Classifier (SEC), which combines multiple base models to improve overall detection accuracy and robustness [55].
- Explainable Decision-Making: Apply the SHapley Additive exPlanations (SHAP) method to the model's predictions. This provides a quantitative explanation for each detection, highlighting which features and signal components were most influential in the decision, thereby building trust for clinical use [55].
Protocol for Motor Imagery Decoding Using a Dual Approach
This protocol compares a traditional feature-based method with a deep learning approach for decoding motor imagery during a pedaling task [54].
Motor Imagery Decoding Approaches
- Experimental Setup: Recruit participants and instrument them with an EEG cap. Position the participant on a cycle ergometer for a lower-limb motor imagery task [54].
- Data Collection: Execute the motor imagery protocol as described in FAQ A4, ensuring a clear contrast between rest and motor imagery states [54].
- Methodology 1: CSP Filter Bank + LDA
- Feature Extraction: Apply a Common Spatial Patterns (CSP) filter bank to the EEG data. CSP is optimal for obtaining spatial features that maximize the variance between two classes (e.g., rest vs. motor imagery) [54].
- Classification: Feed the extracted spatial features into a Linear Discriminant Analysis (LDA) classifier for final decoding [54].
- Methodology 2: Spectro-Spatial CNN (IFNet)
- Preprocessing: Use a filter bank in the frequency domain as a preprocessing pipeline to decompose the signal into key frequency bands [54].
- Deep Learning Classification: Input the processed data into a Convolutional Neural Network (CNN) like IFNet, which is specifically designed for spectro-temporal and spectro-spatial feature extraction from EEG signals. This approach may show higher accuracy but can suffer from greater instability compared to the CSP-LDA method [54].
- Validation: Perform cross-validation to compare the accuracy, stability, and computational demands of both approaches.
The Scientist's Toolkit: Key Research Reagents and Solutions
Table 1: Essential Algorithms and Computational Tools for Neural Signal Decoding
| Tool/Algorithm Name | Type | Primary Function | Key Advantage |
|---|---|---|---|
| Adaptive Decoding Algorithm (ADA) [14] | Decoding Algorithm | Decodes mental contents with variable timing | Handles trial-by-trial latency variations in neural dynamics |
| Multi-source Dynamic Conditional Domain Adaptation (MSDCDA) [52] | Transfer Learning Framework | Improves cross-subject decoding performance | Mitigates multi-source domain conflict via dynamic residual blocks |
| SHapley Additive exPlanations (SHAP) [55] | Explainable AI (XAI) Method | Interprets model predictions | Provides quantitative feature contribution values for clinical trust |
| Common Spatial Patterns (CSP) [54] | Feature Extraction Algorithm | Extracts discriminative spatial features for MI | Highly effective for separating two classes in motor imagery paradigms |
| Stacking Ensemble Classifier (SEC) [55] | Classification Model | Detects epileptic seizures from EEG features | Combines multiple models for higher accuracy and robustness |
Table 2: Key Software and Data Processing Libraries
| Library/Framework | Application Context | Usage Note |
|---|---|---|
| TensorFlow / Keras [56] | General-purpose deep learning | Used for building and training RNNs, CNNs, and other models (e.g., for seizure detection) |
| Dual-Tree Complex Wavelet Transform (DTCWT) [55] | Signal Decomposition | Used in epilepsy detection for analyzing EEG signals and extracting complex features |
| Filter Banks [54] | Signal Preprocessing | Used in motor imagery decoding to separate EEG signals into relevant frequency bands |
Overcoming Practical Hurdles: Optimization Strategies and Bias Mitigation
Addressing Computational Complexity and Real-Time Processing Demands
Frequently Asked Questions (FAQs)
What are the main sources of computational complexity in neural signal decoding? Computational complexity arises from processing high-dimensional, non-stationary neural signals (EEG, ECoG, spike signals) and the sophisticated algorithms required for domain adaptation and feature extraction. Time-frequency transformations and real-time processing demands further contribute to this complexity [8].
How can I determine if my decoding performance issues are due to neural non-stationarity? Performance degradation over time or across sessions, especially with static decoders, often indicates non-stationarity. This manifests as dropping mean firing rates (MFR), decreasing numbers of isolated units (NIU), or shifting neural preferred directions (PDs). Implementing a retraining scheme can help isolate and confirm this issue [37].
What is the practical difference between static and retrained decoder schemes? In a static scheme, the decoder is trained once on initial data and remains fixed. It is simple to deploy but performance degrades with neural changes. A retrained scheme involves regular recalibration of the decoder on new data, improving robustness to non-stationarity at the cost of increased computational overhead and required recalibration data [37].
Why is my system experiencing high latency during real-time decoding? Latency is intrinsic to decoding algorithms that require future signal samples. For instance, a windowed Discrete Wigner-Ville Distribution (DWVD) has a latency limited to half the window duration. The total latency is the sum of the algorithm's intrinsic delay and the computational time for operations like FFTs [57].
Troubleshooting Guides
Issue: High Computational Load and Slow Processing
Problem: Your experiment runs slowly, fails to process data in real-time, or consumes excessive memory.
Possible Causes and Solutions:
Cause 1: Inefficient Feature Extraction or Signal Transformation.
- Solution: Evaluate your signal processing pipeline. For 1D to 2D signal conversion, consider efficient algorithms like Bresenham's line algorithm (time complexity O(n)) as an alternative to more computationally intensive methods like STFT or wavelets [58].
- Solution: Simplify features or reduce the dimensionality of the data before feeding it into the decoder.
Cause 2: Hardware and Software Limitations.
- Solution:
- Check for Memory Leaks: Use system monitoring tools (e.g., Task Manager,
top) to identify if your application is using increasingly large amounts of RAM over time [59]. - Update Software: Ensure your operating system, programming languages (e.g., Python, MATLAB), and critical libraries are updated to the latest stable versions, as updates often include performance optimizations [60].
- Switch to 64-bit Architecture: If you are using a 32-bit version of your software (e.g., MATLAB, Python), migrating to a 64-bit version can dramatically increase the available memory address space, preventing "out of memory" errors with large datasets [60].
- Check for Memory Leaks: Use system monitoring tools (e.g., Task Manager,
- Solution:
Cause 3: Suboptimal Decoder or Training Scheme.
- Solution: For non-stationary signals, select decoders known for robust performance, such as Recurrent Neural Networks (RNNs), which have demonstrated better resilience compared to linear decoders like the Kalman Filter or Optimal Linear Estimation [37]. Weigh the benefits of a static scheme (low computational cost) versus a retrained scheme (higher cost but more accurate over time).
Issue: Poor Decoding Accuracy Over Time
Problem: Your decoder's performance is initially good but degrades across experimental sessions or within a single long session.
Possible Causes and Solutions:
Cause 1: Neural Signal Non-Stationarity.
- Solution: Actively monitor key metrics of non-stationarity from your neural signals, such as Mean Firing Rate (MFR), Number of Isolated Units (NIU), and Preferred Directions (PDs) [37]. A significant change in these metrics confirms the issue.
- Solution: Implement Domain Adaptation (DA) techniques. These are specifically designed to mitigate the effects of distribution shifts between training (source domain) and deployment (target domain) data. DA can be instance-based (weighting source samples), feature-based (transforming features to a common space), or model-based (fine-tuning a pre-trained model) [8].
- Solution: For tasks with uncertain timing (e.g., memory recall, imagery), use algorithms like the Adaptive Decoding Algorithm (ADA), which accounts for trial-specific temporal variability instead of assuming fixed timing [14].
Cause 2: Inadequate Handling of Abrupt Signal Transitions.
- Solution: If your signals have abrupt frequency transitions, standard instantaneous frequency (IF) estimators may fail. Employ specialized IF estimation algorithms designed to handle such non-stationary components and their intersections in the time-frequency domain [61].
Experimental Protocols & Methodologies
Protocol 1: Evaluating Decoder Robustness to Simulated Non-Stationarity
This protocol allows you to systematically test how different decoders perform under controlled, simulated non-stationarity before deploying them in real experiments [37].
1. Objective: To compare the performance of decoders (OLE, KF, RNN) under various types and degrees of simulated neural signal non-stationarity.
2. Materials and Input Data:
- A set of ground-truth kinematic data (e.g., from a 2D center-out task).
- A neural simulation model (e.g., a Population Vector - PV - model).
- Decoder implementations (OLE, KF, RNN).
3. Procedure:
- Step 1: Generate Baseline Spike Data. Use the PV model and the ground-truth kinematic data to simulate baseline spike signals [37].
- Step 2: Introduce Non-Stationarity. Systematically alter the simulated spike data using three key metrics [37]:
- Recording Degradation: Decrease the Mean Firing Rate (MFR) and the Number of Isolated Units (NIU).
- Neural Variance: Change the Preferred Directions (PDs) of the neural units.
- Step 3: Train Decoders. Train the OLE, KF, and RNN decoders on the initial (non-degraded) simulated data (static scheme) or on data from each degradation level (retrained scheme) [37].
- Step 4: Evaluate Performance. Test all decoders on the degraded datasets and quantify performance using a metric like decoding accuracy or kinematic estimation error [37].
4. Expected Output: A comparison of decoder robustness, typically showing that RNNs with a retraining scheme maintain higher performance under moderate non-stationarity [37].
Experimental workflow for simulating and testing neural signal non-stationarity.
Protocol 2: Implementing a Real-Time Decoding Pipeline with Latency Control
1. Objective: To establish a real-time neural decoding pipeline with known and managed latency.
2. Materials:
- Neural signal acquisition system (e.g., EEG, ECoG, or spike recording setup).
- Processing computer meeting specified computational requirements.
3. Procedure:
- Step 1: Signal Preprocessing. Apply necessary filters and downsample the neural data to a rate suitable for your decoding task [8].
- Step 2: Feature Extraction. Choose a computationally efficient feature extraction method. For image-based deep learning models, consider efficient 1D-to-2D conversion algorithms (e.g., Bresenham's line algorithm, O(n) complexity) [58].
- Step 3: Algorithm Selection and Optimization.
- Calculate the intrinsic latency of your chosen decoding algorithm. For a windowed analysis, this is typically half the window length [57].
- Optimize code to minimize computational delay. This may involve using optimized libraries, reducing FFT sizes, or leveraging the realness property of TFDs to halve the number of required FFTs [57].
- Step 4: Latency Measurement. The total latency is the sum of the intrinsic algorithmic latency and the computational delay. Measure the computational delay empirically on your target hardware.
Data Presentation
Table 1: Computational Complexity of Common Signal Processing Operations
| Operation / Algorithm | Time Complexity | Key Characteristics & Notes |
|---|---|---|
| Bresenham's Line Algorithm [58] | O(n) | Efficient rasterization for 1D-to-2D conversion; uses integer arithmetic. |
| Fast Fourier Transform (FFT) [58] | O(N log N) | Standard for spectral analysis; computationally intense for large N. |
| Short-Time Fourier Transform (STFT) [58] | O(N log N) | Adds temporal context to FFT; complexity depends on windowing parameters. |
| Digital Differential Analyzer (DDA) [58] | > O(n) | Less efficient than Bresenham's; uses floating-point operations. |
| 2D Convolution (CNN) [58] | O((M*N) * k²) | High cost with large image sizes (M, N) and kernel size (k). |
| Non-Stationarity Type | OLE (Static) | KF (Static) | RNN (Static) | RNN (Retrained) |
|---|---|---|---|---|
| MFR Decrease (Mild) | Significant Drop | Moderate Drop | Small Drop | Maintained Performance |
| NIU Decrease (Mild) | Significant Drop | Moderate Drop | Small Drop | Maintained Performance |
| PD Shift (Mild) | Significant Drop | Significant Drop | Moderate Drop | Maintained Performance |
| Severe Signal Degradation | Major Performance Drop | Major Performance Drop | Significant Drop | Performance Drop (but best overall) |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Tools for Adaptive Neural Decoding
| Item / Solution | Function in Research | Example/Note |
|---|---|---|
| Domain Adaptation (DA) [8] | Enhances decoder generalizability across subjects/sessions by reducing distribution differences. | Categories: Instance-based (sample weighting), Feature-based (space transformation), Model-based (fine-tuning). |
| Adaptive Decoding Algorithm (ADA) [14] | Decodes tasks with uncertain timing (e.g., recall, imagery) by estimating trial-specific temporal windows. | Non-parametric method; addresses misalignment in neural dynamics. |
| Recurrent Neural Network (RNN) [37] | A non-linear decoder that uses sequential information; shows superior robustness to non-stationarity. | Outperforms OLE and KF under simulated signal degradation and neural variance. |
| Population Vector (PV) Model [37] | A physiologically-inspired model for simulating spike data in motor-related tasks. | Used to generate controlled datasets for testing decoder robustness. |
| Retraining Scheme [37] | A protocol where decoders are regularly updated with new data to combat non-stationarity. | Improves performance compared to static schemes but requires more data and computation. |
Domain adaptation (DA) strategies for handling neural non-stationarity.
Strategies for Mitigating Overfitting in High-Dimensional Neural Data
Troubleshooting Guide: Key Questions and Answers
Q1: Why is my neural network model performing well on training data but poorly on unseen EEG or neural data?
This is a classic sign of overfitting. It occurs when your model learns the noise, random fluctuations, and specific details of the training dataset instead of the underlying patterns that generalize to new data [62] [63]. In high-dimensional neural data, this problem is exacerbated because the vast number of features (e.g., from multi-channel EEG recordings) allows the model to memorize the training examples easily [64] [65]. You can identify this by a large performance gap where training accuracy is very high, but validation or test accuracy is significantly worse [62] [63].
Q2: Our high-dimensional single-cell RNA-seq data is sparse and has few samples. How can we prevent overfitting in this "small n, large p" scenario?
This is a common challenge in biomedical research. A proposed framework to address this combines dimensionality reduction with data augmentation [66].
- Dimensionality Reduction: Use Random Projection (RP) and Principal Component Analysis (PCA) to reduce the feature space while preserving the data's relative structure. This directly counters the "curse of dimensionality" [66] [67].
- Data Augmentation: Generate multiple, lower-dimensional versions of your original samples through random projections. This artificially increases your training set size, which helps the model learn more robust and generalizable patterns [66].
- Ensemble Boosting: During inference, use a majority voting technique on the predictions from these multiple augmented samples to enhance the final classification's reliability and consistency [66].
Q3: How can we improve the decoding accuracy of non-stationary EEG signals where the timing of task-relevant neural activity varies across trials?
Traditional time-locked analysis methods struggle with this. The Adaptive Decoding Algorithm (ADA) is specifically designed for this problem. It operates in two key steps [14]:
- Temporal Window Estimation: For each trial, ADA estimates the temporal window most likely to contain the task-relevant signals, accounting for trial-specific timing variability.
- Decoding Based on Informative Windows: The test trials are then decoded based on the selection of these informative temporal windows, explicitly accounting for the uncertain timing of neural dynamics [14].
Q4: What model architecture choices can help make a deep learning model more robust for EEG analysis?
Consider using an adaptive Transformer-based framework. Standard models like CNNs and LSTMs have limitations in capturing long-range dependencies and spatial interactions in EEG data [11]. An adaptive Transformer offers several advantages:
- Adaptive Attention Mechanism: It uses a domain-specific adaptive attention mask to dynamically focus on the most important temporal and spatial features in the EEG signal, reducing the impact of noise and variability [11].
- Temporal-Spatial Modeling: Its multi-head self-attention mechanism can capture long-range temporal dependencies and, when combined with spatial attention, model interactions between different EEG channels [11].
- Interpretability: The attention weights can be visualized, providing insights into which parts of the EEG data were most important for the model's prediction, which is valuable for clinical decision-making [11].
Experimental Protocols for Cited Methods
This protocol is designed for high-dimensional, sparse data like single-cell RNA-seq.
- Data Preprocessing: Normalize the raw data. Split the dataset into training, validation, and test sets.
- Dimensionality Reduction & Augmentation:
- For each sample in the training set, generate
kdifferent random projections to createknew, lower-dimensional sample representations. - Apply PCA to these projected spaces to further refine and preserve critical structure.
- For each sample in the training set, generate
- Model Training:
- Train a single Neural Network model on the entire augmented training set (the original samples plus their
krandomly projected variants).
- Train a single Neural Network model on the entire augmented training set (the original samples plus their
- Inference with Majority Voting:
- For a new test sample, generate the same
krandom projections to createkrepresentations of the test sample. - Pass each of the
ktest sample representations through the trained network to getkprediction vectors. - Apply a majority voting scheme on these
kpredictions to determine the final, consolidated classification output.
- For a new test sample, generate the same
This protocol outlines the workflow for implementing an adaptive Transformer model for EEG data.
- Data Preprocessing:
- Load raw EEG data (e.g., from TUH EEG Corpus or CHB-MIT).
- Preprocess to remove noise and artifacts (e.g., band-pass filtering, artifact removal).
- Segment the continuous EEG signal into time-series chunks or epochs related to the task (e.g., motor imagery cues).
- Feature Embedding:
- Convert the preprocessed time-series chunks into a structured input for the Transformer.
- Apply channel-wise embeddings to represent the spatial information from different EEG electrodes.
- Incorporate temporal encoding (positional encoding) to retain the sequential order of the data.
- Model Training with Adaptive Transformer:
- Architecture: Implement a Transformer encoder block with a multi-head self-attention mechanism.
- Adaptive Attention Mask: Integrate a domain-specific adaptive attention mask that dynamically weights important temporal and spatial features.
- Training: Train the model end-to-end to classify the EEG epochs, using the preprocessed and embedded data.
- Validation and Interpretation:
- Evaluate performance on a held-out test set using accuracy, precision, recall, and F1-score.
- Visualize the attention maps from the trained model to identify which time points and EEG channels were most critical for the classification, aiding in interpretability.
Table 1: Performance Comparison of Different Feature Selection Methods on Classification Accuracy
| Feature Selection Method | Dataset | Key Principle | Reported Classification Accuracy |
|---|---|---|---|
| Two-phase Mutation Grey Wolf Optimization (TMGWO) [64] | Wisconsin Breast Cancer | Hybrid AI-driven algorithm enhancing exploration/exploitation balance. | 98.85% (on diabetes dataset), 96% (on Breast Cancer with SVM) |
| Improved Salp Swarm Algorithm (ISSA) [64] | Wisconsin Breast Cancer, Sonar | Incorporates adaptive inertia weights and local search techniques. | Outperformed by TMGWO in experimental comparison [64]. |
| Binary Black Particle Swarm Optimization (BBPSO) [64] | Wisconsin Breast Cancer, Sonar | Uses a velocity-free mechanism for global search efficiency. | Outperformed by TMGWO in experimental comparison [64]. |
| BP-PSO (with chaotic model) [64] | Multiple data sets | Combines Backpropagation (BP) neural networks with PSO. | Average accuracy 8.65% higher than a benchmark model (NDFs) [64]. |
Table 2: Performance of Advanced Decoding and Modeling Frameworks on Neural Data
| Model / Framework | Data Type | Key Innovation | Reported Performance |
|---|---|---|---|
| Adaptive Decoding Algorithm (ADA) [14] | MEG (Simulated and real perception/recall data) | Non-parametric, two-level prediction that aligns trial-specific temporal windows. | Outperforms methods assuming fixed temporal structure [14]. |
| Adaptive Transformer [11] | EEG (TUH EEG Corpus, CHB-MIT) | Adaptive attention mask for spatial-temporal modeling of EEG. | 98.24% accuracy, outperforming standard CNNs and LSTMs [11]. |
| RP-PCA Ensemble Framework [66] | scRNA-seq (17 datasets) | Data augmentation via Random Projections and PCA for "small n, large p" problems. | Outperforms state-of-the-art scRNA-seq classifiers and is comparable to XGBoost [66]. |
| Anchored-STFT & Skip-Net [68] | EEG (BCI Competition datasets) | Advanced STFT with variable windows and a shallow CNN with skip connections. | 90.7% accuracy on BCI competition II dataset III [68]. |
Workflow and Conceptual Diagrams
Adaptive Decoding for Non-Stationary Signals
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Tools and Methods
| Item / Method | Function / Purpose | Example Use Case in Research |
|---|---|---|
| Two-phase Mutation Grey Wolf Optimization (TMGWO) [64] | A hybrid AI-driven feature selection algorithm that identifies the most relevant features from high-dimensional datasets, reducing model complexity. | Selecting key biomarkers from high-dimensional genomic or neuroimaging data before classification. |
| Random Projections (RP) [66] | A dimensionality reduction technique that preserves data structure based on the Johnson-Lindenstrauss lemma, used for data augmentation and noise reduction. | Addressing the "small n, large p" problem in single-cell RNA-seq data analysis to improve neural network training. |
| Adaptive Transformer [11] | A deep learning architecture with self-attention and adaptive masks to model complex temporal and spatial relationships in non-stationary signals. | Decoding EEG signals for brain-computer interfaces or classifying cognitive states from MEG/EEG recordings. |
| Anchored-STFT [68] | A feature extraction method that improves upon STFT by using multiple variable-length windows to optimize time-frequency resolution. | Generating enhanced spectrogram representations from raw EEG signals for motor imagery classification. |
| Gradient Norm Adversarial Augmentation (GNAA) [68] | A data augmentation method that generates adversarial inputs to improve model robustness and classification accuracy. | Increasing the effective training set size and harnessing adversarial examples for EEG signal classifiers. |
| Adaptive Decoding Algorithm (ADA) [14] | A non-parametric decoding algorithm that accounts for trial-by-trial timing variability in neural responses. | Analyzing neural data from cognitive tasks like memory recall or imagery, where event timing is not locked to external cues. |
Ensuring Model Generalizability Across Subjects and Recording Sessions
A core challenge in modern neuroscience and drug development is that neural signals are fundamentally non-stationary. Their statistical properties change over time within a single subject and vary significantly between different individuals [37] [8]. This non-stationarity, caused by factors like neuronal adaptation, recording device instability, and individual neurophysiological differences, poses a major threat to the reliability and generalizability of neural decoding models [37]. Adaptive decoding algorithms are designed to overcome this hurdle, enabling robust brain-computer interfaces (BCIs) and reliable pharmacodynamic biomarkers for clinical trials [69] [8]. This guide provides targeted troubleshooting advice for researchers tackling the pervasive issue of model performance degradation across subjects and sessions.
Frequently Asked Questions (FAQs)
FAQ 1: Why does my model's performance drop significantly when I test it on data from a new subject or a new recording session from the same subject?
This performance drop is primarily due to domain shift, a situation where the data used for training (the source domain) and the data encountered during deployment (the target domain) have different probability distributions, despite representing the same underlying tasks or conditions [8]. For neural signals, this shift manifests as:
- Changes in Signal Amplitude and Quality: Chronic recordings often show a decrease in the mean firing rate (MFR) and the number of isolated units (NIU) over time due to biological reactions to implants [37].
- Changes in Neuronal Tuning Properties: The preferred directions (PDs) of neurons can change as subjects adapt to decoders or due to natural learning processes [37].
- Individual Physiological Variability: The brain's functional anatomy and electrical activity patterns are unique to each individual, making a one-size-fits-all model ineffective [8].
FAQ 2: What is the fundamental difference between "cross-subject" and "cross-session" generalization problems?
While both problems stem from non-stationarity, they differ in scope and primary causes:
- Cross-Session Generalization focuses on maintaining performance for the same subject over different time periods (e.g., days or weeks). The main challenges here are recording instability (changes in MFR, NIU) and within-subject neuronal adaptation (changes in PDs) [37].
- Cross-Subject Generalization aims to create a model that works for new, unseen subjects. The dominant challenge is inter-individual variability in brain anatomy, physiology, and cognitive strategy, which leads to a much larger domain shift than typically seen across sessions [8].
FAQ 3: My model performs well during training but fails on new data. Which adaptive decoding strategies should I prioritize?
Your strategy should be chosen based on the amount of labeled data available from the target subject/session. The following table summarizes the core approaches:
| Strategy | Core Principle | Ideal Use Case | Key Advantage |
|---|---|---|---|
| Instance-Based DA [8] | Re-weight or select source domain samples that are most similar to the target domain. | Limited target data; multiple source datasets available. | Reduces negative transfer by focusing on relevant source data. |
| Feature-Based DA [8] | Learn a domain-invariant feature space where source and target distributions are aligned. | Moderate amount of unlabeled target data is available. | Directly minimizes the distributional difference between domains. |
| Model-Based DA [8] | Fine-tune a model pre-trained on the source domain using a small amount of target data. | A small amount of labeled target data can be collected. | Leverages pre-trained knowledge; highly efficient and effective. |
| Deep Domain Adaptation [8] | Use deep learning models (e.g., CNNs, RNNs) to automatically extract features combined with DA losses. | Complex neural signals (EEG, spikes); large and diverse source datasets. | End-to-end learning; superior performance on complex tasks. |
FAQ 4: Are certain types of decoders inherently more robust to non-stationarity?
Yes, decoder architecture significantly impacts robustness. Recurrent Neural Networks (RNNs) have demonstrated superior performance compared to traditional decoders like Kalman Filters (KF) and Optimal Linear Estimation (OLE) when dealing with non-stationary spike signals [37]. RNNs are better at capturing temporal dynamics and sequential patterns in neural data, which can be more stable across sessions than moment-to-moment firing rates. However, combining a powerful decoder like an RNN with explicit domain adaptation techniques typically yields the best overall performance [8].
Troubleshooting Guides
Problem: Poor Cross-Session Performance for a Single Subject
Symptoms: Model accuracy degrades when applied to data recorded from the same subject on a different day.
Step-by-Step Diagnostic and Solution Protocol:
Quantify the Non-Stationarity:
- Calculate the change in key neural metrics between your training session and the new session. Compute the mean firing rate (MFR) and the number of isolated units (NIU) for both sessions. A significant drop suggests recording degradation [37].
- If tuning model data is available, compare the neural preferred directions (PDs). Significant changes indicate neuronal property variance [37].
Select and Apply a Remediation Strategy:
- If MFR/NIU have dropped but PDs are stable: Employ a retraining scheme. Retrain your decoder using a small amount of newly collected data from the target session. This is a form of model-based adaptation that directly addresses the shifted distribution [37] [8].
- If PDs have significantly changed: Relying on a static decoder will lead to persistent failure. Prioritize a feature-based DA method. Algorithms that project source and session data into an aligned feature space can compensate for these tuning property changes [8].
- For the most robust long-term solution: Implement an adaptive decoding algorithm that dynamically adjusts its parameters based on recent neural activity. This creates a continuously learning system that can track non-stationarity in real-time [37].
Problem: Poor Cross-Subject Generalization
Symptoms: A model trained on a group of subjects fails to perform accurately on a new, unseen subject.
Step-by-Step Diagnostic and Solution Protocol:
Preprocess with Domain Alignment in Mind:
- Beyond standard filtering, use algorithmic techniques to explicitly reduce inter-subject differences. Techniques like Euclidean Alignment can spatially filter data to create a more subject-invariant signal space before feature extraction [8].
Choose a Domain Adaptation Framework:
- If you have no labeled data from the new target subject: Use unsupervised domain adaptation. Employ feature-based DA methods such as Transfer Component Analysis (TCA) or Domain Adversarial Neural Networks (DANN) to minimize distribution divergence between the source subjects and the unlabeled target subject data [8].
- If you can collect a small labeled dataset from the target subject: Use supervised domain adaptation. A model-based approach is highly effective. Fine-tune a model pre-trained on your source subjects using the small target dataset. This is often the most practical and efficient path to high performance [8].
- If your source data comes from many different subjects: Treat this as a multi-source domain adaptation problem. Use methods that can selectively weight contributions from different source subjects based on their similarity to the target subject, preventing "negative transfer" from dissimilar sources [8].
Problem: Model is Overfitting to Subject-Specific Noise
Symptoms: The model performs exceptionally well on the training subject but fails to generalize to any other subject, indicating it has learned idiosyncratic noise rather than the underlying neural code.
Step-by-Step Diagnostic and Solution Protocol:
Increase Data Diversity and Augmentation:
- Ensure your training set incorporates data from a large number of subjects (if available) to force the model to learn invariant features.
- Artificially augment your training data by adding controlled noise or simulating common non-stationarities (e.g., slight amplitude scaling, random channel drops) to improve robustness.
Implement Feature-Based Domain Adaptation:
- This is the primary solution. Methods like Subspace Alignment (SA) or Geodesic Flow Kernel (GFK) explicitly learn features that are discriminative for the task but invariant to the subject-specific domain. This strips away subject-specific noise and retains the core signal of interest [8].
Regularize the Model:
- Apply stronger regularization techniques (e.g., L2 regularization, dropout) during training to discourage the model from relying on complex, subject-specific patterns that do not generalize.
Experimental Protocols & Performance Data
Protocol 1: Benchmarking Decoder Robustness to Simulated Non-Stationarity
This protocol, derived from simulation studies [37], allows for controlled testing of decoders against specific types of non-stationarity.
1. Objective: To evaluate and compare the robustness of different decoders (OLE, KF, RNN) against controlled introductions of recording degradation and neuronal variance.
2. Materials & Signals:
- A pre-existing neural dataset (e.g., spike times) and corresponding kinematic data from a center-out task.
- A validated neural signal simulator based on the Population Vector (PV) model [37].
3. Methodology:
- Step 1 - Baseline Simulation: Use the PV model and kinematic data to generate a baseline spike signal.
- Step 2 - Introduce Non-Stationarity: Systematically alter the simulated neural data using three metrics:
- MFR & NIU: Decrease values to simulate recording degradation.
- Neural PDs: Change values to simulate neuronal property variance.
- Step 3 - Decoder Training & Evaluation: Train OLE, KF, and RNN decoders on the baseline data (static scheme) or retrain them on the altered data (retrained scheme). Evaluate their performance (e.g., with Pearson's correlation coefficient) on the non-stationary simulated data.
4. Expected Outcomes: Simulation results consistently show that the RNN decoder outperforms OLE and KF under non-stationary conditions. Furthermore, the retrained scheme is crucial for maintaining performance when neuronal PDs change [37].
Protocol 2: A Standardized Pipeline for Cross-Subject EEG Decoding
This protocol outlines a standard workflow for applying Domain Adaptation (DA) to cross-subject EEG analysis, as surveyed in [8].
1. Objective: To build a robust EEG-based classifier (e.g., for emotion recognition or drug effect detection) that generalizes to new, unseen subjects.
2. Materials & Signals:
- Multi-subject EEG dataset with task labels.
- Standard EEG preprocessing pipeline (filtering, artifact removal).
3. Methodology:
- Step 1 - Data Preparation: Designate one or more subjects as the source domain and one or more as the target domain. Apply preprocessing.
- Step 2 - Feature Extraction: Extract features from all domains. Common features include time-frequency features (from a Wavelet transform or STFT) or functional connectivity metrics [8].
- Step 3 - Domain Adaptation: Apply a chosen DA method. For a practical setup with a small labeled target dataset, a feature-based method like Transfer Component Analysis (TCA) or a model-based fine-tuning approach is recommended.
- Step 4 - Classification & Validation: Train a classifier (e.g., SVM) on the adapted source features (and limited target labels) and validate its performance strictly on the held-out target subject data.
4. Expected Outcomes: Studies show that employing DA can significantly improve cross-subject classification accuracy compared to non-adaptive models. The table below summarizes the performance of various adaptive decoding frameworks on different tasks:
| Framework / Method | Core Adaptive Mechanism | Task / Context | Key Performance Findings |
|---|---|---|---|
| RNN Decoder with Retraining [37] | Model retraining on new session data. | iBCI cursor control (simulated). | Maintains high performance with small recording degradation or PD changes. |
| Feature-Based DA (e.g., TCA) [8] | Learning domain-invariant features. | Cross-subject EEG classification. | Significantly outperforms non-adaptive models; accuracy improvements of 10-20% are common. |
| Instance-Based DA [8] | Re-weighting source domain samples. | Cross-subject/session neural decoding. | Effective in selecting relevant source data, improving robustness. |
| Deep DA (e.g., DANN) [8] | End-to-end deep learning with domain adversarial loss. | Complex EEG/ECoG decoding tasks. | Achieves state-of-the-art performance by learning robust, invariant features directly from data. |
Signaling Pathways & Workflows
Adaptive Decoding for Non-Stationary Signals Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item / Solution | Function in Research | Example Application in Context |
|---|---|---|
| Recurrence Quantification Analysis (RQA) [70] | A non-linear method to quantify the complexity and dynamics of a time series. | Used to extract entropy indices from EEG to detect changes in brain complexity associated with multidrug dependence, serving as a biomarker. |
| Bresenham's Line Algorithm [58] | A computationally efficient algorithm (O(n)) to convert 1D non-stationary signals into 2D image representations. | Used as a preprocessing step to transform neural spikes or EEG segments into 2D images for classification with image-based deep learning models (e.g., 2D CNN). |
| Population Vector (PV) Model Simulator [37] | A computational model to simulate neural spike data based on kinematic parameters and neuronal tuning properties. | Critical for conducting controlled simulation studies to evaluate how decoders perform under specific types of introduced non-stationarity (e.g., changing PDs). |
| Transfer Component Analysis (TCA) [8] | A feature-based domain adaptation method that learns a set of transfer components that minimize the distribution difference between domains. | Applied to EEG features to create a domain-invariant representation, improving cross-subject classification accuracy for tasks like emotion recognition or seizure detection. |
| Kalman Filter (KF) Decoder [37] | A classical state-space model decoder that uses a series of measurements over time to produce estimates of kinematic variables. | Serves as a baseline decoder in iBCI studies; performance is compared against more robust decoders like RNN under non-stationary conditions. |
| Recurrent Neural Network (RNN) Decoder [37] | A deep learning decoder designed to handle sequential data, capturing temporal dependencies in neural activity. | The preferred decoder for handling non-stationary neural signals due to its inherent ability to model temporal dynamics and its superior robustness compared to KF and OLE. |
In the field of adaptive decoding algorithms for non-stationary neural signals, data quality is paramount. Neural signals, such as EEG and sEEG, are inherently non-stationary and susceptible to low signal-to-noise ratios (SNR) and various artifacts. These data quality issues can severely compromise the performance and reliability of decoding algorithms, hindering both scientific discovery and clinical application. This guide provides researchers and drug development professionals with targeted troubleshooting strategies to identify, address, and prevent common data quality problems in their neural signal research.
FAQs: Addressing Common Data Quality Challenges
What are the most prevalent data quality issues in neurological data?
The most common issues can be categorized as follows [71]:
- Inaccurate Data: Errors introduced during data entry or recording.
- Incomplete Data: Essential information or data segments that are missing.
- Low Signal-to-Noise Ratio (SNR): The target neural signal is obscured by background biological or environmental noise [72].
- Artifacts: Unwanted signals from non-neural sources, such as muscle activity (EMG), eye blinks (EOG), and head movements [8] [33].
- Non-Stationarity: The statistical properties of the neural signal (e.g., mean, variance) change over time, leading to domain shifts between training and testing data [8] [73].
How do data quality issues impact the development of adaptive decoding algorithms?
Data quality issues directly undermine algorithm performance [74] [73]:
- Reduced Generalizability: Non-stationarity causes models trained on one dataset to fail on another from a different subject or session, a core challenge known as domain shift [8] [73].
- Obscured Treatment Effects: In clinical trials for neurological conditions, excessive noise and artifacts can mask a drug's true efficacy, leading to false negatives or inflated placebo responses [74].
- Inaccurate Models: Artifacts can be mistakenly learned as features by the model, leading to artificially inflated decoding accuracy and incorrect conclusions about brain function [33].
Can artifact correction improve the decoding performance of my model?
The relationship is nuanced. A 2025 study evaluating artifact correction and rejection found that while these steps are crucial, they do not always significantly boost decoding accuracy and their primary value is in ensuring validity [33].
Table: Impact of Artifact Correction on Decoding Performance
| Scenario | Impact on Decoding Performance | Recommendation |
|---|---|---|
| Simple Binary Tasks (e.g., P3b, N400) | Minimal performance gain from correction + rejection | Artifact correction is still essential to prevent confounds. |
| Challenging Multi-Way Tasks (e.g., stimulus orientation) | Minor performance improvements possible | Prioritize artifact correction; rejection may help if trial count is high. |
| Preventing Inflated Accuracy | Critical | Always use artifact correction to ensure features are neural in origin. |
The study strongly recommends artifact correction (e.g., using Independent Component Analysis (ICA) for ocular artifacts) before decoding analyses to eliminate the risk of artifact-related confounds that could lead to invalid results [33].
What strategies can mitigate the effects of non-stationarity in neural signals?
Non-stationarity is a fundamental challenge, but several adaptive techniques can address it [8] [73]:
- Domain Adaptation (DA): This family of techniques aims to minimize distributional differences between source (training) and target (testing) domains. This can be done by re-weighting source samples, transforming features into a shared space, or fine-tuning model parameters [8].
- Test-Time Adaptation (TTA): This method adjusts a pre-trained model online using unlabeled test data. For example, entropy minimization on incoming test batches can help the model adapt to distribution shifts during inference without needing the original training data [73].
- Multi-Scale Feature Learning: Using architectures that capture hierarchical temporal dynamics (from rapid articulation to slower planning) can make models more robust to variations at specific time scales [73].
Troubleshooting Guides
Guide 1: Resolving Low Signal-to-Noise Ratio (SNR) in EEG Recordings
A low SNR is a common bottleneck for decoding inner speech and other cognitive processes [72].
Symptoms:
- Poor performance of a decoding model that works well on other datasets.
- Weak or indistinguishable event-related potentials (ERPs) in grand averages.
- High variability in features across trials or subjects.
Methodology:
Preprocessing with Advanced Filtering:
Feature Extraction Optimization:
- Move beyond manually engineered features (e.g., Power Spectral Density) to automatic feature learning. Frameworks that combine discrete wavelet transform with deep learning can adaptively extract robust time-frequency representations, enhancing cross-task generalization [75].
Algorithm Selection:
- Employ models designed for noise robustness. Spiking Neural Networks (SNNs), for instance, offer efficient computation and have shown success in tasks like emotion recognition and auditory attention decoding under low-SNR conditions [75].
Guide 2: Correcting for Artifacts to Prevent Inflated Decoding Accuracy
Artifacts can create misleadingly high decoding performance if not properly managed.
Symptoms:
- Surprisingly high decoding accuracy (>95%) in a difficult task.
- Analysis reveals the model's decisions are based on signals from non-brain regions (e.g., forehead, eyes).
- The top predictive features correspond to known artifact channels or components.
Methodology:
Systematic Preprocessing:
- Step 1: Artifact Correction. Use ICA to identify and remove components associated with blinks and eye movements. This is crucial to remove confounds without losing data [33].
- Step 2: Artifact Rejection. Follow with automated or manual inspection to reject trials with large, non-ocular artifacts (e.g., muscle spikes, electrode pops). Weigh this against the loss of trial count [33].
Validation and Control Analysis:
- Perform a "sham" decoding analysis where the labels are shuffled. A well-preprocessed dataset should yield results at chance level in this control analysis.
- Verify that the model's learned features are neurophysiologically plausible (e.g., originate from scalp locations relevant to the task).
Diagram: Workflow for troubleshooting artifact-inflated decoding accuracy.
Guide 3: Adapting to Non-Stationarity and Domain Shifts
Domain shifts across subjects or sessions are a major obstacle to robust real-world BCI applications [8] [73].
Symptoms:
- A model demonstrates high accuracy for the training subject/session but fails to generalize to new ones.
- Performance degrades significantly over time within the same subject.
Methodology:
Architectural Strategy: Multi-Scale Learning:
- Implement a Multi-Scale Decomposable Mixing (MDM) module. This architecture constructs a temporal pyramid of the input signal, processing it at multiple resolutions to capture both rapid and slow neural dynamics, which is a more invariant representation [73].
Algorithmic Strategy: Test-Time Adaptation:
- Integrate a source-free TTA method like Tent. This technique minimizes the prediction entropy on batches of unlabeled test data by updating only the model's normalization layers. This adapts the model to the new data distribution in real-time without requiring source data or costly re-training [73].
Table 2: Performance of Adaptive Algorithms Against Benchmarks
| Decoding Model | Approach to Non-Stationarity | Average Accuracy (Sample Subjects) |
|---|---|---|
| EEGNet [73] | Not Specified | ~36.83% (subj-04) |
| EEG-Conformer [73] | Not Specified | ~50.44% (subj-04) |
| DU-IN [73] | Self-supervised features | ~60.60% (subj-04) |
| MDM-Tent (Proposed) [73] | Multi-Scale Learning + Test-Time Adaptation | ~71.58% (subj-04) |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Robust Neural Signal Decoding
| Tool / Technique | Function | Relevance to Adaptive Decoding |
|---|---|---|
| Discrete Wavelet Transform (DWT) [75] [76] | Provides time-frequency analysis and denoising for non-stationary signals. | Enables automatic feature extraction and noise reduction, forming a robust input for models. |
| Spiking Neural Networks (SNNs) [75] | Bio-inspired, energy-efficient models that process data via discrete spike events. | Ideal for portable BCI devices; can be combined with attention mechanisms for high performance. |
| Domain Adaptation (DA) [8] | Transfers knowledge from a labeled source domain to a related target domain with different statistics. | Directly addresses cross-subject/session variability, enhancing decoder generalizability. |
| Test-Time Adaptation (TTA) [73] | Adapts a pre-trained model to distribution shifts during inference using unlabeled test data. | Mitigates performance decay over time without needing source data or re-training. |
| Independent Component Analysis (ICA) [33] | Identifies and separates independent source signals, such as neural activity and artifacts. | Critical preprocessing step to remove biological confounds and ensure decoding validity. |
| Multi-Scale Decomposable Mixing (MDM) [73] | Models hierarchical temporal dynamics in neural signals across different time scales. | Learns stable neural representations that are more invariant to transient noise and shifts. |
Diagram: A two-stage framework combining multi-scale feature learning with test-time adaptation.
In the field of research on adaptive decoding algorithms for non-stationary neural signals, minimizing bias is not merely a methodological preference but a fundamental requirement for scientific validity. The inherent variability of brain signals and the complexity of decoding models create multiple points where conscious and unconscious biases can affect study outcomes. This guide establishes a technical support framework for implementing two cornerstone methodologies for bias mitigation: blinding and independent review.
- Blinding (or masking) is the process of concealing information about assigned interventions or experimental conditions from one or more parties involved in the research to prevent bias from influencing the results [77] [78].
- Blinded Independent Central Review (BICR) is a specific, methodical assessment process where qualified, independent reviewers evaluate data without knowledge of treatment assignments, group allocations, or initial investigator assessments [79].
The following sections provide a structured troubleshooting guide to help you implement these practices effectively in your neural signal research.
Frequently Asked Questions (FAQs)
FAQ 1: Why is blinding critical in neural signal decoding research, even for seemingly objective outcomes? Even when using quantitative measures, bias can significantly affect results. Knowledge of group allocation can influence how data is preprocessed, how features are selected, and how model outputs are interpreted. For instance, empirical evidence shows that non-blinded versus blinded outcome assessors can generate exaggerated effect sizes—by an average of 36% for binary outcomes and 68% for measurement scale outcomes [77]. In studies involving subjective outcomes or those requiring interpretation (like classifying cognitive states from EEG), the risk is even higher [77] [80].
FAQ 2: Who should be blinded in an experiment involving adaptive decoding algorithms? Blinding is a graded continuum, not an all-or-nothing phenomenon. The following groups should be considered for blinding, where feasible [77] [78]:
- Participants: To prevent biased expectations or altered behavior.
- Intervention Administrators: Those applying a stimulus or therapy concurrent with signal recording.
- Data Collectors and Pre-processors: Those handling raw neural data.
- Outcome Assessors: Personnel who evaluate the final decoded output or model performance.
- Statisticians and Data Analysts: Those performing the final analysis should be blinded to group labels (e.g., using non-identifying codes like "Group A" and "Group B") to prevent selective reporting [78].
FAQ 3: What is the difference between allocation concealment and blinding? A common point of confusion, these are distinct concepts. Allocation concealment occurs before assignment and ensures researchers and participants are unaware of the upcoming group assignment until the moment of randomization, preventing selection bias. Blinding occurs after assignment and refers to concealing the group allocation from the parties listed above throughout the trial to prevent performance and detection bias [77].
FAQ 4: Our study is "open-label" (e.g., comparing an invasive stimulus to a control). How can we still minimize bias? When full blinding of participants and interventionists is impossible, you can still implement blinded evaluation. This involves ensuring that all subsequent parties, especially data pre-processors, feature engineers, and outcome assessors, are blinded to the group allocation. A real-world study on diabetic foot infections demonstrated that unblinded site investigators tended to exaggerate treatment efficacy compared to blinded central reviewers, with a 27% discrepancy in evaluations [80].
FAQ 5: What are the practical challenges of maintaining a blind, and how can we test its integrity? Challenges include accidental unblinding through side effects, data patterns, or logistical errors. To manage this:
- Plan for Blinding Maintenance: Use centralized procedures for data handling and pre-processing [77] [81].
- Test Blinding Success: In some studies, you can ask blinded participants and researchers to guess the group assignment at the end of the study. The success of blinding can be quantified using a blinding index, helping to assess the potential for bias [81].
Troubleshooting Guides
Issue: Implementing Blinding in Complex or Open-Label Study Designs
Problem: It is not always feasible to blind all parties, especially in studies with distinct interventions or when using participant-specific adaptive models.
Solution: Adopt a partial blinding strategy and implement a Blinded Independent Central Review for the analysis pipeline.
Detailed Protocol:
- Role Separation and Delegation: Clearly define and separate roles within your research team. Authorize different personnel for tasks such as:
- Blinded Data Handling Workflow:
- After data collection, the unblinded team member should anonymize the datasets by replacing group labels with non-identifiable codes (e.g., Dataset001, Dataset002).
- This anonymized dataset is then transferred to the blinded data pre-processing and analysis team.
- The blinded team performs all steps—filtering, artifact removal, feature extraction (e.g., using Permutation Conditional Mutual Information Common Space Pattern), and model training (e.g., using an optimized back propagation neural network)—using only the anonymous codes [82].
- Final Analysis and Unblinding: The blinded statistician performs the final comparative analysis on the model outputs (e.g., accuracy, F1-score) using the anonymous codes. The unblinding of what "Group A" and "Group B" represent should only occur after the analysis plan is finalized and the results are locked [78].
Issue: Managing Discordance Between Local and Central Assessments
Problem: In a multi-center study or when comparing a site's initial assessment to a central blinded review, discrepancies can arise, complicating the interpretation of results.
Solution: Implement a pre-specified adjudication process.
Detailed Protocol:
- Pre-define Assessment Criteria: In your study protocol, establish clear, objective criteria for evaluating the primary outcome (e.g., decoding accuracy thresholds, specific patterns in the neural signal). This is crucial for non-stationary signals where interpretation may vary [79].
- Independent Parallel Review: Have at least two qualified, blinded reviewers assess the outcomes independently.
- Adjudication Pathway: If the two primary reviewers disagree, a third, senior blinded adjudicator should review the data and make a final determination based on the pre-defined criteria [79].
- Documentation and Analysis: Document all initial and adjudicated outcomes. During manuscript preparation, report both the initial and centrally-adjudicated results, if applicable, and perform sensitivity analyses to show the robustness of your findings [79].
Experimental Protocols & Data Presentation
Protocol for a Blinded Analysis of an Adaptive Decoding Algorithm
This protocol outlines the key steps for evaluating a new adaptive decoding algorithm, like the Adaptive Decoding Algorithm (ADA), while maintaining blinding to minimize bias [83].
Quantitative Impact of Unblinded Assessment on Effect Size Table: Empirical evidence demonstrating the exaggeration of effects in non-blinded studies [77]
| Type of Bias Mitigated | Outcome Type | Average Exaggeration of Effect Size |
|---|---|---|
| Participant Blinding | Participant-reported outcomes | 0.56 standard deviations |
| Outcome Assessor Blinding | Binary Outcomes | 36% (exaggerated odds ratio) |
| Outcome Assessor Blinding | Measurement Scale Outcomes | 68% (exaggerated effect size) |
| Outcome Assessor Blinding | Time-to-Event Outcomes | 27% (exaggerated hazard ratio) |
Protocol for Implementing Blinded Independent Central Review (BICR) in a Multi-Center Trial
For large-scale validation studies, a formal BICR process ensures consistency and objectivity across sites [79].
Discordance Analysis Between Site and Central Review Table: Example from a clinical study showing the impact of blinded review on outcome assessment [80]
| Subject Group | Non-Blinded Site Evaluation (IDSA Grade) | Blinded Central Review (IDSA Grade) | Number of Cases | Interpretation of Discrepancy |
|---|---|---|---|---|
| Experimental Group | 1 (Mild) | 2 (Moderate) | 3 | Potential overestimation of treatment benefit by site investigator. |
| Control Group | 2 (Moderate) | 1 (Mild) | 3 | Potential underestimation of treatment effect in control group. |
| Total Discrepancies | --- | --- | 6/22 (27%) | High rate of discordance necessitates blinded review. |
The Scientist's Toolkit
Table: Essential methodological "reagents" for minimizing bias in neural decoding research
| Research Reagent Solution | Function in Experiment | Key Considerations |
|---|---|---|
| Anonymized Data Pipeline | Replaces group labels with non-identifiable codes before analysis, blinding data processors and model trainers. | Ensure the code-key is held securely by an independent data manager and not accessible to the analysis team. |
| Blinded Independent Central Review (BICR) | Uses independent, blinded experts to adjudicate the primary study outcomes (e.g., decoding success/failure). | Critical for multi-center trials and subjective outcomes. Requires pre-specified adjudication rules [79]. |
| Standard Operating Procedures (SOPs) | Documents exact procedures for data handling, pre-processing, and analysis to ensure consistency and reduce operator-dependent variability. | Especially important for managing non-stationary signals and ensuring all team members follow the same blinded protocol [80]. |
| Sham Procedure / Placebo Intervention | Serves as a control that mimics the active intervention without its critical component, blinding participants to their group assignment. | In neural studies, this could be a sham stimulation or a control task designed to be perceptually similar to the experimental task [77]. |
| Active Placebo | A placebo designed to mimic the minor side effects or sensations of the active intervention, thereby strengthening the blind. | Helps prevent participants from guessing their assignment based on peripheral sensations, thus protecting the blind [77]. |
Benchmarking Performance: Validation Frameworks and Comparative Analysis
Frequently Asked Questions
1. When should I use Accuracy versus the F1-Score to validate my neural decoder?
Answer: The choice between Accuracy and F1-Score depends heavily on the class balance of your neural data and the relative importance of different error types in your experimental goals [84].
- Use Accuracy when your test dataset is balanced and the cost of false positives and false negatives is roughly equal [85] [84]. It provides an intuitive measure of overall correctness. However, it can be highly misleading for imbalanced datasets, which are common in BCI applications, as a model can achieve high accuracy by simply always predicting the majority class [86] [87].
- Use the F1-Score when your data is imbalanced or when you care more about the correct identification of the positive class (e.g., detecting a specific movement intention or cognitive state) [88] [84]. The F1-Score is the harmonic mean of Precision and Recall, providing a single metric that balances the concern of false positives (Precision) and false negatives (Recall) [85]. This makes it a robust metric for many neural decoding tasks.
Table: Guidance for Choosing Between Accuracy and F1-Score
| Situation | Recommended Metric | Reasoning |
|---|---|---|
| Balanced classes, equal cost of FP/FN | Accuracy | Gives a good overview of overall performance [84]. |
| Imbalanced classes | F1-Score | Prevents inflated performance estimates from predicting the majority class [88] [86]. |
| High cost of False Negatives (e.g., disease detection) | F1-Score (or F2-Score) | Recall, a component of F1, prioritizes finding all positive instances [86] [84]. |
| High cost of False Positives | Precision | Prioritizes the correctness of positive predictions [87]. |
| Initial model benchmarking | Multiple Metrics | Always evaluate with a suite of metrics (Accuracy, F1, Precision, Recall) for a complete picture [87]. |
2. Why does my model have a high F1-Score but low Accuracy, and is this a problem?
Answer: A high F1-Score coupled with low Accuracy is a classic indicator of a highly imbalanced dataset [88]. This is not necessarily a problem with your model, but rather a reflection of the data and what the metrics are measuring.
- Explanation: Accuracy considers both true positives (TP) and true negatives (TN). The F1-Score, however, is calculated from Precision and Recall, which focus primarily on the positive class and do not directly consider true negatives [85]. In an imbalanced scenario where the negative class is the majority, a model can be very good at finding the positive class (high F1) but perform poorly on the abundant negative class (e.g., many false positives), leading to low overall accuracy [88].
- Actionable Steps:
- Verify Class Balance: Check the distribution of classes in your test set.
- Examine the Confusion Matrix: This will show you the exact counts of true positives, false positives, true negatives, and false negatives, providing clarity on where the model is failing [86] [85].
- Re-evaluate Your Objective: If the positive class is your primary interest (e.g., detecting the onset of an epileptic seizure), a high F1-Score might be exactly what you want, and the low accuracy can be disregarded as misleading [84].
3. My neural decoder's performance drops over time. Is this a metric problem or a signal problem?
Answer: This is most likely a signal problem related to the inherent non-stationarity of neural signals, not a flaw in the metrics themselves [8] [89]. Neural recordings, especially in chronic implants, are dynamic and change over time due to factors like electrode drift, immune response, and neural plasticity [89].
- Root Cause: The joint probability distribution of the neural signals and the decoded output, ( P_s(x, y) ), changes between the training (source) domain and the deployment (target) domain over time [8]. This violates the common assumption in machine learning that training and test data are independently and identically distributed (i.i.d.).
- Solutions & mitigation strategies:
- Domain Adaptation (DA): Employ DA techniques to reduce the distributional shift. This can be instance-based (re-weighting training samples), feature-based (transforming features to a common space), or model-based (fine-tuning a pre-trained model) [8].
- Adaptive Decoding Algorithms: Use algorithms specifically designed for temporal variability. For example, the Adaptive Decoding Algorithm (ADA) estimates the most informative temporal window for each trial, improving robustness to timing jitters in cognitive tasks [83].
- Retraining Strategy: Move from a static training scheme (model trained once) to a retrained scheme where the decoder is periodically updated with new calibration data. Research has shown that decoders like Recurrent Neural Networks (RNNs) under a retrained scheme maintain higher performance under simulated non-stationarity [89].
4. What is a comprehensive experimental protocol for validating a new adaptive decoder?
Answer: A robust validation protocol must account for non-stationarity and ensure results are statistically sound. Below is a detailed methodology.
Table: Key Phases for Validating an Adaptive Decoder
| Phase | Key Activities | Outputs/Deliverables |
|---|---|---|
| 1. Experimental Design | - Define decoding task (e.g., classification, regression).- Plan for longitudinal data collection across multiple sessions.- Deliberately introduce controlled variations (e.g., different days, subjects). | Experimental protocol. |
| 2. Data Preparation | - Split data into training, validation, and test sets by session or subject (not randomly) to simulate real-world use.- Apply preprocessing: filtering, artifact removal, and feature extraction [8]. | Preprocessed datasets for each subject/session. |
| 3. Model Training & Tuning | - Train multiple decoder types (e.g., OLE, Kalman Filter, RNN) [89] [90].- Use k-fold cross-validation within the training data only to tune hyperparameters and prevent overfitting [87] [90].- Implement Domain Adaptation techniques if applicable [8]. | A set of tuned decoder models. |
| 4. Model Evaluation | - Evaluate each model on the held-out test sessions.- Calculate a suite of metrics: Accuracy, Precision, Recall, F1-Score, ROC-AUC [85] [84].- For continuous outputs, use Bit Rate or other information-theoretic measures. | A table of performance metrics for all models and sessions. |
| 5. Statistical Testing | - Use a paired statistical test (e.g., Wilcoxon signed-rank test) to compare metric distributions across sessions or against a baseline model [85].- Correct for multiple comparisons if testing many models. | p-values, confidence intervals. |
| 6. Reporting | - Report all metrics, not just one.- Clearly state the cross-validation and testing procedure.- Discuss performance in the context of non-stationarity. | Final validation report. |
The following workflow diagram illustrates this protocol:
Diagram 1: Workflow for validating an adaptive neural decoder.
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Components for a Neural Decoding Research Pipeline
| Item / Solution | Function / Role in the Experiment |
|---|---|
| Electrophysiology Recording System | Acquires raw neural signals (e.g., EEG, ECoG, spike data) from the brain [8]. |
| Preprocessing Pipeline (Custom Scripts) | Performs essential steps like downsampling, filtering (artifact removal), and normalization to clean the raw signals [8]. |
| Feature Extraction Algorithms | Transforms preprocessed signals into informative features (e.g., time-domain, frequency-domain, time-frequency features) [8]. |
| Domain Adaptation (DA) Library | Provides algorithms (e.g., instance-based, feature-based, model-based DA) to mitigate performance decay from non-stationarity [8]. |
| Machine Learning Library (e.g., PyTorch, scikit-learn) | Offers implementations of various decoders, from traditional filters (Kalman) to modern neural networks (RNNs), and evaluation metrics [91] [90]. |
| Statistical Analysis Software | Used to perform rigorous statistical tests (e.g., Wilcoxon signed-rank test) to compare decoder performance across conditions [85]. |
Troubleshooting Guide: Common Problems and Solutions
Table: Diagnostic and Resolution Steps for Common Issues
| Problem | Possible Causes | Diagnostic Steps | Resolution Steps |
|---|---|---|---|
| Poor Accuracy on Imbalanced Data | Metric is biased towards the majority class [88] [86]. | Check class distribution in the dataset. Analyze the confusion matrix. | Switch to F1-Score, Precision, or Recall. Use resampling techniques or cost-sensitive learning [84]. |
| Performance Degradation Over Time | Non-stationarity of neural signals [8] [89]. | Track performance metrics across different sessions or days. | Implement Domain Adaptation [8]. Use adaptive algorithms like ADA [83]. Adopt a retraining strategy [89]. |
| Inconsistent Results Across Validation Folds | High variance in model performance; possible overfitting [87]. | Examine the standard deviation of metrics across k-fold cross-validation. | Increase training data. Tune model hyperparameters (e.g., increase regularization). Use ensemble methods [87] [90]. |
| Model Fails to Generalize to New Subjects | High inter-subject variability; subject-specific features not learned [8]. | Evaluate model performance on a per-subject basis. | Apply feature-based Domain Adaptation to align distributions [8]. Train subject-specific models or use a multi-subject pre-training approach. |
In neural signal processing, a fundamental challenge is the non-stationary nature of neural activity, where statistical properties like mean firing rates and neural preferred directions change over time [2] [89]. This phenomenon poses significant problems for traditional fixed-algorithm decoders trained on initial data periods, as their performance degrades when the relationship between neural activity and behavior evolves [2]. Non-stationarity arises from various factors including recording degradation from glial scarring, neuronal property variance as subjects adapt to tasks, and physiological changes like electrode drift [89] [92].
Adaptive decoding algorithms address this limitation by continuously updating their parameters to track these dynamic changes [2]. This technical support document provides a comparative analysis and practical guidance for researchers selecting, implementing, and troubleshooting these approaches in experimental settings, particularly within the context of intracortical Brain-Computer Interfaces (iBCI) and motor brain-machine interfaces (BMI) [89].
Algorithm Performance & Selection Guide
Quantitative Performance Comparison
Table 1: Performance comparison of decoders under non-stationary conditions
| Decoder Type | Static Scheme Performance | Retrained Scheme Performance | Best Suited Non-Stationarity Type |
|---|---|---|---|
| Optimal Linear Estimation (OLE) | Performance drops significantly with recording degradation and neural variation [89]. | Improved performance, but still influenced by serious recording degradation [89]. | Stable signals with minimal recording degradation [89]. |
| Kalman Filter (KF) | Performance drops with both recording degradation and neural variation; outperforms OLE in some sequential tasks [89]. | Maintains high performance when changes are limited to Preferred Directions (PDs) [89]. | Scenarios with gradual neuronal property variance (e.g., PD changes) [2] [89]. |
| Recurrent Neural Network (RNN) | More robust than OLE and KF under small recording degradation and neural variation [89]. | Shows consistent better performance under small recording degradation; significantly outperforms OLE and KF [89]. | Complex non-stationarities and sequential decoding tasks [89]. |
| Adaptive Linear Regression | Becomes inappropriate over time as neural signals evolve [2]. | N/A (The algorithm itself is adaptive) [2]. | Scenarios requiring efficient, real-time updates of linear mapping [2]. |
| Adaptive Kalman Filter | N/A (The algorithm itself is adaptive) [2]. | N/A (The algorithm itself is adaptive) | Online situations with non-stationary neural activity; more accurate and efficient than non-adaptive versions [2]. |
Performance Under Specific Non-Stationarity Types
Table 2: Decoder performance across different non-stationarity metrics
| Non-Stationarity Metric | Effect on Neural Signal | OLE Performance | KF Performance | RNN Performance |
|---|---|---|---|---|
| Mean Firing Rate (MFR) Decrease [89] | Simulates recording degradation; reduces overall neural activity strength [89]. | Performance drops with decreasing MFR [89]. | Performance drops with decreasing MFR [89]. | Robust to small decreases; performance drops with serious degradation [89]. |
| Number of Isolated Units (NIU) Loss [89] | Simulates recording degradation; reduces the number of detectable neurons [89]. | Performance drops with NIU loss [89]. | Performance drops with NIU loss [89]. | Robust to small losses; performance drops with significant loss [89]. |
| Neural Preferred Directions (PDs) Change [89] | Simulates neuronal property variance; alters the tuning of neurons to movement [89]. | Performance drops with PD changes under static scheme [89]. | Maintains performance with PD changes under retrained scheme [89]. | Maintains performance with PD changes under retrained scheme [89]. |
Experimental Protocols & Methodologies
Implementing an Adaptive Kalman Filter for Motor Decoding
The Adaptive Kalman Filter enhances the standard Kalman filter by recursively updating the model parameters (state transition and observation matrices) to track the dynamic relationship between neural activity and kinematics [2].
Core Methodology:
- Neural Signal Preprocessing: Extract firing rates from spike-sorted single-unit activity by counting spikes in 50-100ms bins [2].
- Kinematic Data Alignment: Align neural activity with kinematic parameters (e.g., hand position, velocity) using a time lag (typically ~100ms) to account for neural processing and signal transmission delays [2].
- Recursive Parameter Update: Implement a recursive maximum likelihood estimation or a similar approach to update the encoding model parameters as new batches of neural and kinematic data arrive. This can be done efficiently without reprocessing the entire dataset [2].
Figure 1: Adaptive Kalman Filter Workflow
Benchmarking Decoder Performance with Simulated Non-Stationarity
A 2D-cursor simulation study allows for controlled evaluation by introducing specific non-stationarities separately [89].
Core Methodology:
- Data Generation with Population Vector (PV) Model: Generate synthetic spike data using a PV model and real kinematic data from a center-out or random target pursuit task. Simulate spike counts using a Poisson process [89].
- Introduce Controlled Non-Stationarity: Systematically vary one of the three key metrics across simulated sessions:
- Decoder Training & Evaluation: Train decoders (OLE, KF, RNN) using both static (trained on first session only) and retrained (retrained each session) schemes. Evaluate decoding accuracy against the known ground-truth kinematics [89].
Figure 2: Simulation-Based Benchmarking
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key resources for neural decoding research
| Item Name | Specification / Example | Primary Function in Research |
|---|---|---|
| Silicon Microelectrode Arrays | 100 platinized-tip electrodes (e.g., Cyberkinetics Inc.) [2]. | Chronic implantation in motor cortex for long-term recording of single- and multi-unit activity [2]. |
| Neural Signal Acquisition System | Cerebus system (e.g., Cyberkinetics Neurotechnology Systems) [2]. | Filters, amplifies, and digitally records raw neural signals at high sampling rates (e.g., 30 kHz) [2]. |
| Spike Sorting Software | Offline Sorter (Plexon Inc.) [2]. | Isolates action potentials from individual neurons from multi-unit recordings based on waveform shape [2] [92]. |
| Behavioral Task & Robot | KINARM system for a Random Target Pursuit (RTP) task [2]. | Presents visual targets and precisely measures the subject's actual joint angles and hand kinematics (ground truth) [2]. |
| Neural Signal Simulator | Population Vector (PV) model with Poisson process for spike generation [89]. | Generates synthetic neural data with controlled non-stationarities for controlled algorithm testing and validation [89]. |
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: My decoder performance drops significantly a few days after calibration. Should I switch to a different decoder type? The problem may not be the decoder type but its training scheme. A decoder using a static scheme (trained once) will inevitably degrade over time due to neural non-stationarity [2] [89]. Before switching decoders, try implementing a retrained scheme where the decoder is regularly updated with new calibration data [89]. If continuous recalibration is impractical, an adaptive algorithm (like the Adaptive Kalman Filter) that updates itself in real-time is strongly recommended [2].
Q2: How do I know if my neural data is non-stationary, and what type of non-stationarity I am dealing with? Monitor these key metrics over sessions: a steady decrease in Mean Firing Rates (MFR) or the Number of Isolated Units (NIU) indicates recording degradation [89]. A shift in the Preferred Directions (PDs) of your neurons, calculated through a tuning curve analysis, indicates neuronal property variance [89]. Observing consistent changes in these metrics confirms non-stationarity.
Q3: I am using an adaptive algorithm, but it seems slow to converge or is unstable. What is the critical parameter to check? The learning rate is the most critical parameter. It creates a fundamental trade-off: a rate that is too high causes instability and large steady-state error, while a rate that is too low leads to slow convergence [93]. Use a principled calibration algorithm to select a learning rate that balances convergence speed with steady-state error based on your desired performance [93].
Q4: When should I choose an RNN over a classic Kalman Filter? An RNN is a superior choice when decoding complex, sequential neural patterns and when you have sufficient computational resources and data to train it. It generally shows better robustness to various types of non-stationarity compared to OLE and KF [89]. However, for many real-time BMI applications where simplicity and efficiency are key, the Adaptive Kalman Filter remains a highly effective and preferred option [2].
Common Problem-Solving Guide
Table 4: Troubleshooting common decoder performance issues
| Problem | Potential Causes | Suggested Solutions |
|---|---|---|
| Gradual performance decay over days/weeks. | Neural non-stationarity due to recording degradation or neural adaptation [89]. | Switch from a static to a retrained or adaptive training scheme [2] [89]. |
| Sudden, sharp drop in performance. | Failure of recording hardware (e.g., electrode breakage); sudden large shift in neural population [89]. | Check impedance and integrity of recording system. Re-initialize decoder with a new calibration session. |
| Decoder is slow to respond to intended movements. | Incorrect time lag alignment between neural activity and kinematics [2]. | Re-estimate the optimal latency (typically ~100ms) between neural firing and behavior [2]. |
| Poor performance from the first session. | Ineffective spike sorting; insufficient or poor-quality training data; incorrect decoder model assumptions [92]. | Revisit spike sorting quality; collect more robust calibration data; verify the decoder's encoding model matches your neural activity's properties. |
Frequently Asked Questions (FAQs)
Q1: What is the primary performance bottleneck when deploying adaptive decoders in a real-time clinical Brain-Machine Interface (BMI) system?
A1: The primary bottleneck is often the computational complexity and latency of the adaptation algorithm itself. While adaptive decoders like Unsupervised Adaptation methods [94] or Adaptive Learned Belief Propagation [95] can significantly improve accuracy, their real-time execution requires substantial processing power. For clinical readiness, the algorithm must complete its adaptation and decoding within a strict time budget (e.g., a few tens of milliseconds) to provide seamless feedback to the user. High complexity can also lead to increased power consumption, which is a critical concern for fully implantable devices.
Q2: Our decoder's performance degrades significantly a few weeks after initial calibration. What are the most common causes of this non-stationarity in neural signals?
A2: Performance degradation is typically caused by the inherent non-stationarity of neural recordings [94] [96]. Key factors include:
- Biological Changes: The immune response to the implanted electrode, leading to glial scarring, which changes the signal quality and properties of recorded neurons over time [97].
- Neuronal Adaptation: The user's brain itself adapts and learns during BMI control, causing the relationship between neural activity and intended movement to evolve. This is a natural part of neuroplasticity but poses a challenge for static decoders [94].
- Electrode Instability: Micromotions of the electrode array within the brain tissue can change the population of neurons being recorded from [94].
Q3: Can we use signals other than single-neuron spikes to make our adaptive decoder more robust for long-term use?
A3: Yes, incorporating Local Field Potentials (LFPs) is a highly promising strategy. LFPs are lower-frequency signals that are more stable over long periods than single-unit spikes. Research has demonstrated that adaptive decoders can be driven by LFP signals alone, with periodic adaptation improving offline decoding accuracy by 5% to 50% [97]. Using a combination of spikes and LFPs can provide redundancy and enhance overall system robustness.
Q4: What is a key difference between "supervised" and "unsupervised" adaptive decoding, and why does it matter for clinical use?
A4: The key difference lies in the need for recalibration data where the user's intent is known.
- Supervised Adaptation requires periodic, interruptive calibration sessions where the user is instructed to perform specific movements so the decoder can learn the new neural mapping. This burdens the user and interrupts autonomous device use [94].
- Unsupervised Adaptation continuously adjusts decoder parameters during normal, autonomous use without knowing the user's precise movement intentions. This is achieved by leveraging internal cost functions or consistency metrics, drastically reducing the burden on the user and making the system more practical for daily life [94].
Q5: How can we quantitatively assess the benefit-risk ratio of implementing a new, more complex adaptive decoder?
A5: Assessing the benefit-risk ratio requires measuring both the performance gains and the associated costs. The following table summarizes key quantitative metrics for this assessment:
Table 1: Quantitative Metrics for Benefit-Risk Assessment of Adaptive Decoders
| Assessment Dimension | Benefit Metrics (To Maximize) | Risk/Burden Metrics (To Minimize) |
|---|---|---|
| Performance | - Bit Error Rate (BER) reduction [95]- Improvement in target acquisition accuracy (%) [94]- Increase in information transfer rate (bits/sec) | - Decoding latency (milliseconds)- Performance variability across sessions |
| Clinical Burden | - Reduction in required supervised recalibration sessions per week- Increase in stable operation time (days/weeks) | - Computational complexity (FLOPS)- Power consumption increase (mW) |
| Technical Robustness | - Stability across signal non-stationarities [94] | - Sensitivity to hyperparameter tuning- Generalization error on unseen data |
Troubleshooting Guides
Issue: High Error Floor in Decoding Performance
Problem: Your adaptive decoder's performance plateaus at a high error rate and fails to improve further, despite algorithm adjustments.
Possible Causes and Solutions:
- Cause: Inadequate Adaptation to Signal Drift
- Solution: Implement a more frequent or stronger adaptation rule. For belief propagation-based decoders, investigate Adaptive Learned Belief Propagation (A-BP) where weights are dynamically determined for each received word, which has been shown to achieve Bit Error Rates (BER) up to an order of magnitude lower than static decoders [95].
- Actionable Protocol:
- Train a secondary, small neural network to predict optimal weights for the primary decoder based on real-time input features (the two-stage decoder method) [95].
- Run multiple decoders with different weight sets in parallel and select the output with the highest confidence (e.g., lowest syndrome weight) [95].
- Cause: Overfitting to Short-Term Noise Instead of Long-Term Drift
- Solution: Introduce a "forgetting factor" or use a longer time window for the adaptation algorithm to learn from. This helps the decoder distinguish between momentary noise and a genuine, persistent shift in the neural tuning properties.
Issue: Unacceptable Computational Latency in Real-Time Operation
Problem: The adaptive decoding algorithm takes too long to process, causing lag in the BMI's response and degrading user experience.
Possible Causes and Solutions:
- Cause: Complex Model-Based Neural Decoder Architecture
- Solution: Optimize the decoder architecture. Weighted Belief Propagation (WBP), which unfolds a Tanner graph into a recurrent network, is a model-based approach that typically has fewer parameters and lower computational complexity than fully model-agnostic deep learning decoders, making it more suitable for real-time applications [95].
- Actionable Protocol:
- Implement parameter sharing across layers of the unrolled WBP network to reduce the total number of parameters that need to be stored and updated [95].
- For algorithms like those using Graph Neural Networks (GNNs), analyze the complexity, which is often
O(E * F^2)(where E is edges and F is feature dimensions). Focus on optimizing the feature dimensionFas a primary lever to reduce latency [98].
- Cause: Inefficient Hardware/Software Implementation
- Solution: Profile the code to identify computational bottlenecks. For deployment, consider using specialized hardware accelerators (FPGAs, ASICs) designed for low-latency inference of neural network models [98].
Issue: Failure to Generalize Across Users or Sessions
Problem: The adaptive decoder performs well on the initial user or dataset it was trained on but fails to maintain performance for new users or even the same user in a different session.
Possible Causes and Solutions:
- Cause: Lack of Robustness to Inter-User Variability
- Solution: Employ transfer learning techniques. Start with a decoder pre-trained on a large population dataset and then perform a rapid, user-specific calibration to fine-tune the model. This balances generalizability with personalization.
- Actionable Protocol:
- Use a base model trained on a diverse set of neural signals from multiple subjects.
- During a new user's initial calibration session, run a few trials to collect user-specific data and update only the final layer (or a small subset) of the decoder's parameters.
- Cause: Ignoring Multi-Modal Signal Sources
- Solution: Enhance the feature set. Fuse information from multiple neural signal types to create a more robust representation. For instance, combine the high-temporal resolution of spikes with the long-term stability of LFPs [97]. Advanced algorithms like Joint Independent Component Analysis (jICA) or Bayesian Data Fusion can be used for this integration [99].
Experimental Protocols for Key Cited Studies
Objective: To enable a BMI decoder to adapt autonomously during use without knowledge of the user's movement intentions, countering performance degradation from neural non-stationarities.
Workflow:
Methodology:
- Initialization: Perform a one-time, supervised calibration to establish a baseline decoder (e.g., using linear regression or a Kalman filter) [94] [96].
- Autonomous Operation: Switch to autonomous mode where the user freely controls the effector (e.g., cursor, robotic arm).
- Data Collection: Continuously record neural activity and the decoder's output (e.g., predicted kinematics) during operation.
- Cost Function Evaluation: Compute an unsupervised cost function derived from the neural data itself. This function is designed to guide learning without ground-truth movement labels, for instance, by promoting consistency with a model of population activity or leveraging optimal feedback control principles [94].
- Parameter Update: Use the output of the cost function to guide a learning algorithm (e.g., stochastic gradient descent) to update the decoder's parameters.
- Iteration: Continuously repeat steps 3-5, creating a closed-loop system where the decoder adapts in real-time.
Objective: To assess and improve the performance of a BMI decoder that uses the more stable LFP signals through periodic unsupervised adaptation.
Workflow:
Methodology:
- Signal Acquisition: Implant a microelectrode array in the relevant brain area (e.g., motor cortex) of a non-human primate model. Configure the system to record LFP signals [97].
- Closed-Loop Setup: Set up a closed-loop BMI where the primate can control a robotic platform using decoded brain signals.
- Feature Extraction: In real-time, extract features from the LFPs, such as spectral power in specific frequency bands.
- Baseline Decoding: Use a simple decoder (e.g., linear regression) to map LFP features to movement commands.
- Offline Analysis for Adaptation:
- Record LFP data and concurrent decoder outputs during closed-loop control.
- Periodically re-analyze the data to perform "signal and channel selection." This involves identifying which LFP features and electrode channels are most informative for decoding at that point in time.
- Use this analysis to adapt the decoder's feature pre-processing and model parameters.
- Validation: Compare the decoding accuracy before and after adaptation to quantify improvement (reported 5-50% in offline analysis) [97].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for Adaptive Decoding Research
| Item | Function/Explanation | Example Use Case |
|---|---|---|
| Intracortical Microelectrode Array | A multi-electrode implant for recording single-unit (spike) and local field potential (LFP) signals from the brain. The foundation of signal acquisition. | Chronic implantation in motor cortex for closed-loop BMI control studies [97]. |
| Neural Signal Processor | Hardware and software for real-time amplification, filtering, and processing of raw neural signals. | Converting raw neural data into spike times and LFP band power for decoding. |
| Optimal Feedback Control (OFC) Model | A computational model of motor control used to derive internal cost functions for unsupervised adaptation. | Simulating a realistic BMI user to test unsupervised adaptation algorithms [94]. |
| Graph Neural Network (GNN) Framework | A deep learning library (e.g., PyTorch Geometric) for implementing GNN-based decoders like ABPGNN [98] or Adaptive WBP [95]. | Dynamically adapting message-passing rules in a decoder graph to improve performance and reduce error floors. |
| Bayesian Data Fusion Algorithm | A statistical method for integrating data from different modalities (e.g., fMEG and EEG) to create a more robust brain map [99]. | Improving the spatial and temporal resolution of source-localized neural features used for decoding. |
The Emergence of Brain Foundation Models (BFMs) as a Validation Benchmark
Frequently Asked Questions (FAQs) on BFMs and Neural Decoding
FAQ 1: What exactly is a Brain Foundation Model (BFM), and how does it differ from traditional neural decoders?
A Brain Foundation Model (BFM) is a foundational model built using deep learning and neural network technologies that is pre-trained on large-scale neural data, designed to decode or simulate brain activity [100]. Unlike traditional decoders like Optimal Linear Estimation (OLE) or Kalman Filters (KF), which are often trained for a single, specific task and struggle with distribution shifts, BFMs learn universal representations from vast datasets. This enables them to generalize across multiple scenarios, tasks, and neural signal modalities (e.g., EEG, fMRI) with minimal or no additional training (zero-shot or few-shot learning) [100]. Their architecture is specifically designed to handle the spatiotemporal complexity and low signal-to-noise ratio inherent to neural data.
FAQ 2: Why are BFMs particularly suited for validating algorithms designed for non-stationary neural signals?
Non-stationarity—where the statistical properties of neural signals change over time due to factors like recording degradation or neuronal adaptation—is a core challenge in chronic brain-computer interfaces (BCIs) [89]. BFMs directly address this through their foundational design principles. Their large-scale pre-training exposes them to a wide diversity of neural patterns and variations, inherently building robustness to distributional shifts [100]. Furthermore, their architecture often supports efficient fine-tuning or adapter modules (like Hypergraph Dynamic Adapters), allowing for rapid, low-resource adaptation to new subjects or sessions, which is crucial for compensating for non-stationarity in real-world applications [101] [8].
FAQ 3: What are the primary categories of BFMs, and how do I choose one for my research?
BFMs can be broadly classified into three categories based on their application paradigm [100]:
- Pretrained Only: These models are deployed directly after pre-training for general brain activity analysis, leveraging in-context learning during inference.
- Pretrained with Fine-tuning: These models are first pre-trained on large-scale data and then fine-tuned on specific downstream tasks (e.g., disease diagnosis, motor imagery classification). This is the most common approach for task-specific applications.
- Pretrained with Interpretability for Brain Discovery: These models combine pre-training with interpretability techniques to not only perform tasks but also to simulate and explore key biological mechanisms of brain function. Your choice depends on the task: use "Pretrained Only" for exploratory analysis, "Pretrained with Fine-tuning" for a specific classification/decoding task, and "Interpretability" models for hypothesis-driven brain discovery.
FAQ 4: My decoder performance drops significantly across sessions. Could non-stationarity be the cause, and how can a BFM help?
Yes, performance drops across sessions are a classic symptom of neural non-stationarity. This can be caused by a decrease in the Mean Firing Rate (MFR) or Number of Isolated Units (NIU) due to recording device degradation, or by a shift in neuronal tuning properties like Preferred Directions (PDs) [89]. A BFM can help in two key ways. First, it can serve as a robust feature extractor that is less sensitive to these shifts. Second, its framework allows for the integration of Domain Adaptation (DA) techniques. DA techniques—such as feature space transformation or fine-tuning the pre-trained model with a small amount of new data—can explicitly minimize the distributional differences between your old and new sessions, thereby restoring decoder performance [8].
Troubleshooting Guide: Common BFM Experimental Challenges
Table 1: Troubleshooting Common Issues in BFM and Neural Decoding Experiments
| Problem Symptom | Potential Cause | Diagnostic Steps | Solution & Validation Approach |
|---|---|---|---|
| Poor Generalization to New Subjects | High inter-subject variability; domain shift between training and test data. | 1. Check subject demographic and experimental condition mismatches.2. Measure distribution distance (e.g., MMD) between source and target feature domains [8]. | Apply Feature-based Domain Adaptation. Use algorithms like CORrelation Alignment (CORAL) to transform feature spaces, or employ Model-based DA by fine-tuning the BFM on a small dataset from the new subject [8]. |
| Performance Degradation Over Time (Within Subject) | Non-stationarity of neural signals: decreasing MFR, NIU, or shifting PDs [89]. | 1. Track MFR and NIU metrics across sessions.2. Analyze if neuronal tuning properties (e.g., PDs) have changed. | Implement a retraining scheme. Periodically retrain or fine-tune the decoder on the most recent data. Use a BFM with a dynamic adapter (e.g., HyDA) for patient-specific adaptation [89] [101]. |
| Low Decoding Accuracy Despite Large Pre-training | Task or modality mismatch; insufficient fine-tuning; suboptimal model architecture for the specific neural signal. | 1. Verify the BFM's pre-training modalities (EEG, fMRI) match your data.2. Evaluate if the decoder (OLE, KF, RNN) is suitable for your task's dynamics [89]. | Fine-tune the BFM on your specific task. Consider switching to a more powerful decoder like an RNN, which has shown better performance under non-stationarity compared to OLE and KF [89]. |
| Overfitting on Small Fine-tuning Datasets | The BFM has a high number of parameters, and the target domain dataset is too small. | Monitor the sharp gap between training and validation accuracy during fine-tuning. | Use lightweight adapter modules like Hypergraph Dynamic Adapter (HyDA) instead of full model fine-tuning. This allows for efficient adaptation with fewer parameters [101]. Apply strong regularization and data augmentation. |
Experimental Protocols for BFM Validation
Protocol: Benchmarking Decoder Robustness to Simulated Non-Stationarity
This protocol outlines a method to quantitatively evaluate and compare different decoding algorithms under controlled non-stationary conditions, using BFMs as a benchmark.
1. Objective: To assess the robustness of a BFM-based decoder against traditional decoders (OLE, KF, RNN) when faced with simulated recording degradation and neuronal variance [89].
2. Materials & Data:
- Base Dataset: Neural data (e.g., spike signals) with corresponding kinematic data (e.g., from a 2D center-out task) [89].
- Simulation Model: A Population Vector (PV) model to synthesize spike data based on kinematic input [89].
- Decoders: The BFM under test, along with OLE, KF, and RNN decoders for comparison.
3. Methodology:
- Step 1: Simulate Non-Stationarity. Systematically modify the simulated spike data to introduce specific types of non-stationarity across multiple "sessions" [89]:
- Recording Degradation: Linearly decrease the Mean Firing Rate (MFR) and Number of Isolated Units (NIU).
- Neuronal Variance: Apply random shifts to the neural Preferred Directions (PDs).
- Step 2: Train Decoders. Train all decoders on the first (least degraded) session.
- Step 3: Test with Two Schemes. Evaluate performance on subsequent, more degraded sessions using two training schemes [89]:
- Static Scheme: Keep decoder parameters fixed.
- Retrained Scheme: Retrain the decoder on data from each target session.
- Step 4: Quantitative Analysis. Measure decoding performance (e.g., correlation coefficient, success rate) for each decoder and training scheme across all sessions.
4. Expected Outcome: The experiment will generate data showing how much performance degrades for each decoder as non-stationarity increases. A robust BFM should maintain higher performance under the static scheme and adapt more efficiently with the retrained scheme compared to traditional models.
Protocol: Validating a BFM with Domain Adaptation for Cross-Subject Transfer
This protocol tests the effectiveness of integrating Domain Adaptation (DA) techniques with a BFM to overcome the challenge of cross-subject variability.
1. Objective: To demonstrate that a BFM, when combined with DA, can achieve higher decoding accuracy for a new subject with minimal labeled data compared to a BFM without DA.
2. Materials:
- BFM: A pre-trained Brain Foundation Model.
- Data: A multi-subject neural dataset. Designate one or several subjects as the source domain, and one new subject as the target domain.
- DA Method: A feature-based DA algorithm (e.g., a method that minimizes Maximum Mean Discrepancy - MMD).
3. Methodology:
- Step 1: Feature Extraction. Use the BFM to extract high-level features from the neural data of both the source and target subjects.
- Step 2: Domain Alignment. Apply the DA algorithm to the extracted features. The goal is to project the features from both the source and target domains into a shared, domain-invariant feature space by minimizing a distribution distance metric like MMD [8].
- Step 3: Classifier Training. Train a simple classifier (e.g., linear classifier) on the aligned features from the source domain.
- Step 4: Evaluation. Test the classifier directly on the aligned features from the target subject. Compare the performance against a baseline where the BFM and classifier are used without DA.
4. Expected Outcome: The BFM with DA should show significantly improved performance on the target subject, demonstrating its utility as a validation benchmark for generalizable neural decoding algorithms.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Tools and Datasets for BFM Research in Neural Decoding
| Item Name | Type | Primary Function in Research | Example / Source |
|---|---|---|---|
| UK Biobank | Large-scale Biomedical Dataset | Provides massive volumes of brain imaging data (e.g., MRI) for self-supervised pre-training of BFMs, forming the foundational knowledge base [102]. | UK Biobank Dataset |
| BraTS (Brain Tumor Segmentation) | Benchmarking Challenge & Dataset | Serves as a key downstream task for fine-tuning and evaluating the performance of BFMs on specific, clinically relevant problems like brain tumor segmentation [102]. | BraTS Challenge |
| Hypergraph Dynamic Adapter (HyDA) | Algorithmic Module / Software | A lightweight adapter that enables efficient fine-tuning of BFMs; it uses hypergraphs to fuse multi-modal data and dynamically generates patient-specific parameters for personalized adaptation [101]. | [101] |
| SAM-Brain3D | Pre-trained Model | An example of a brain-specific foundation model pre-trained on over 66,000 image-label pairs, capable of segmenting diverse brain targets and adaptable to classification tasks [101]. | [101] |
| Non-Stationarity Simulation Framework | Computational Model | A tool (e.g., based on the Population Vector model) to systematically generate neural data with controlled levels of degradation (MFR, NIU) and neuronal variance (PDs) for controlled robustness testing [89]. | [89] |
Conclusion
Adaptive decoding algorithms represent a paradigm shift in neural signal processing, directly addressing the fundamental challenge of non-stationarity to unlock more accurate and clinically viable applications. The synthesis of Bayesian methods, adaptive transformers, and specialized algorithms like ADA provides a powerful toolkit for dynamic brain state decoding. While significant progress has been made in methodological innovation and validation, future work must focus on enhancing computational efficiency, improving model interpretability for clinical adoption, and facilitating seamless integration with real-time neurotherapeutic systems. The convergence of these adaptive algorithms with large-scale Brain Foundation Models and AI-driven clinical trial designs holds immense promise for accelerating the development of personalized diagnostics, closed-loop neuromodulation therapies, and high-performance neural prostheses, ultimately translating complex neural data into tangible patient benefits.
References
- 1. Non-stationary signal - (Intro to Electrical Engineering) [https://fiveable.me/key-terms/introduction-electrical-systems-engineering-devices/non-stationary-signal]
- 2. Real-Time Decoding of Nonstationary Neural Activity in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4484753]
- 3. frequency - Why is doing fft on a non - stationary a problem? signal [https://dsp.stackexchange.com/questions/44136/why-is-doing-fft-on-a-non-stationary-signal-a-problem]
- 4. Non Sinusoidal and Non Stationary Properties of Neural ... [https://sapienlabs.org/non-sinusoidal-and-non-stationary-properties-of-neural-signals/]
- 5. Nonstationary Signal - an overview [https://www.sciencedirect.com/topics/computer-science/nonstationary-signal]
- 6. Navigating Non with FFCS Analysis - FasterCapital Stationary Signals [https://fastercapital.com/content/Navigating-Non-Stationary-Signals-with-FFCS-Analysis.html]
- 7. Signal, noise, and variation in neural and sensory-motor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4824642/]
- 8. A survey of neural signal decoding based on domain ... [https://www.sciencedirect.com/science/article/abs/pii/S0925231225023252]
- 9. Machine Learning - Limitations [https://www.tutorialspoint.com/machine_learning/machine_learning_limitations.htm]
- 10. Subject-adaptive SSVEP decoding based on time– ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809425006524]
- 11. An adaptive transformer-based framework for advanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12528197/]
- 12. Machine learning based brain signal decoding for intelligent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10521329/]
- 13. Business Analytics & Machine Learning 2025: Key Trends [https://kanerika.com/blogs/machine-learning-for-business-analytics/]
- 14. A decoding algorithm for temporally-variable brain responses [https://www.sciencedirect.com/science/article/pii/S2001037025004465]
- 15. Hierarchical attention enhanced deep learning achieves ... [https://www.nature.com/articles/s41598-025-17922-1]
- 16. Super-resolution time-frequency decomposition with ... [https://www.sciencedirect.com/science/article/pii/S0031320325012610]
- 17. Five Predictions for Clinical Research in 2025 [https://www.muralhealth.com/blog/five-predictions-for-clinical-research-in-2025]
- 18. The Future of Clinical Trials: 2025 Outlook [https://signanthealth.com/resources/blog/the-future-of-clinical-trials-2025-trends]
- 19. When Is Simultaneous Recording Necessary? A Guide for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8276697/]
- 20. Getting to know EEG artifacts and how to handle them in ... [https://pressrelease.brainproducts.com/eeg-artifacts-handling-in-analyzer/]
- 21. Concordance between distributed EEG source localization ... [https://www.sciencedirect.com/science/article/abs/pii/S1053811907006994]
- 22. Avoiding EEG Recording Failures During a Clinical Trial [https://lifelinesneuro.com/eeg-recording-failures/]
- 23. fMRI functional networks for EEG source imaging - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6869924/]
- 24. Multimodal single-neuron, intracranial EEG, and fMRI brain ... [https://www.nature.com/articles/s41597-024-03029-1]
- 25. Adaptive Decoding for Brain-Machine Interfaces through ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3335277/]
- 26. A high-performance speech neuroprosthesis [https://www.nature.com/articles/s41586-023-06377-x]
- 27. Troubleshooting & Clarification (Beginner) [https://openbci.com/forum/index.php?p=/discussion/3689/troubleshooting-clarification-beginner]
- 28. Statistical innovations in clinical trial design with a focus on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12627888/]
- 29. Bayesian Methods to Optimise Trial Design in Clinical ... [https://marksmanhealthcare.com/2025/05/13/bayesian-methods-to-optimise-trial-design-in-clinical-development/]
- 30. A Beginner's Guide to the Bayesian Neural Network [https://www.coursera.org/articles/bayesian-neural-network]
- 31. Tutorial 1: Bayes with a binary hidden state [https://compneuro.neuromatch.io/tutorials/W3D1_BayesianDecisions/student/W3D1_Tutorial1.html]
- 32. Adaptive Artifact Removal From Intracortical Channels for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6476983/]
- 33. and LDA-based decoding of EEG signals [https://www.sciencedirect.com/science/article/pii/S1053811925003076]
- 34. Full-Process Adaptive Encoding and Decoding Framework ... [https://www.mdpi.com/2072-4292/16/9/1529]
- 35. Accelerating LLM Decoding with Adaptive Layer Parallelism [https://arxiv.org/html/2506.03700v1]
- 36. A modified transformer based on adaptive frequency ... [https://www.nature.com/articles/s41598-025-18187-4]
- 37. The influence of non-stationarity of spike signals on ... [https://www.frontiersin.org/journals/computational-neuroscience/articles/10.3389/fncom.2023.1135783/full]
- 38. Increasing Transformer Model Efficiency Through Attention ... [https://medium.com/data-science/increasing-transformer-model-efficiency-through-attention-layer-optimization-fefa6f87b1d6]
- 39. Advanced Attention Mechanisms in Transformer LLMs | by M [https://pub.towardsai.net/advanced-attention-mechanisms-in-transformer-llms-44cac04ec356]
- 40. Determination of the Time-frequency Features for Impulse ... [https://link.springer.com/article/10.1007/s12021-024-09698-y]
- 41. Single-trial characterization of neural rhythms: Potential ... [https://www.sciencedirect.com/science/article/pii/S105381191930922X]
- 42. Trial-to-Trial Variability of Spiking Delay Activity in Prefrontal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8549532/]
- 43. Personalized preictal EEG pattern characterization [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1526963/full]
- 44. Investigating the Impact of Rational Dilated Wavelet ... [https://arxiv.org/html/2510.09242]
- 45. Deep learning for network security: an Attention-CNN ... [https://www.nature.com/articles/s41598-025-07706-y]
- 46. IntrusionX: A Hybrid Convolutional–LSTM Deep Learning ... [https://arxiv.org/html/2510.00572v1]
- 47. A Hybrid Convolutional Neural Network-Long Short-Term ... [https://pubmed.ncbi.nlm.nih.gov/41225966/]
- 48. CNN-LSTM Hybrid Model for AI-Driven Prediction of ... [https://arxiv.org/abs/2505.23879]
- 49. A Robust Hybrid CNN–LSTM Model for Predicting Student ... [https://www.mdpi.com/2673-6470/5/2/16]
- 50. Automated drug design for druggable target identification ... [https://www.nature.com/articles/s41598-025-18091-x]
- 51. A model for epileptic EEG detection and recognition based ... [https://www.nature.com/articles/s41598-025-17256-y]
- 52. Motor imagery decoding network with multisubject dynamic ... [https://braininformatics.springeropen.com/articles/10.1186/s40708-025-00267-w]
- 53. Enhancing motor imagery decoding via transfer learning [https://www.sciencedirect.com/science/article/pii/S2352648322000733]
- 54. Exploring EEG-based motor imagery decoding: a dual ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10937463/]
- 55. A Case Study on Epilepsy Diagnosis Using EEG Signals [https://pubmed.ncbi.nlm.nih.gov/38507373/]
- 56. Automated-Seizure-Detection-Using-Machine-Learning [https://aesnet.org/abstractslisting/automated-seizure-detection-using-machine-learning--a-tool-for-better-patient-care]
- 57. Real Time Computation - an overview [https://www.sciencedirect.com/topics/computer-science/real-time-computation]
- 58. Non-stationary neural signal to image conversion ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10079945/]
- 59. 11 Common Computer Issues (and How to Fix Them) [https://www.rednightconsulting.com/11-common-computer-issues-fix/]
- 60. How to troubleshoot available resources errors in Excel [https://learn.microsoft.com/en-us/troubleshoot/microsoft-365-apps/excel/available-resources-errors]
- 61. Tackling abrupt transitions in aliased non-stationary signals [https://www.sciencedirect.com/science/article/pii/S2773186325000076]
- 62. How to Fix Overfitting and Underfitting | by Tahir | Oct, 2025 [https://medium.com/@tahirbalarabe2/how-to-fix-overfitting-and-underfitting-4cf8f383ea96]
- 63. What is Overfitting and How to Avoid Overfitting in Neural ... [https://pub.towardsai.net/what-is-overfitting-and-how-to-avoid-overfitting-in-neural-networks-17508fc37f0c]
- 64. Optimizing high dimensional data classification with a ... [https://www.nature.com/articles/s41598-025-08699-4]
- 65. The Relationship Between High Dimensionality and Overfitting [https://vtiya.medium.com/the-relationship-between-high-dimensionality-and-overfitting-5bca0967b60f]
- 66. Boosting neural network performance for high dimensional ... [https://www.sciencedirect.com/science/article/abs/pii/S0167865525003551]
- 67. Managing High-Dimensional Data in Machine Learning [https://www.geeksforgeeks.org/machine-learning/managing-high-dimensional-data-in-machine-learning/]
- 68. Enhancing the decoding accuracy of EEG signals by ... [https://www.nature.com/articles/s41598-022-07992-w]
- 69. Electroencephalogram-based pharmacodynamic measures [https://pmc.ncbi.nlm.nih.gov/articles/PMC3637024/]
- 70. An automated drug dependence detection system based ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482523003189]
- 71. 9 Common Data Quality Issues to Fix in 2025 [https://atlan.com/data-quality-issues/]
- 72. Neural signals, machine learning, and the future of inner ... [https://www.frontiersin.org/journals/human-neuroscience/articles/10.3389/fnhum.2025.1637174/full]
- 73. A Robust Multi-Scale Framework with Test-Time ... [https://arxiv.org/html/2509.24700v1]
- 74. Impact of Data Concerns in Neurological Clinical Trials [https://signanthealth.com/resources/blog/impact-of-data-concerns-in-neurological-clinical-trials-why-quality-matters]
- 75. Spiking neural networks for EEG signal analysis using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12572938/]
- 76. Wavelet-Based Denoising Strategies for Non-Stationary ... [https://www.mdpi.com/2079-9292/14/16/3190]
- 77. Blinding in Clinical Trials: Seeing the Big Picture - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8308085/]
- 78. Blinding: Who, what, when, why, how? - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2947122/]
- 79. Blinded Independent Central Review (BICR) in Clinical Trials [https://collectiveminds.health/articles/blinded-independent-central-review-bicr-complete-guide-for-clinical-trials]
- 80. Enhancing open clinical trials through blinded evaluations [https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-023-07652-y]
- 81. Blinding in pharmaceutical clinical trials: An overview of ... [https://www.sciencedirect.com/science/article/abs/pii/S1551714415300185]
- 82. Towards decoding motor imagery from EEG signal using ... [https://www.nature.com/articles/s41598-025-05423-0]
- 83. ADA: A decoding algorithm for temporally-variable brain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12657816/]
- 84. vs ROC AUC vs F vs PR AUC: Which Evaluation... 1 Score Accuracy [https://neptune.ai/blog/f1-score-accuracy-roc-auc-pr-auc]
- 85. Evaluation metrics and statistical tests for machine learning [https://www.nature.com/articles/s41598-024-56706-x]
- 86. On evaluation metrics for medical applications of artificial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8993826/]
- 87. 5 Key Metrics: Boosting Performance with Cross- Validation Folds Study [https://www.numberanalytics.com/blog/5-key-metrics-boosting-performance-cross-validation]
- 88. machine learning - What could explain a much higher F in... 1 score [https://datascience.stackexchange.com/questions/44206/what-could-explain-a-much-higher-f1-score-in-comparision-to-accuracy-score]
- 89. The influence of non-stationarity of spike signals on decoding ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10213332/]
- 90. Machine Learning for Neural Decoding [https://www.eneuro.org/content/7/4/ENEURO.0506-19.2020]
- 91. Tutorial 1: Decoding Neural Responses [https://compneuro.neuromatch.io/tutorials/W1D5_DeepLearning/student/W1D5_Tutorial1.html]
- 92. Classification of non-stationary neural signals [https://www.sciencedirect.com/science/article/abs/pii/S0165027098001101]
- 93. Optimizing the learning rate for adaptive estimation of neural ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1006168]
- 94. Unsupervised Adaptation of Brain-Machine Interface Decoders - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3499737/]
- 95. Adaptive Learned Belief Propagation for Decoding Error- ... [https://www.mdpi.com/1099-4300/27/8/795]
- 96. Unlocking Neural Secrets [https://www.numberanalytics.com/blog/ultimate-guide-neural-decoding-algorithms-bcis]
- 97. (PDF) Adaptive using local field potentials in... decoding [https://www.academia.edu/143247695/Adaptive_decoding_using_local_field_potentials_in_a_brain_machine_interface]
- 98. [DOCS] Enhanced Low-Density Parity-Check (LDPC) Decoding via... [https://gist.github.com/freederia/cb7992e0fe9c91fe3b582f279d558f81]
- 99. Systematic Review of Advanced Algorithms for Brain Mapping ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12650610/]
- 100. Brain Foundation Models: A Survey on Advancements in ... [https://arxiv.org/html/2503.00580v1]
- 101. Brain Foundation Models with Hypergraph Dynamic ... [https://www.sciencedirect.com/science/article/pii/S0031320325012580]
- 102. lab-smile/BrainSegFounder - BrainFounder [https://github.com/lab-smile/BrainSegFounder]